Clear Sky Science · en
Effect of taxonomical distance and scriptaid on iSCNT embryo development in suidae
Bringing Back Vanishing Pigs
Many wild pig species are sliding toward extinction, yet collecting eggs from these rare animals is difficult and often impossible. One proposed workaround is a kind of biological "surrogacy": taking the DNA from a threatened animal and placing it into an egg from a related farm animal, in hopes of growing an embryo in the lab. This study asks how far we can stretch that approach within the pig family, and whether a small drug molecule can help overcome hidden compatibility problems inside these hybrid embryos.
Building Hybrid Embryos in the Lab
The researchers focused on members of the pig family, Suidae, which includes familiar domestic pigs as well as more elusive forest dwellers like the bearded pig and the babirusa. Using a technique called somatic cell nuclear transfer, they removed the genetic material from domestic pig eggs and replaced it with skin-cell nuclei from three sources: domestic pig (the baseline), bearded pig (a different species in the same genus), and babirusa (a more distant cousin from another subfamily). All embryos grew in the same domestic pig egg environment, allowing the team to isolate the effect of how closely related the donor and the egg species were. 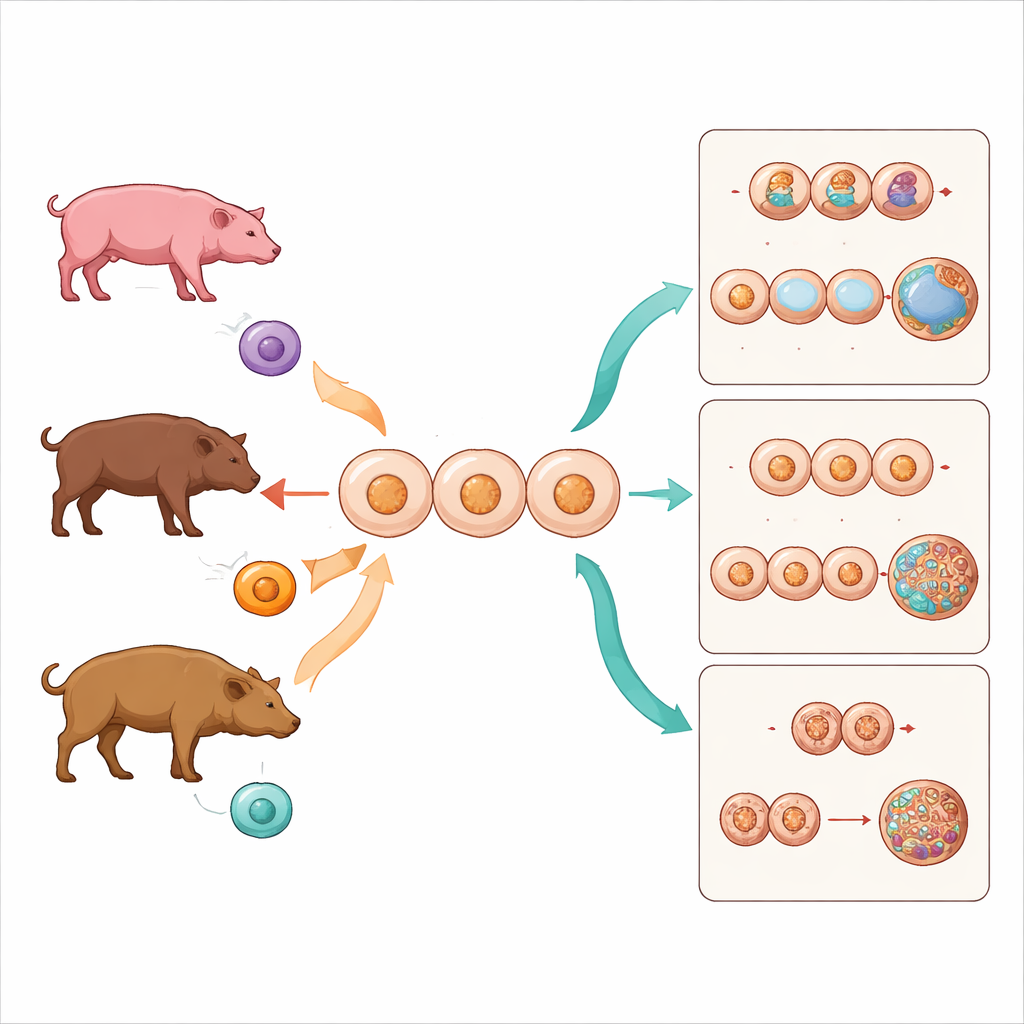
When Relatedness Shapes Chance of Life
The outcome was stark. Embryos made from domestic pigs cloned into domestic pig eggs had the best success, with more than a quarter reaching the blastocyst stage, an advanced point suitable for implantation. Bearded pig nuclei, which come from a different but closely related species, performed more poorly but still produced some blastocysts. In contrast, babirusa nuclei, drawn from a more distant branch of the pig family tree, never produced blastocysts at all, stalling earlier as small balls of cells. This steady decline in developmental success with increasing evolutionary distance highlights a practical barrier sometimes called the "taxonomic wall": beyond a certain gap between donor and egg, the embryo simply cannot continue.
A Chemical Push for Stalled Embryos
The team then tested whether Scriptaid, a drug that loosens how DNA is packaged, could improve these odds. By making the genetic material more accessible, Scriptaid can, in principle, help reset a donor nucleus to an early embryonic state. When bearded pig nuclei were treated with Scriptaid after transfer into domestic pig eggs, more embryos reached the blastocyst stage, and their early cell divisions occurred sooner, more closely matching those of domestic pig clones. The drug also briefly raised levels of a chemical mark on DNA-packaging proteins (H3K9 acetylation) during the first two cell stages, a sign that their genetic programs were more actively turned on. However, Scriptaid could not rescue the more distant babirusa embryos, which still failed to pass the morula stage.
