Clear Sky Science · en
Scalable flow synthesis of ultrasmall inorganic nanoparticles for biomedical applications via a confined impinging jet mixer
Why Tiny Particles Could Make a Big Difference
Hospitals increasingly rely on microscopic particles to see disease earlier, deliver drugs more precisely, and fight stubborn infections. But making these particles in large, consistent batches has been surprisingly difficult, often requiring high heat, harsh chemicals, and complex equipment. This study introduces a simple, water-based production method that can churn out uniform, ultrasmall particles suitable for medical use, potentially smoothing the path from lab discovery to real-world treatments and imaging tools. 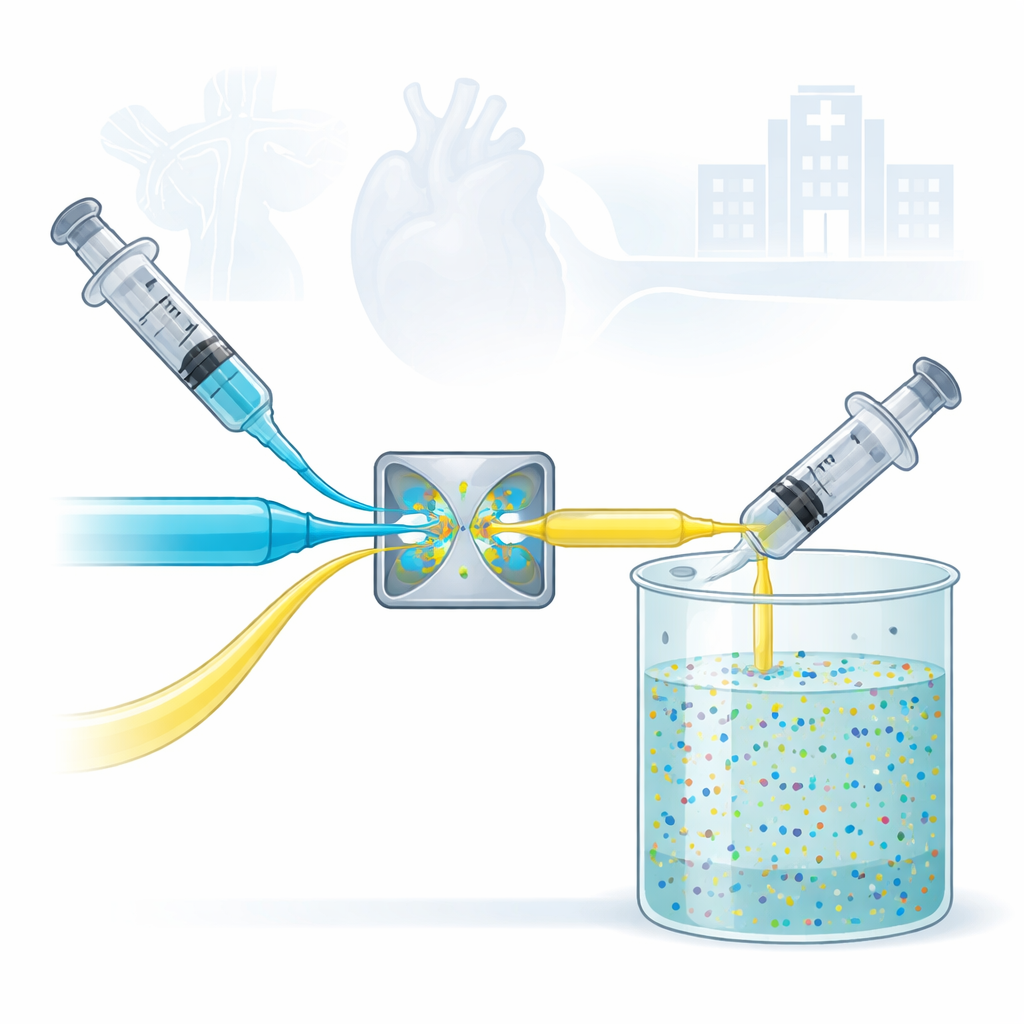
A New Way to Mix at the Smallest Scales
The heart of the work is a device called a confined impinging jet mixer, or CIJM. It looks unassuming: two liquid streams are pushed toward each other and collide head-on inside a small chamber. That violent but precisely controlled collision instantly mixes the ingredients and causes tiny inorganic particles to form in a process known as flash precipitation. Unlike many traditional methods, the entire reaction happens in water at room temperature, without toxic organic solvents, special gas atmospheres, or long heating steps. This makes the approach both safer and easier to scale for industrial production.
