Clear Sky Science · en
A new technique inducing mitral valve regurgitation as an experimental porcine model of volume-overload induced heart failure
Why this pig heart study matters
Heart failure is one of the leading causes of illness and death worldwide, and many cases are driven by a leaky heart valve that forces the heart to pump extra blood over and over again. Before new drugs, devices, or surgical approaches can be safely tested in people, scientists need animal models that closely mimic what happens in human hearts. This study introduces a simple but powerful way to create a stable form of valve-related heart failure in pigs, whose hearts behave much like our own, opening the door to better testing of future treatments.
A leaky gate in the heart’s main pump
In many patients, heart failure develops because the mitral valve, which sits between the heart’s main pumping chamber and the chamber that feeds it, no longer closes tightly. Each heartbeat then sends some blood backward instead of forward, creating a chronic volume overload that gradually stretches and weakens the heart muscle. Existing large-animal models of this problem have drawbacks: they often rely on damaging heart arteries to make the valve leak, which introduces heart attack–like injury and makes results less predictable. The authors set out to build a model focused purely on the leak itself, without additional hidden damage to the heart.
A direct but controlled way to make the valve leak
The team worked with 25 healthy Piétrain pigs, a common farm breed whose heart structure and function resemble those of humans. Seventeen pigs underwent a procedure to create a strong leak across the mitral valve, while eight had a similar "sham" surgery without damaging the valve. Under general anesthesia and through an open-chest approach, the researchers gently opened the sac around the heart and inserted a custom-made tool through the tip of the left ventricle. Guided by high-quality ultrasound imaging placed directly on the heart surface, they hooked and cut one of the fine strings (chordae tendineae) that help the valve close, adjusting which string to cut until they achieved a strong, directed jet of backward blood flow. In the sham animals, the tool was introduced but the strings were left intact, so their valves continued to work normally.
Watching the heart adapt over four weeks
After surgery, all animals received careful pain control, antibiotics, and, in the leak group, water pills to relieve fluid buildup. The pigs were followed for four weeks, after which the team repeated the ultrasound scans and examined heart tissue under the microscope. In every pig with an induced leak, imaging confirmed a very severe valve problem with an eccentric jet of blood shooting back into the upper chamber. Compared to the sham group, these pigs developed much larger upper chambers and pumping chambers, as shown by marked increases in the volumes of the left atrium and left ventricle at the end of both filling and squeezing phases of the heartbeat. At the same time, key measures of pump performance, such as ejection fraction and fractional shortening, fell sharply, indicating that the hearts were already weakening.
Scars in the muscle and how this model compares
Tissue samples told a similar story. Hearts exposed to the valve leak had roughly double the amount of fibrous scar-like tissue in the main pumping chamber compared with sham hearts. This pattern—chamber enlargement, reduced pumping strength, and increased fibrosis—mirrors the early stages of human heart failure caused by mitral valve regurgitation. Importantly, both male and female pigs showed similar changes, and the leak was strong and uniform across nearly all animals, something many earlier models struggled to achieve. Because the method targets only the valve strings and avoids blocking heart arteries, it isolates the effects of volume overload without the confounding damage of a heart attack.
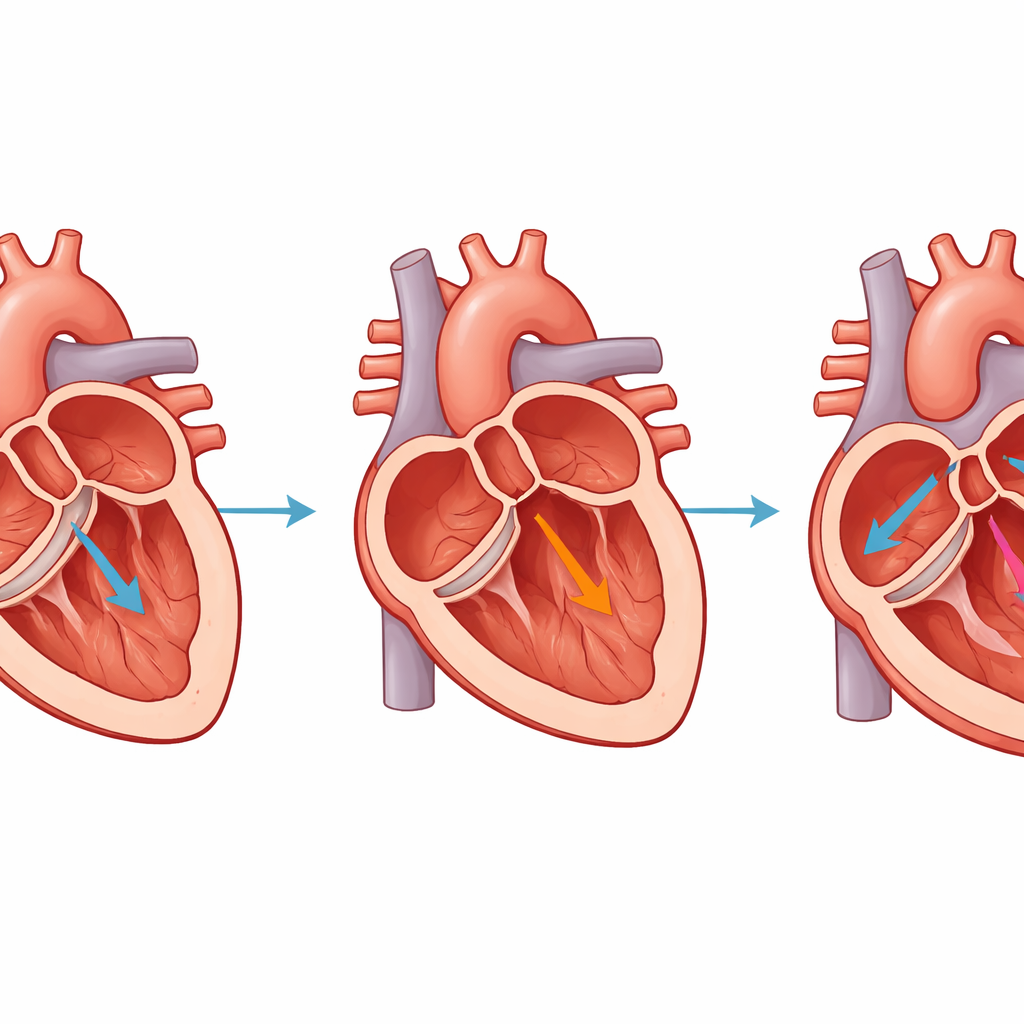
What this means for future treatments
In plain terms, the researchers have created a dependable way to make pig hearts develop the same kind of stretching, weakening, and scarring that human hearts experience when a major valve leaks badly. Within just four weeks, the animals progress to a clinically meaningful stage of heart failure driven purely by chronic volume overload. This makes the model especially useful for testing new imaging techniques, drugs, and devices aimed at either fixing the valve or protecting the heart muscle from long-term damage. By providing a cleaner and more controlled experimental setup, this pig model may help speed the path from laboratory insights to therapies that improve the lives of patients with valve-related heart failure.
Citation: Van Laer, S.L., Goovaerts, B., Laga, S. et al. A new technique inducing mitral valve regurgitation as an experimental porcine model of volume-overload induced heart failure. Sci Rep 16, 13500 (2026). https://doi.org/10.1038/s41598-026-43623-4
Keywords: heart failure, mitral valve leak, animal models, porcine cardiology, volume overload