Clear Sky Science · en
Tumor-specific cytokine therapy mediated by engineered Salmonella with a synthetic protein delivery system
Turning Germs into Cancer Fighters
Cancer doctors have long known that certain immune-boosting drugs called cytokines can help the body attack tumors, but they are so powerful that they often harm healthy tissues, too. This study explores an unusual workaround: using a weakened form of the food‑poisoning bacterium Salmonella as a tiny delivery vehicle that sneaks into tumors and releases a designer immune signal only where it is needed, aiming to supercharge local defenses while sparing the rest of the body.
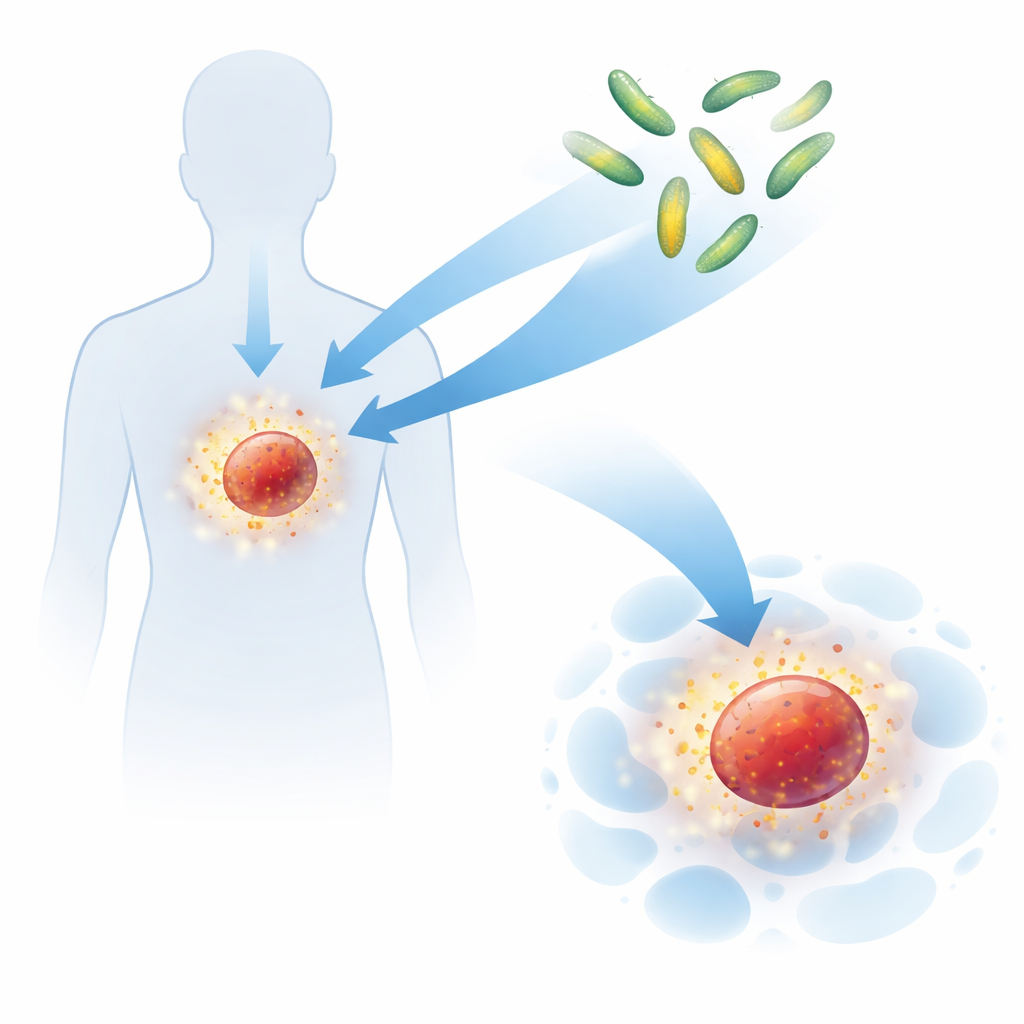
Why Existing Immune Signals Are Hard to Use
Cytokines act like molecular text messages that tell immune cells when to rest and when to fight. Drugs based on natural cytokines such as interleukin‑2 and interleukin‑15 can strongly activate cancer‑killing T cells and natural killer cells, but they come with serious drawbacks: they circulate throughout the body, cause dangerous side effects, and break down quickly in the bloodstream. A newer, computer‑designed cytokine called Neoleukin‑2/15 was created to keep the good effects—waking up cancer‑fighting cells—while avoiding pathways that switch on suppressive immune cells. In mice, this designer protein worked well, but in human trials it still caused off‑target problems, suggesting that the real issue was not just the molecule itself, but where and how it was delivered.