Building Four Types of Helpful Nanoparticles
Using this single mixer design, the team produced four different kinds of nanoparticles, each with its own medical promise. Silver sulfide and silver telluride particles can act as bright contrast agents for X-ray–based scans such as mammography and computed tomography. Cerium oxide particles behave like tiny antioxidants, mopping up damaging reactive oxygen molecules that contribute to inflammation and tissue injury. Iron oxide particles are magnetic and catalytic, making them useful both as imaging agents and as helpers in breaking down harmful bacterial biofilms. All four types ended up extremely small—typically between one and five billionths of a meter across—and were coated with biocompatible molecules to keep them stable in the body.
Dialing in Size and Shape Like a Control Knob
For many medical uses, the exact size of a particle matters. Smaller particles can slip through the kidneys and be cleared from the body, reducing long-term buildup in organs, while size also influences how strongly they show up in scans or catalyze reactions. The researchers systematically adjusted how fast the fluids flowed, how concentrated the ingredients were, and how they were mixed or diluted after formation. For silver sulfide particles, simply shifting the ratio of the incoming ingredient streams changed the particle diameter from about two to just over five nanometers without affecting quality. Silver telluride particles responded instead to the strength of a chemical that donates electrons and to how quickly the product was diluted after mixing. Iron oxide particles showed only modest changes in core size, but their overall “cloud” size in water—a key feature for how they travel in the body—could be tuned over a wide range. Cerium oxide particles were less adjustable in size, but could still be produced reliably at room temperature. 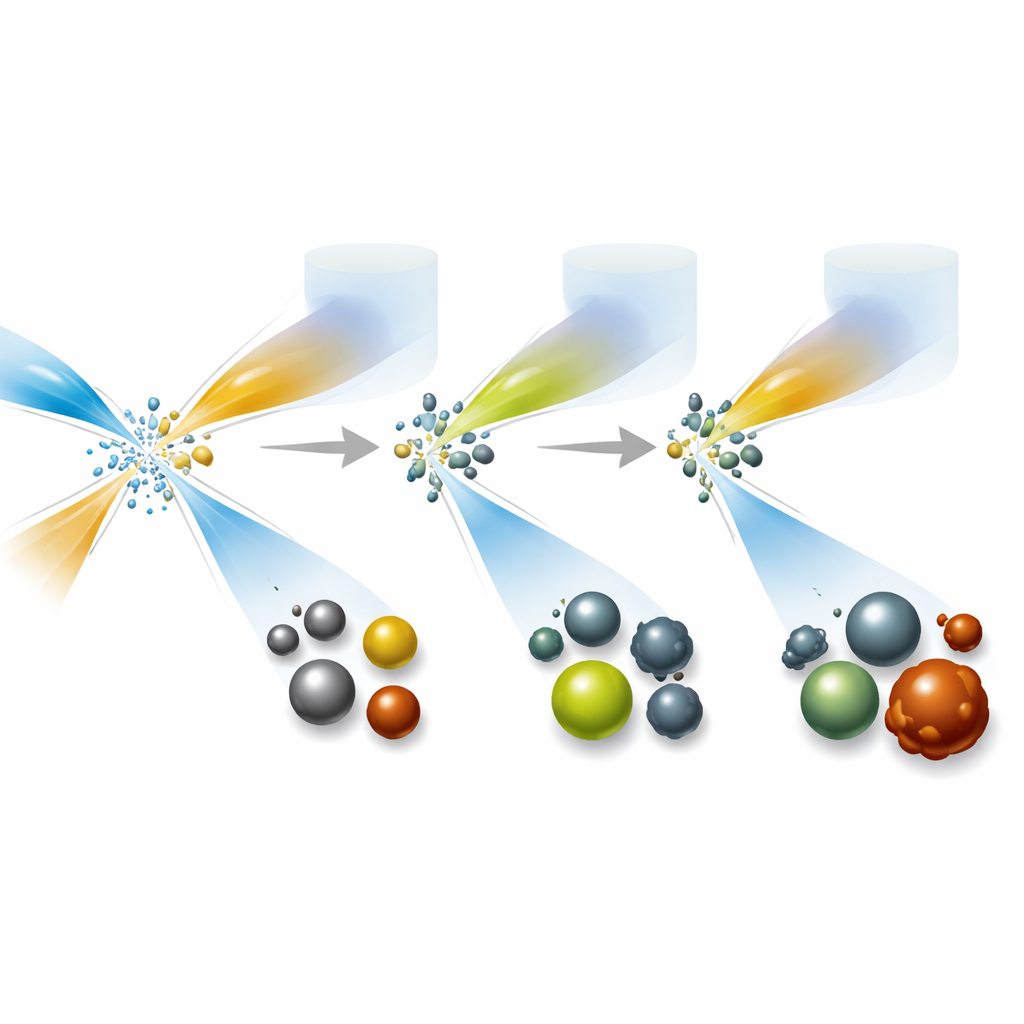
Proving They Work in Real-World Tests
