Clear Sky Science · en
NOX1/4 drives hepatic iron and lipid dysregulation, redox imbalance, and inflammation in ethanol-fed mice
Why this research matters for people who drink
Heavy drinking is known to damage the liver, but the reasons why some people progress from simple fat buildup to serious disease are still being uncovered. This study takes a close look at one pair of enzyme "spark plugs" in liver cells that help generate harmful oxidants and shows that blocking them with an experimental drug can calm several damaging processes at once in alcohol-exposed mice. The work hints at a future pill that might protect the liver by easing iron overload, fat buildup, and inflammation together, rather than tackling each problem separately.
A closer look at alcohol’s hidden liver damage
Alcoholic liver disease spans a spectrum from fatty liver to cirrhosis and liver cancer. The authors focus on four tightly linked troublemakers: excess iron stored in the liver, accumulation of fat droplets, chemical stress caused by reactive oxygen species, and chronic inflammation driven by immune cells. In people who drink heavily, alcohol weakens the gut barrier and alters gut microbes, allowing bacterial products to reach the liver and fan the flames of inflammation. At the same time, alcohol metabolism in the liver produces reactive molecules that injure cells and can trigger a form of iron-driven cell death called ferroptosis. Together, these processes create a vicious cycle that accelerates liver injury.
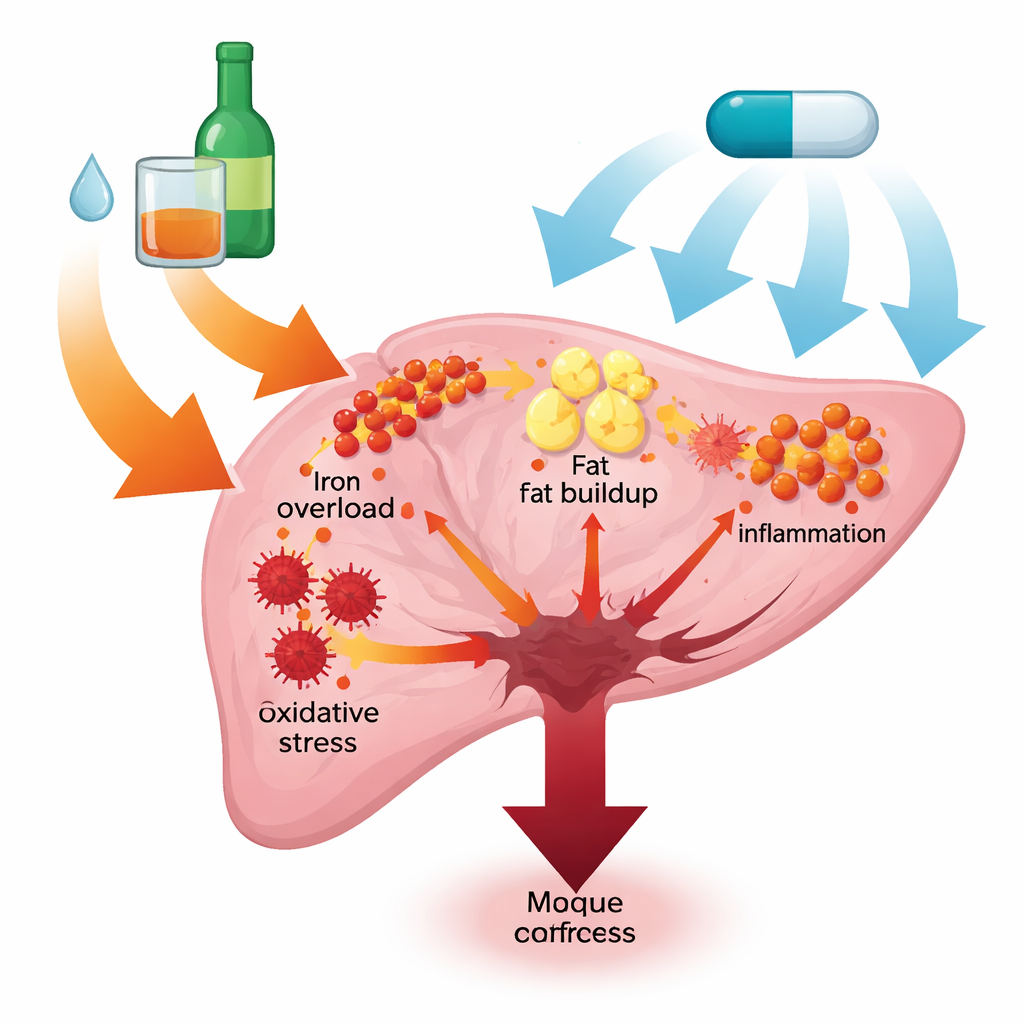
The enzyme pair that lights the fire
The study zooms in on two related enzymes, NOX1 and NOX4, which sit in cell membranes and produce reactive oxygen species. Using a standard mouse model of chronic-plus-binge alcohol exposure, the researchers found that alcohol sharply increased both the gene and protein levels of NOX1 and NOX4 in the liver. This surge was accompanied by classic signs of liver injury: higher blood levels of liver enzymes, ballooned and dying liver cells under the microscope, and clear evidence of iron accumulation and fat deposits in liver tissue. These findings suggest that NOX1/4 act as upstream igniters that connect alcohol exposure to widespread metabolic and inflammatory damage.