Recruiting Bacteria That Naturally Home to Tumors
Certain bacteria naturally prefer the harsh, oxygen‑poor environment found inside tumors and tend to multiply there while being cleared from healthy organs. The researchers took advantage of this behavior by working with Salmonella, a bacterium that can be genetically tamed so it no longer invades or damages normal tissues. They deleted two key genetic regions that normally enable Salmonella to invade host cells, creating a highly weakened strain that still migrates to tumors. Into this strain they introduced a carefully engineered protein export system and a separate set of genes encoding the designer cytokine Neo‑2/15, turning each bacterium into a controllable factory for making and releasing the immune‑stimulating protein directly inside tumor tissue.
Building a Switchable Protein Delivery Machine
To move Neo‑2/15 out of the bacterial cell and into the tumor surroundings, the team rewired a molecular syringe that Salmonella usually uses during infection, known as a type 3 secretion system. They compacted dozens of genes onto a single plasmid—a circular DNA piece—so that the entire secretion machine could be turned on by a common antibiotic, doxycycline. A short protein “tag” from a natural Salmonella protein was fused to Neo‑2/15 so that, when the system was induced, the bacteria pumped this tagged cytokine out into the surrounding fluid. In lab tests, adding doxycycline triggered steady secretion of the fusion protein without killing the bacteria, and standard biochemical assays confirmed that the released product was present at measurable levels and retained the expected size and structure.
Proving the Designer Signal Still Works
Releasing a cytokine is only useful if it still speaks the right language to immune cells. The team tested this by exposing a mouse T‑cell line, which normally depends on cytokines to survive and multiply, to the bacterial culture fluid containing the secreted Neo‑2/15 fusion. After concentrating this fluid to reach biologically relevant doses, the researchers found that T cells grew nearly as well as they did in the presence of standard interleukin‑2 or purified Neo‑2/15 made in a more conventional way. Even when the exact amount of cytokine in the fluid was low, the combination of the designer signal and other bacterial components still boosted T‑cell numbers, showing that the fusion protein remained functional after secretion.
