Clear Sky Science · en
Quantitative immunohistochemistry and the use of cellular calibrators for HER2 receptor number determination
Why counting cell signals matters
When doctors test a breast cancer sample for the marker HER2, the result can determine whether a patient receives powerful targeted drugs or not. Yet the standard staining test, called immunohistochemistry, still behaves more like an art than a measurement: different labs can get different answers, and very weak signals are especially easy to miss. This study shows how to turn that color stain into an actual number – by adding specially prepared cells that act as built‑in rulers for how much HER2 is present on each tumor cell.
From yes-or-no to real numbers
Most current stain-based tests classify HER2 in broad steps such as “0,” “1+,” “2+,” or “3+.” These categories are useful but crude; they do not tell how many HER2 receptors sit on each cancer cell, especially in the newly important “HER2-low” and “HER2-ultralow” groups. The authors argue that the main reason for high error rates is the lack of quantitative yardsticks within the staining process itself. Other lab tests, like blood-based antibody assays, routinely rely on carefully prepared standards and quality controls. Bringing the same discipline into tissue staining could make pathologists’ reads more consistent and allow much finer grading of HER2 levels.
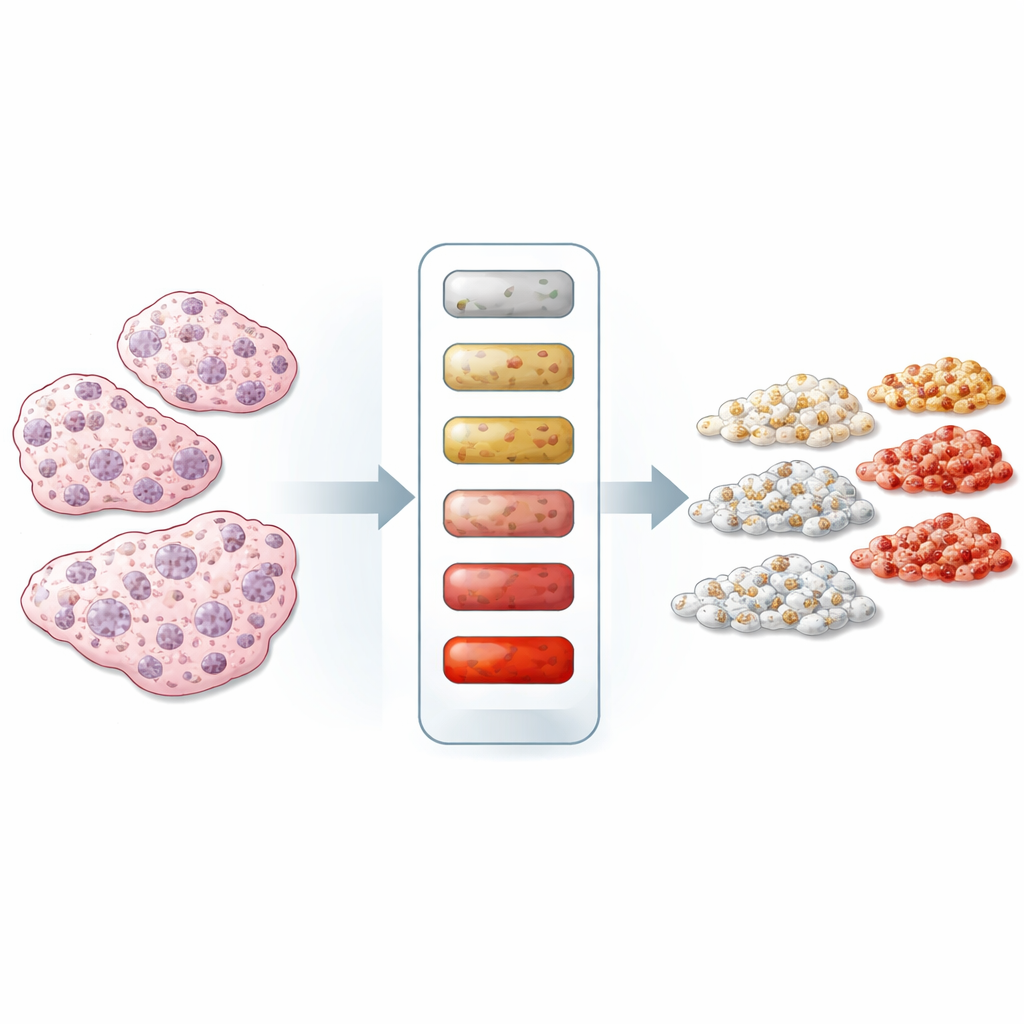
Building cell-based measuring sticks
To create such standards, the team chose a panel of breast cancer cell lines that naturally carry different amounts of HER2, ranging from almost none to very high. They used two independent technologies – an electrochemiluminescent immunoassay, which measures HER2 in broken‑open cells, and flow cytometry, which counts receptors on intact cells – to assign each line an average number of HER2 receptors per cell. Most cell lines agreed well between the two methods, confirming that their HER2 content was known with confidence, although the very highest-HER2 lines showed some disagreement between techniques. These characterized cell lines then became “cellular calibrators” that could be processed and stained just like patient tissue.
