Clear Sky Science · en
A CAM bioimaging model reveals the connection between VEGFA vascular remodeling and enhanced sarcoma progression via tumor secretome
Why this research matters
Cancers called sarcomas arise in bones and soft tissues and often strike children and young adults. They are dangerous not only because they grow quickly, but because they spread through the body with the help of newly formed blood vessels. This study uses fertilized chicken eggs as a living testbed to watch, in real time, how sarcoma cells reshape nearby vessels and how blocking a single key signal can slow their growth and spread. The work points toward faster, cheaper ways to test drugs and refines our understanding of how tumors prepare new territory before they arrive.
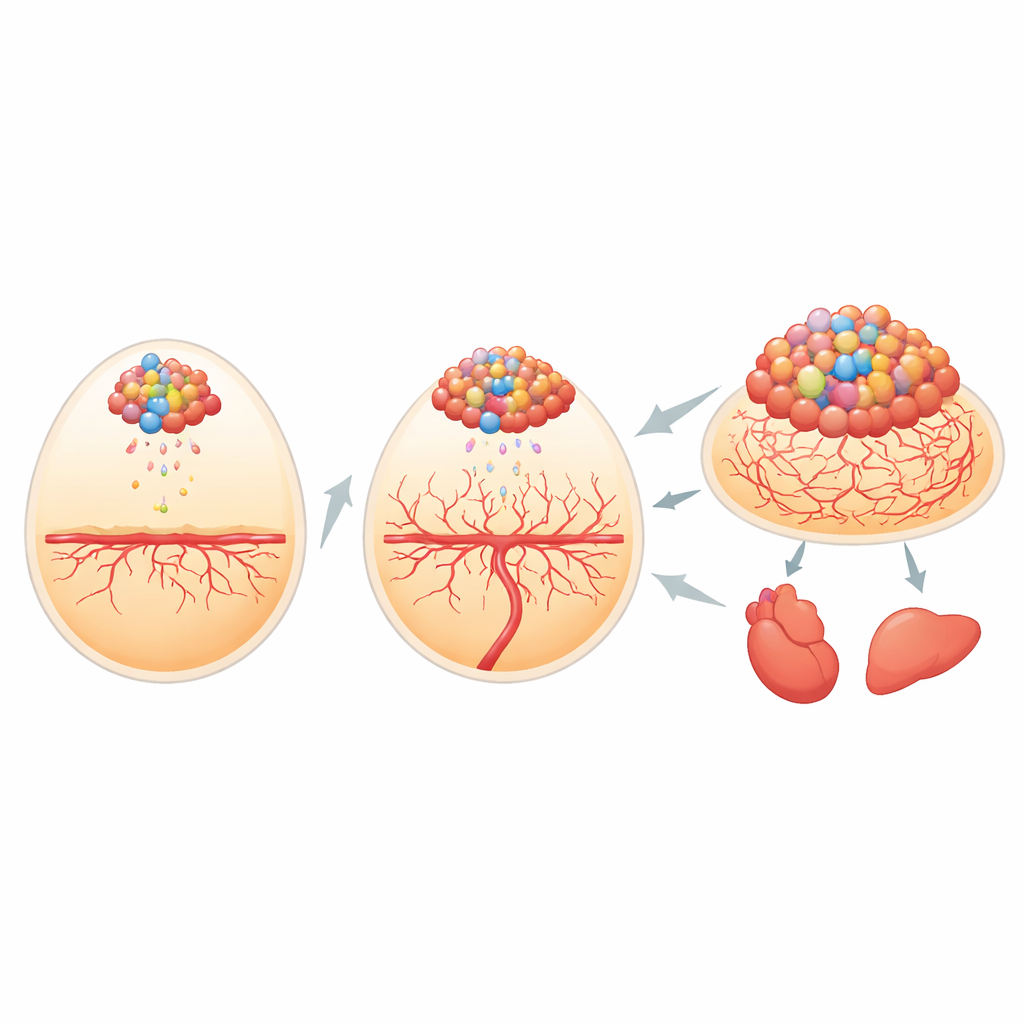
Building a window into tumor growth
The researchers first needed a way to track tiny clusters of cancer cells inside the developing chick. They engineered human bone cancer and mouse soft tissue cancer cells to carry two light-producing tags: one that glows under specific colored light and one that emits its own light when given a chemical fuel. When these cells were placed on the richly supplied membrane that surrounds the chick embryo, the team could measure the brightness of the signal and match it almost perfectly to the actual size of the tumors. The self-lit tag was especially powerful for spotting very small groups of cells that had broken away and lodged in organs such as the heart and liver, letting the scientists follow metastasis with high sensitivity.
How tumor “messages” prepare new ground
Before adding cancer cells, the team soaked the membrane with tumor cell–conditioned medium, essentially the broth in which sarcoma cells had been growing. This liquid contains the tumor secretome—hundreds of molecules shed by the cells. Membranes pretreated in this way formed tumors more often, and those tumors were more regular, larger, and packed with dividing cells than tumors on membranes that saw only plain medium. The secretome also drove cancer cells to leave the original site and appear in distant embryonic organs, mimicking how sarcomas spread in the body. These findings suggest that tumor-released factors can “precondition” nearby tissue, making it friendlier terrain for cancer to take root and travel.
