Clear Sky Science · en
Extraction and characterization of a novel glycosylated naphthazarin pigment from mangrove Aspergillus unguis AUMC15225
Why Color from Nature Matters
From the clothes we wear to the foods we eat, synthetic dyes are everywhere—and many of them raise health and environmental concerns. This study explores a new way to get strong, stable color from a surprising source: a fungus living on mangrove tree roots in the Red Sea. The researchers discovered and carefully mapped the structure of a previously unknown red pigment that blends a classic dye-like core with a natural sugar, making it both water‑friendly and remarkably tough. Their work points toward future dyes that are safer for people and ecosystems while still standing up to heat, light and time.
A Hidden Factory on Mangrove Roots
Mangrove forests sit at the edge of land and sea, where roots are bathed in salty, shifting waters. These harsh conditions favor microbes with unusual chemistry, including fungi that make protective molecules to cope with stress. The team isolated one such fungus, Aspergillus unguis, growing on aerial roots in a Red Sea mangrove reserve. When they grew this fungus in a salty broth in the lab, the clear liquid slowly turned red after about nine days. The timing showed that the pigment is a “secondary” product—something the fungus makes not for basic growth, but likely for defense or survival—hinting at a specialized and potentially useful compound.
From Red Broth to Pure Pigment
To work with the pigment, the scientists first had to pull it out of the culture medium. Because the color stayed stubbornly in the water layer when mixed with oil‑like solvents, they knew it was highly polar—that is, naturally drawn to water rather than fats. After removing proteins and other impurities, they obtained a dark reddish‑brown powder that dissolved completely in high‑grade alcohol. This ease of extraction is an advantage over pigments locked inside cells, which require mechanical disruption. Early tests also showed the pigment was not mixed with common plant‑like chemicals such as phenols or flavonoids, but did include carbohydrate components, suggesting that a sugar was chemically attached to the colored core.
Color that Stays Strong