How blocking NOX1/4 cools down iron, fat, and stress
The team tested Setanaxib, a drug in clinical development that selectively blocks NOX1 and NOX4. Mice given Setanaxib along with alcohol showed much less liver injury than alcohol-only animals. Iron measurements revealed that the drug reduced both ferrous and ferric iron in the liver and shrank the area of iron staining, while lowering a key iron-storage protein. At the molecular level, Setanaxib appeared to rebalance iron traffic: it reduced levels of hepcidin, a hormone that traps iron in tissues, restored the iron exporter protein ferroportin, and lowered levels of transferrin receptor 1, which brings iron into cells. In parallel, oil-stained sections and blood tests showed that Setanaxib cut liver and blood triglycerides and free fatty acids, indicating relief from alcohol-induced fat overload.
Rebuilding the liver’s defenses and calming inflammation
Beyond iron and fat, Setanaxib helped repair the liver’s antioxidant shield. Alcohol alone depleted glutathione, the cell’s main protective molecule, increased its oxidized form, and raised markers of lipid peroxidation. It also suppressed the activity of Nrf2, a master switch that turns on antioxidant genes, leading to lower levels of protective proteins such as HO-1, SLC7A11, and GPX4, while boosting a stress marker called Prx2. Setanaxib reversed these changes, restoring glutathione balance and Nrf2 signaling and lowering oxidative damage markers. The drug also dampened the inflammatory response: it reduced levels of the chemoattractant MCP-1, cut down the number of infiltrating macrophages in liver tissue, and decreased both liver and blood levels of the pro‑inflammatory messengers IL‑1β and IL‑6.
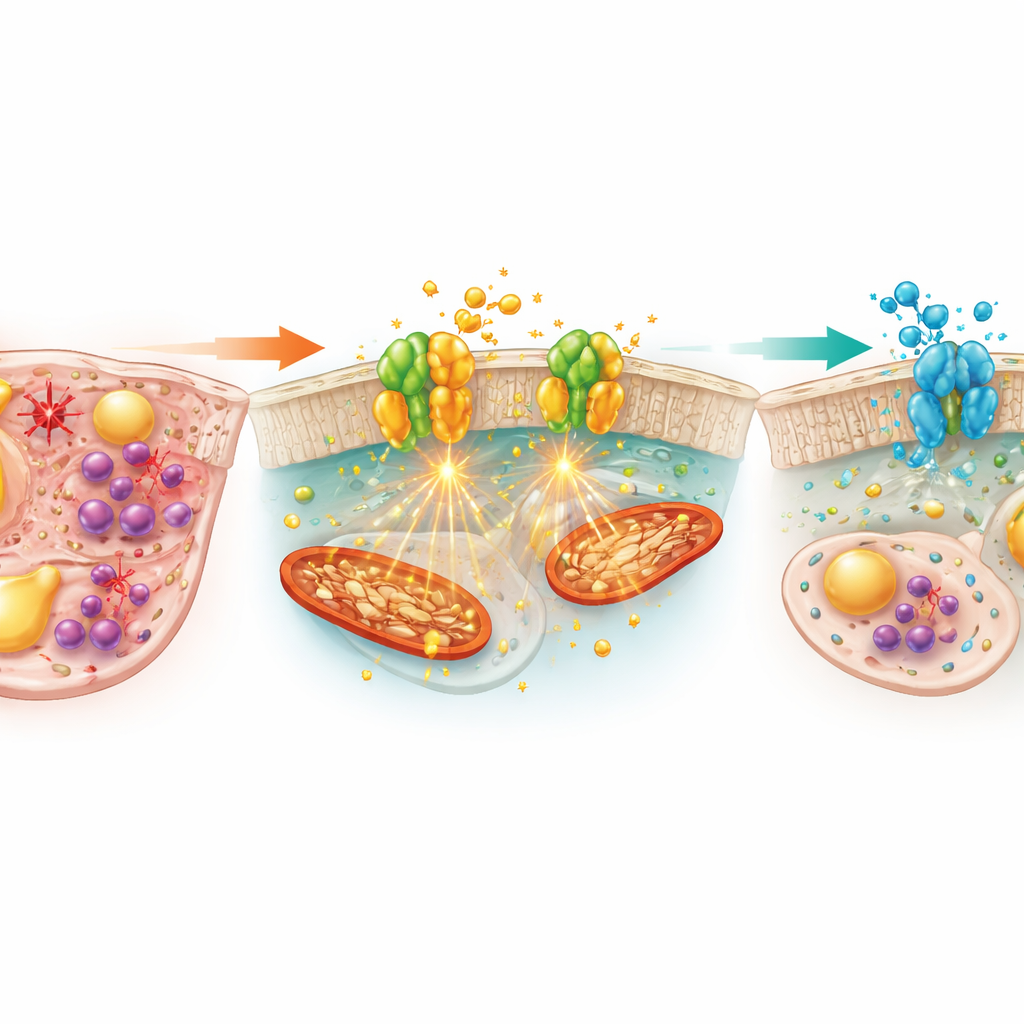
What this could mean for future treatments
Taken together, the findings suggest that NOX1 and NOX4 sit at a central crossroads of alcohol-induced liver damage, linking excess iron, fat accumulation, oxidative stress, and inflammation. By blocking this enzyme pair, Setanaxib was able to ease all four processes at once in ethanol-fed mice, offering broader protection than strategies that only replenish antioxidants or only target inflammation. While the work was done in animals and important questions remain about long-term safety, dosing, and effects in humans, the study provides a strong rationale for testing NOX1/4 inhibitors as multidimensional treatments for alcoholic liver disease, especially in patients showing signs of iron overload and oxidative stress.
Citation: Yu, L., He, T., Zhang, P. et al. NOX1/4 drives hepatic iron and lipid dysregulation, redox imbalance, and inflammation in ethanol-fed mice. Sci Rep 16, 12283 (2026). https://doi.org/10.1038/s41598-026-42716-4
Keywords: alcoholic liver disease, oxidative stress, iron overload, liver inflammation, Setanaxib