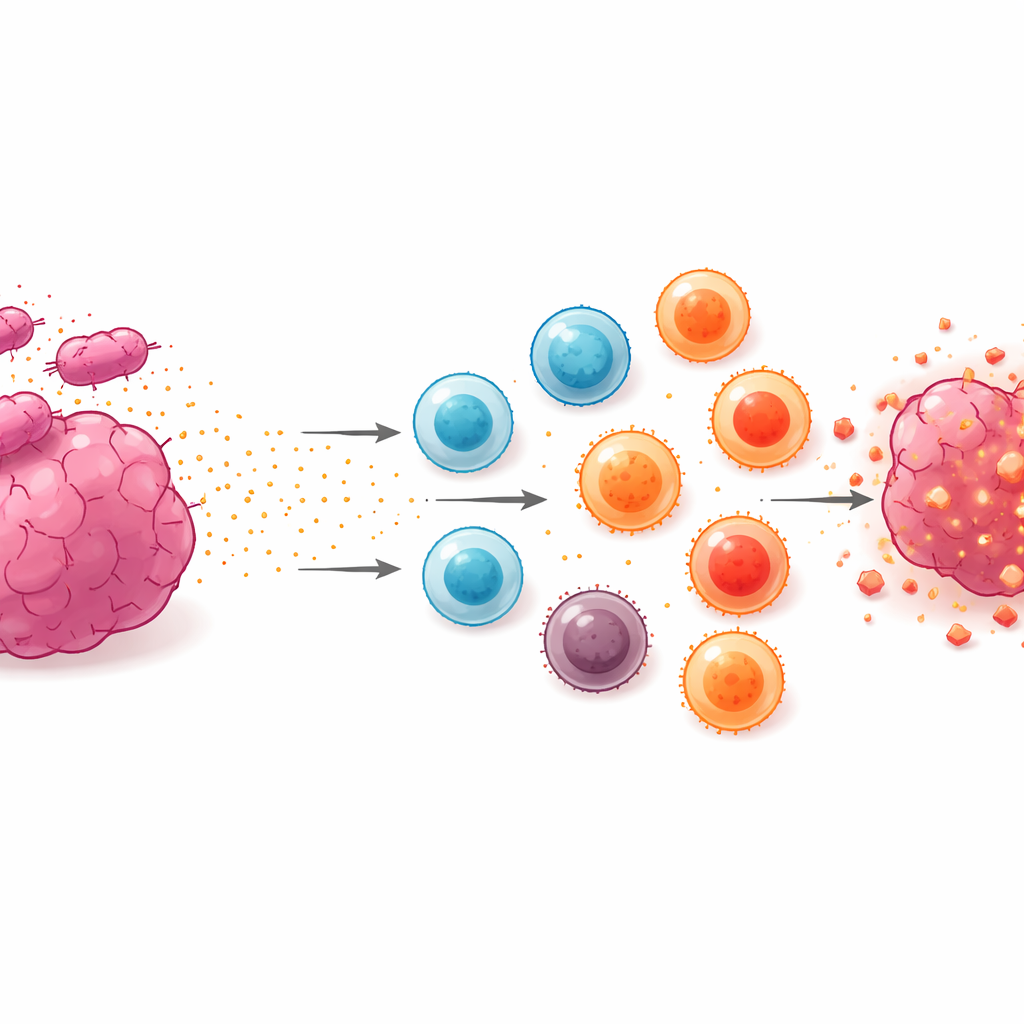
Slowing Tumors and Extending Survival in Mice
The ultimate test was whether this bacterial delivery strategy could help animals fight cancer. Mice were implanted with colon cancer cells and, once tumors had formed, injected with the weakened Salmonella carrying the secretion system and Neo‑2/15 cargo. When the doxycycline switch was left off, tumors continued to grow, though the mice lived slightly longer than untreated controls, likely because some cytokine leaked out as bacteria died inside the tumor. When the switch was turned on and secretion was fully activated, tumor growth slowed markedly, and the treated mice survived roughly twice as long as untreated animals, all without obvious liver or kidney damage in blood tests. The benefit was not a complete cure, in part because one of the plasmids carrying the secretion machinery was gradually lost over time inside the body, limiting how long high‑level cytokine release could be sustained.
What This Could Mean for Future Cancer Care
This work shows that live, weakened bacteria can be re‑engineered into programmable couriers that carry powerful immune‑boosting proteins straight into tumors, activating local defenses while limiting whole‑body exposure. Although the treated mice were not fully cured, the approach slowed tumor growth and extended life without major toxicity, suggesting that further refinements—such as making the delivery system more stable or combining it with other treatments—could make it more effective. For non‑specialists, the main message is that microbes once known only as enemies might soon be harnessed as precision tools, turning the body’s own immune system against cancer in a far more targeted way.
Citation: Ha, J., Song, M. Tumor-specific cytokine therapy mediated by engineered Salmonella with a synthetic protein delivery system. Sci Rep 16, 14240 (2026). https://doi.org/10.1038/s41598-026-44265-2
Keywords: bacteria-based cancer therapy, tumor-targeted cytokine delivery, engineered Salmonella, cancer immunotherapy, synthetic protein secretion