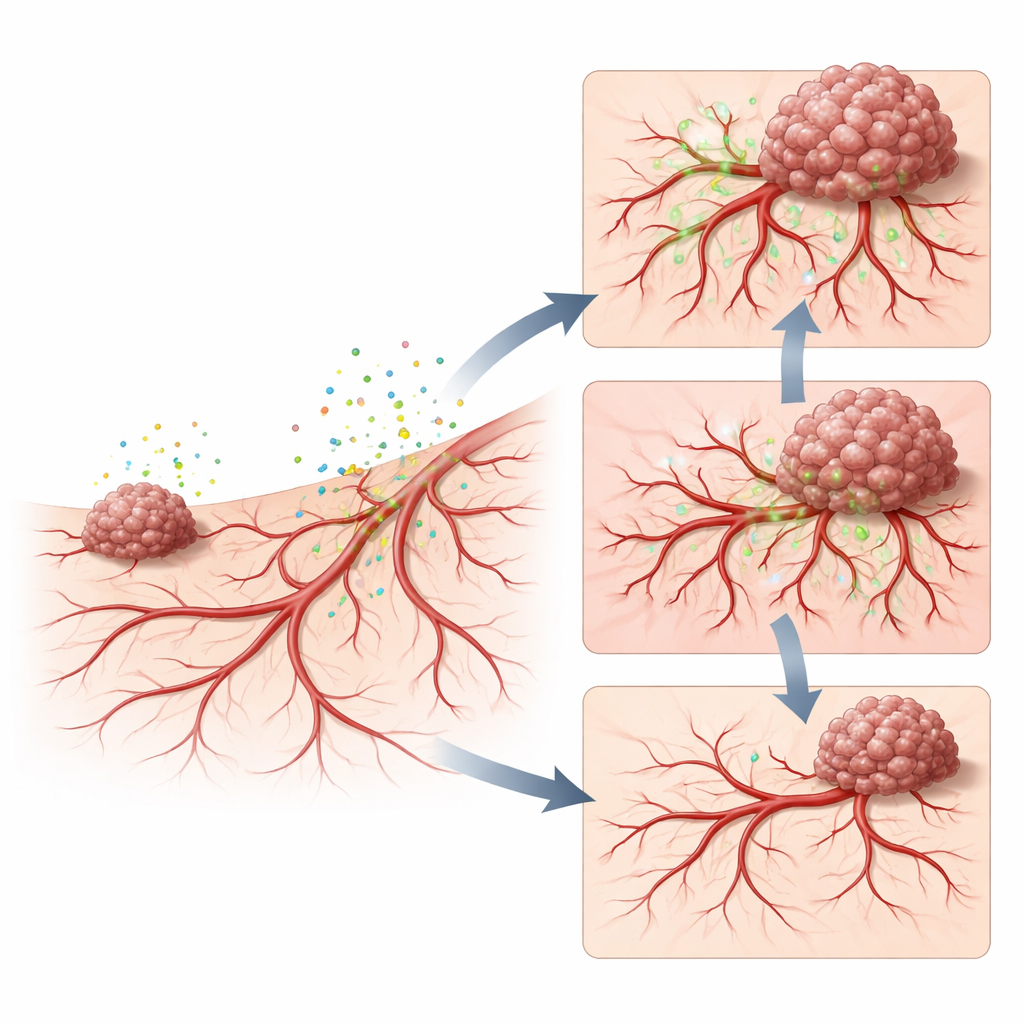
Rewiring blood vessels and making them leaky
To uncover what the secretome was doing to the host tissue, the scientists examined blood vessel growth and leakiness. Tumor-conditioned medium caused a surge of fine new vessels in the membrane and around early tumors, building a dense network that can feed rapidly growing cancer. When a fluorescent dye linked to a large sugar molecule was injected into the circulation, much more of it seeped out into tumor tissue after secretome treatment, showing that the new vessels were unusually leaky. This leaky, overgrown network mirrors what is seen in aggressive human cancers, where abnormal vessels not only supply nutrients but also give cancer cells easy exits into the bloodstream.
Pinpointing VEGFA as a key driver
The team then asked which secreted molecules were responsible. By profiling dozens of signaling proteins in the tumor broth, they found that several vessel-growing factors were elevated, with one called VEGFA standing out as both common to different sarcoma lines and especially abundant in the more metastatic cells. When they neutralized VEGFA in the secretome using a clinically used antibody drug, the membrane showed far fewer new vessels, less leakage, smaller tumors, and almost no detectable metastases. Briefly re-adding VEGFA partially restored abnormal vessels, leakage, and tumor spread. These experiments place VEGFA at the center of a chain that links tumor secretions, remodeling of the vascular niche, and enhanced sarcoma progression.
What this means for patients and future studies
This work shows that sarcoma cells do not just adapt to their environment; they actively reshape it ahead of time by releasing factors that call in new, fragile blood vessels. In the chick embryo system developed here, that remodeling can be watched and measured in days, using light-emitting tumor cells as a readout of growth and spread. By revealing VEGFA as a major switch that turns on this pro-tumor remodeling, the study supports efforts to target this signal and related pathways in sarcoma. At the same time, it offers researchers a practical platform to test combinations of drugs that calm the tumor secretome, stabilize vessels, and ultimately reduce the chance that these cancers will grow and metastasize.
Citation: Wang, Y., Xue, W., Sakr, N. et al. A CAM bioimaging model reveals the connection between VEGFA vascular remodeling and enhanced sarcoma progression via tumor secretome. Sci Rep 16, 12525 (2026). https://doi.org/10.1038/s41598-026-42154-2
Keywords: sarcoma, tumor microenvironment, angiogenesis, VEGFA, CAM model