Power Plants and Crossed Signals
To understand why relatedness mattered so much, the scientists examined which genes were turned on or off at several early stages in all embryo types. They found that many of the differences between successful and failing embryos involved the cell’s energy factories, the mitochondria. Mitochondria carry their own small set of genes but also depend heavily on genes in the cell nucleus. In the hybrid embryos, the nucleus and mitochondria came from different species, and their communication appeared to falter as the evolutionary distance widened. Many affected genes belonged to families that build a key part of the energy chain known as oxidative phosphorylation. In more distant pairings, these nuclear genes tended to be dialed down, while in closer pairings they could be dialed up. When Scriptaid was added, another set of mitochondrial genes—this time encoded inside the mitochondria themselves—responded strongly, hinting that the drug partly retuned this nuclear–mitochondrial partnership. 
What This Means for Saving Species
Taken together, the work shows that interspecies cloning within the pig family is not just a mechanical exercise in swapping DNA; it hinges on a delicate conversation between nuclear and mitochondrial genes that evolved together over millions of years. When donor and egg are closely related, this conversation can be strained but still function well enough to produce blastocysts, especially if helped along by a chromatin-modifying drug like Scriptaid. When the species are more distant, the mismatch in their cellular "power grid" becomes too great, and development stalls despite chemical assistance. For conservation efforts, this suggests that interspecies cloning is most promising between very closely related species and that future strategies will need to directly tackle the energy-production mismatch, not only reprogram the nucleus.
Citation: Liu, HJ., Wong, K.W., Ma, X. et al. Effect of taxonomical distance and scriptaid on iSCNT embryo development in suidae. Sci Rep 16, 11288 (2026). https://doi.org/10.1038/s41598-026-41963-9
Keywords: interspecies cloning, endangered pigs, mitochondria, embryo development, epigenetic drugs