Making particles is one thing; showing that they actually perform in useful ways is another. In imaging phantoms that mimic human tissue, both silver-based particles produced stronger X-ray contrast than a widely used iodine dye, meaning they could help radiologists see subtle features more clearly at the same metal dose. The cerium oxide particles protected cultured human cells from otherwise damaging bursts of hydrogen peroxide, acting like miniature antioxidant enzymes. The iron oxide particles worked together with hydrogen peroxide to rapidly kill mouth bacteria living in tough protective films, suggesting a way to enhance treatments for tooth decay and other oral infections. Across these tests, the new particles matched or exceeded the performance of versions made with more complicated methods.
From Bench-Top to Bucket Without Losing Quality
A frequent stumbling block in nanomedicine is that processes that work in tiny vials break down when scaled to factory-sized batches. The team showed that their mixer can overcome this. By increasing the flow rate and starting solution volumes, they produced about a liter of silver sulfide nanoparticle suspension in just fifteen minutes—a roughly hundredfold scale-up. Measurements of size, structure, and optical properties revealed that the large-batch particles were nearly indistinguishable from those made at small scale. Because the CIJM is commercially available, relatively inexpensive, and resistant to clogging, it could be adopted without custom engineering.
What This Could Mean for Future Medicine
In plain terms, the study demonstrates a practical “assembly line” for making very small, medically useful particles in water, at room temperature, and in large quantities. The confined impinging jet mixer can be tuned to produce several kinds of inorganic nanoparticles that keep their intended properties—whether that is bright X-ray contrast, antioxidant protection, or bacteria-killing power—while remaining small enough for safe clearance from the body. This kind of reliable, scalable production technology is a key missing link between promising nanomedicine discoveries and the routine tools used in clinics, and it may help speed the arrival of more precise imaging agents and targeted therapies.
Citation: Kian, A.C., Gupta, M., Hong, H. et al. Scalable flow synthesis of ultrasmall inorganic nanoparticles for biomedical applications via a confined impinging jet mixer. Sci Rep 16, 11135 (2026). https://doi.org/10.1038/s41598-026-41509-z
Keywords: nanoparticles, biomedical imaging, microfluidics, drug delivery, antimicrobial therapy