Many natural colors fade or shift when exposed to heat or extreme acidity and alkalinity, limiting their real‑world use. The new pigment proved unusually stubborn. It kept its color across a pH range from extremely acidic to strongly alkaline and withstood temperatures up to the boiling point of water, losing only a few percent of its intensity. Even after six months at room temperature, no visible change or settling was observed. These traits, combined with its water‑loving nature, make the pigment a strong candidate for applications where color stability is critical, from foods and textiles to cosmetics and coatings that must survive processing and storage.
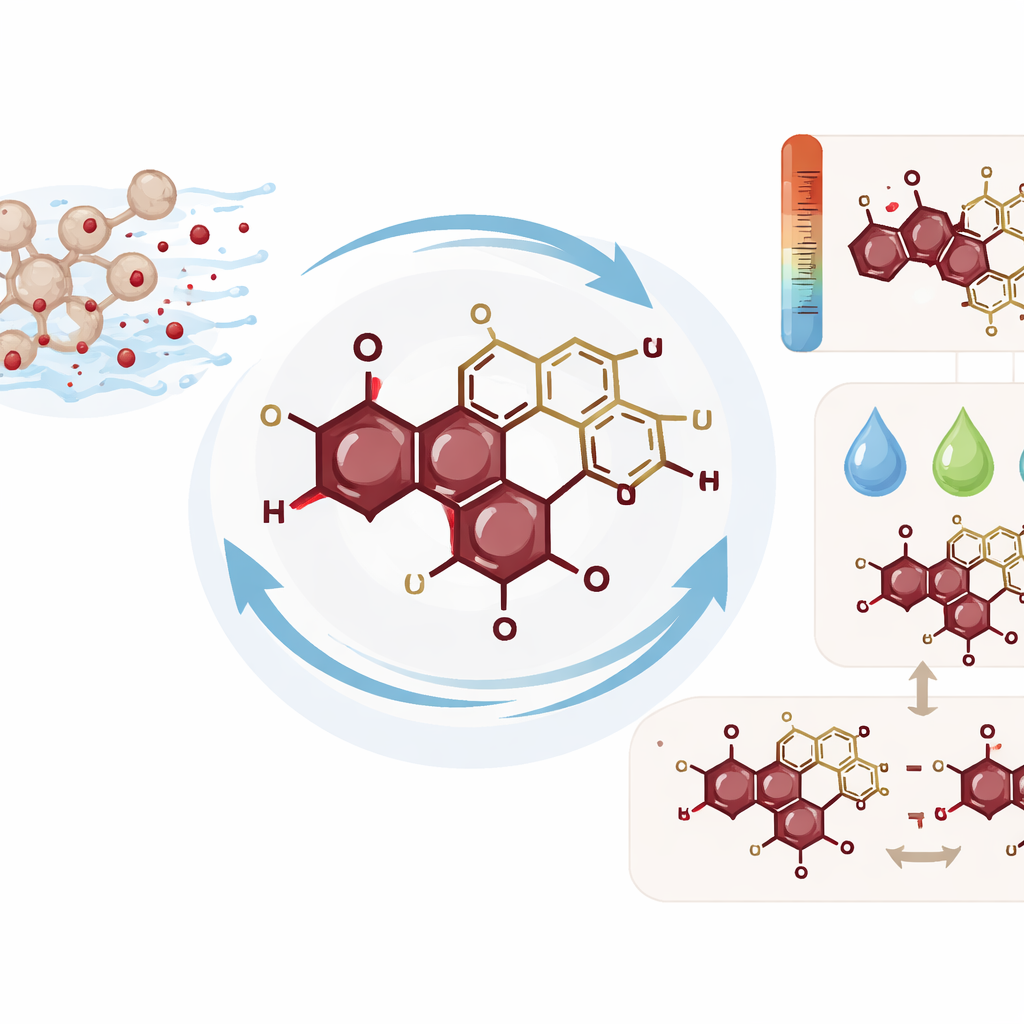
Unpacking the Pigment’s Architecture
To understand why the pigment behaves so well, the researchers turned to a toolkit of structural methods. Light‑absorption measurements showed a signature peak typical of a family of compounds known as naphthoquinones, long used as dyes and studied for medical potential. High‑performance liquid chromatography separated the pigment into two nearly identical forms, both carrying the same color but differing subtly in 3‑D arrangement. Infrared analysis, mass spectrometry and nuclear magnetic resonance then converged on a single architecture: a classic naphthazarin ring—the part that gives the deep red hue—decorated with a five‑carbon sugar called arabinose attached through an oxygen “bridge.” The final structure, named 2‑O‑β‑L‑arabinofuranosyl‑5,8‑dihydroxy‑1,4‑naphthoquinone, is the first of its kind reported from nature, and the two forms are best explained as stereoisomers—mirror‑like variants in how the sugar ring is twisted in space.
What This Discovery Could Lead To
By tracing this pigment from mangrove roots to molecular blueprint, the study opens a door to greener color technologies. The sugar attachment improves water solubility and likely shields the dye core, helping explain its stability and hinting at favorable biological behavior, such as lower nonspecific toxicity and better compatibility with living tissues. While the work stops short of testing biological effects or fully mapping the fungus’s pigment‑making pathway, it establishes mangrove‑associated fungi as a promising source of new natural dyes. In plain terms, the authors have found and decoded a tough, water‑friendly red color made by a marine‑influenced fungus—exactly the sort of ingredient that could one day help replace problematic synthetic dyes with safer, more sustainable alternatives.
Citation: Alkersh, B.M., Ghozlan, H.A., Sabry, S.A. et al. Extraction and characterization of a novel glycosylated naphthazarin pigment from mangrove Aspergillus unguis AUMC15225. Sci Rep 16, 11238 (2026). https://doi.org/10.1038/s41598-026-43500-0
Keywords: natural pigments, mangrove fungi, naphthoquinone, biobased dyes, fungal secondary metabolites