Clear Sky Science · en
Raloxifene relaxes the rat aorta via a mitochondria-dependent mechanism
Why keeping blood vessels flexible matters
As we age, our blood vessels can become stiffer and more prone to narrowing, raising the risk of high blood pressure, heart attacks, and strokes. Many women take the drug raloxifene to protect their bones after menopause, and doctors have noticed that it also seems to help the heart and blood vessels. This study asks a simple but important question: how, exactly, does raloxifene help blood vessels relax? The answer turns out to involve the tiny “power stations” inside cells—mitochondria—and the reactive molecules they produce.
A bone drug with hidden heart benefits
Raloxifene is best known as a treatment to prevent osteoporosis and lower the risk of certain breast and uterine cancers in postmenopausal women. It acts on estrogen receptors in a tissue-specific way, blocking estrogen’s action in some organs while mimicking it in others. Earlier work showed that raloxifene can lower blood pressure in hypertensive rats and relax arteries and veins in several species. Proposed explanations included boosting nitric oxide, blocking calcium entry into muscle cells, and dampening inflammation. However, these ideas did not fully explain how the drug helps blood vessels widen, especially in the short term.
Testing how rat arteries respond
The researchers focused on the main body artery (the aorta) from male rats and studied small ring segments of this vessel in the lab. First, they tightened the rings using phenylephrine, a compound that causes blood vessels to constrict. Then they added increasing amounts of raloxifene and measured how much the tension in the rings dropped. Removing the delicate inner lining of the vessel—the endothelium—reduced, but did not abolish, raloxifene’s relaxing effect. Blocking prostaglandins (with indomethacin) or estrogen receptors made little difference. This showed that while the vessel lining helps, raloxifene’s main relaxing action does not rely on classic estrogen signaling or common hormone-like messengers.
Spotlight on cellular power plants
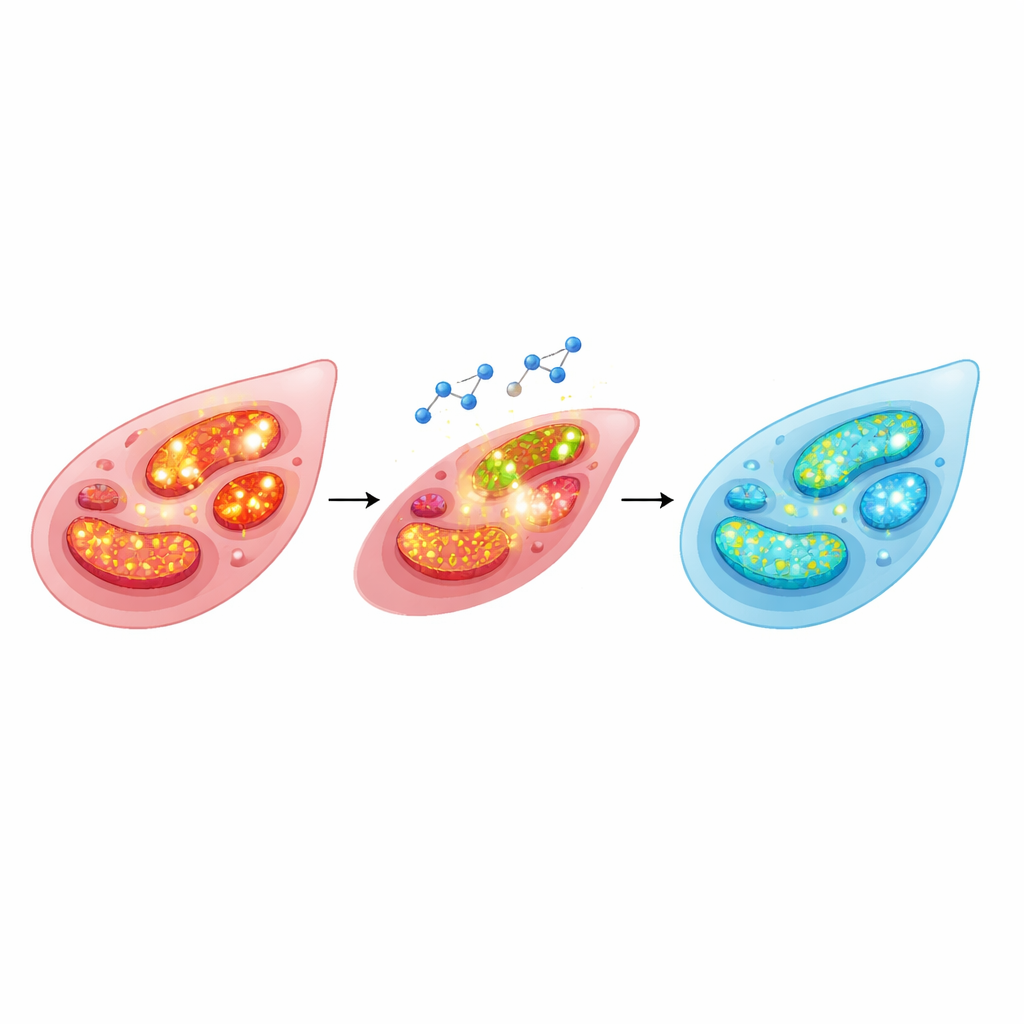
Attention then turned to reactive oxygen species—short-lived, chemically active forms of oxygen often described as “oxidative stress.” These molecules are produced by several enzyme systems, including a membrane enzyme complex (NADPH oxidase) and the mitochondrial energy chain. In arteries stripped of their inner lining, the team used drugs that reduce reactive oxygen species from different sources. Blocking NADPH oxidase only modestly weakened phenylephrine-induced tightening, whereas targeting mitochondria with rotenone or a mitochondria-focused antioxidant clearly reduced constriction. When they combined these approaches with raloxifene, they found that interfering with the mitochondrial machinery strongly blunted raloxifene’s ability to relax the vessel, pointing to mitochondrial reactive oxygen species as a central player.
How cells calm down their inner sparks
To see what happens inside individual vascular smooth muscle cells, the cells that actually squeeze the artery, the scientists used fluorescent dyes that light up in the presence of reactive oxygen species or when mitochondrial voltage is high. Phenylephrine caused a jump in both total and mitochondrial reactive oxygen species, and raised the mitochondrial membrane potential—signs that the cellular power plants were working in an overcharged, stress-generating mode. Raloxifene reversed these changes, bringing both reactive oxygen levels and mitochondrial potential back down. The team also examined a protein called UCP2, which acts as a built-in “brake” to gently leak the energy gradient across the mitochondrial membrane and thereby limit reactive oxygen production. Phenylephrine lowered UCP2 levels, but raloxifene pretreatment preserved them, helping keep the mitochondrial environment more stable.
What this means for future treatments
Overall, the study shows that raloxifene helps rat arteries relax in large part by dialing down stress signals coming from mitochondria in the vessel wall. By maintaining the activity of UCP2 and modestly loosening the mitochondrial energy gradient, raloxifene reduces the buildup of harmful reactive oxygen species that would otherwise promote vessel tightening and damage. For lay readers, the key message is that a drug already used to protect bones may also protect blood vessels by calming the “sparks” inside cellular power plants. This mitochondrial pathway could become a target for new treatments aimed at preventing or easing vascular diseases driven by oxidative stress.
Citation: Ji, KD., Song, B., Shen, WL. et al. Raloxifene relaxes the rat aorta via a mitochondria-dependent mechanism. Sci Rep 16, 14175 (2026). https://doi.org/10.1038/s41598-026-44551-z
Keywords: raloxifene, mitochondria, reactive oxygen species, vascular relaxation, UCP2