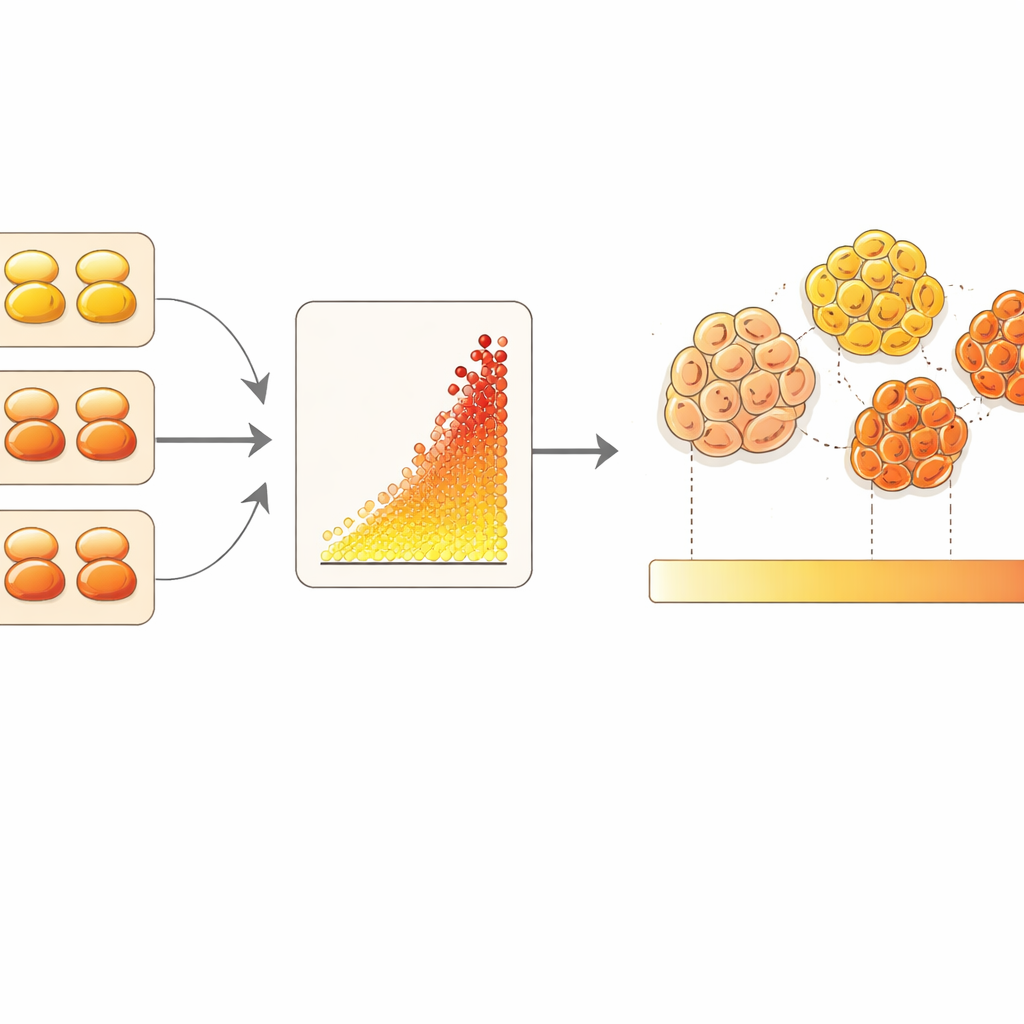
Turning microscope images into counts
The researchers embedded the calibrator cells into a wax block, cut sections, and stained them alongside a microarray of 85 breast cancer tumors using a sensitive fluorescent version of the HER2 test. A machine-learning image analysis tool was trained to recognize tumor cells based on a separate stain for structural proteins and to measure only the HER2 signal around those cells. By plotting the known receptor numbers of the calibrator cells against their measured brightness, the team built a calibration curve. They then used that curve to translate the average brightness of each patient’s tumor cells into an estimated number of HER2 receptors per cell, over a usable range of roughly ten to almost two hundred thousand receptors.
What the new numbers reveal
When they compared these quantitative HER2 counts with the standard clinical scores (using a commonly employed antibody called 4B5), the relationship was surprisingly inverted: tumors with higher measured receptor numbers sometimes carried lower traditional scores, and vice versa. Many samples labeled as “HER2 zero” actually showed clearly measurable HER2 levels with the new method. The authors caution that their assay is not yet fully validated and that tumor-to-tumor variability, differences in antibodies, and technical details could all contribute to the mismatch. Still, the findings highlight how semi‑quantitative categories can obscure a continuous spectrum of HER2 expression in real cancers.
Promise and hurdles for better cancer testing
Overall, the work demonstrates that using whole cells as calibrators can turn HER2 staining into a numerical test that more closely resembles other regulated laboratory assays. Because these calibrator cells mimic real tissue – they have membranes, internal structures, and can be stained in the same way – they provide a realistic yardstick for future quantitative methods. At the same time, the approach is demanding: it depends on well-characterized cell lines, careful cross-checking with multiple measurement platforms, and clear standards for accuracy and precision. The authors argue that if such quantitative immunohistochemistry is to guide treatment decisions, formal validation guidelines and proof of clinical benefit will be essential, but cell-based calibrators offer a practical path toward that goal.
Citation: McKinski, K., Chen, B. Quantitative immunohistochemistry and the use of cellular calibrators for HER2 receptor number determination. Sci Rep 16, 14573 (2026). https://doi.org/10.1038/s41598-026-42898-x
Keywords: HER2 breast cancer, immunohistochemistry, cellular calibrators, biomarker quantification, diagnostic assay quality