Clear Sky Science · en
Data-driven explainable chronic kidney disease detection using RF based data imputation and meta-ensemble learning
Why this matters for everyday health
Chronic kidney disease often creeps in silently, damaging the body long before symptoms appear. For many people, especially in low-resource settings, simple blood and urine tests may be available, but doctors do not always have tools that make the most of that information. This study shows how a carefully designed artificial intelligence (AI) system can turn routine lab data into an early warning signal for kidney trouble, while still letting clinicians understand why the computer is flagging a patient.
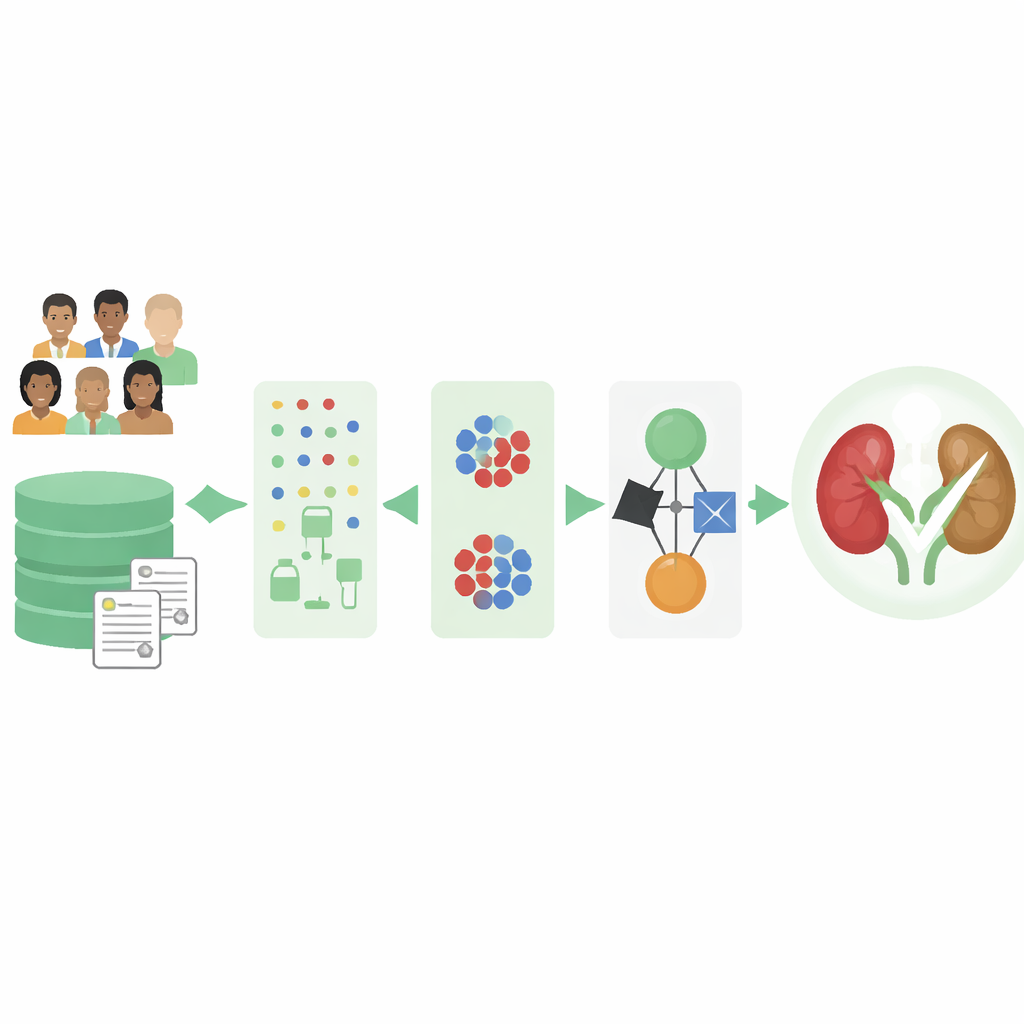
Turning messy clinic records into usable clues
Real-world medical records are rarely complete. Lab results may be missing, and some types of patients may be recorded far more often than others. The authors worked with a well-known public dataset of 400 people, each described by 25 basic measurements such as age, blood pressure, blood counts, and kidney-related chemicals. Many entries had gaps, and there were more people without kidney disease than with it, which can bias computer models. To fix this, the team first built a smart cleaning step that learns patterns from the existing data to fill in missing values rather than simply discarding incomplete records or using crude averages.
Balancing the scales between sick and healthy
Because the dataset contained more non-kidney-disease cases, a model trained naively might learn to play it safe by mostly predicting “healthy” and still achieve a deceptively high score. To counter this imbalance, the researchers used a method that creates realistic synthetic examples of the underrepresented group. In essence, it studies patients who do have chronic kidney disease and generates new, slightly varied cases that resemble them. After this step, the computer sees a more balanced picture of sick and healthy individuals, which helps it pay attention to those early, easy-to-miss warning signs.
Many simple minds working together
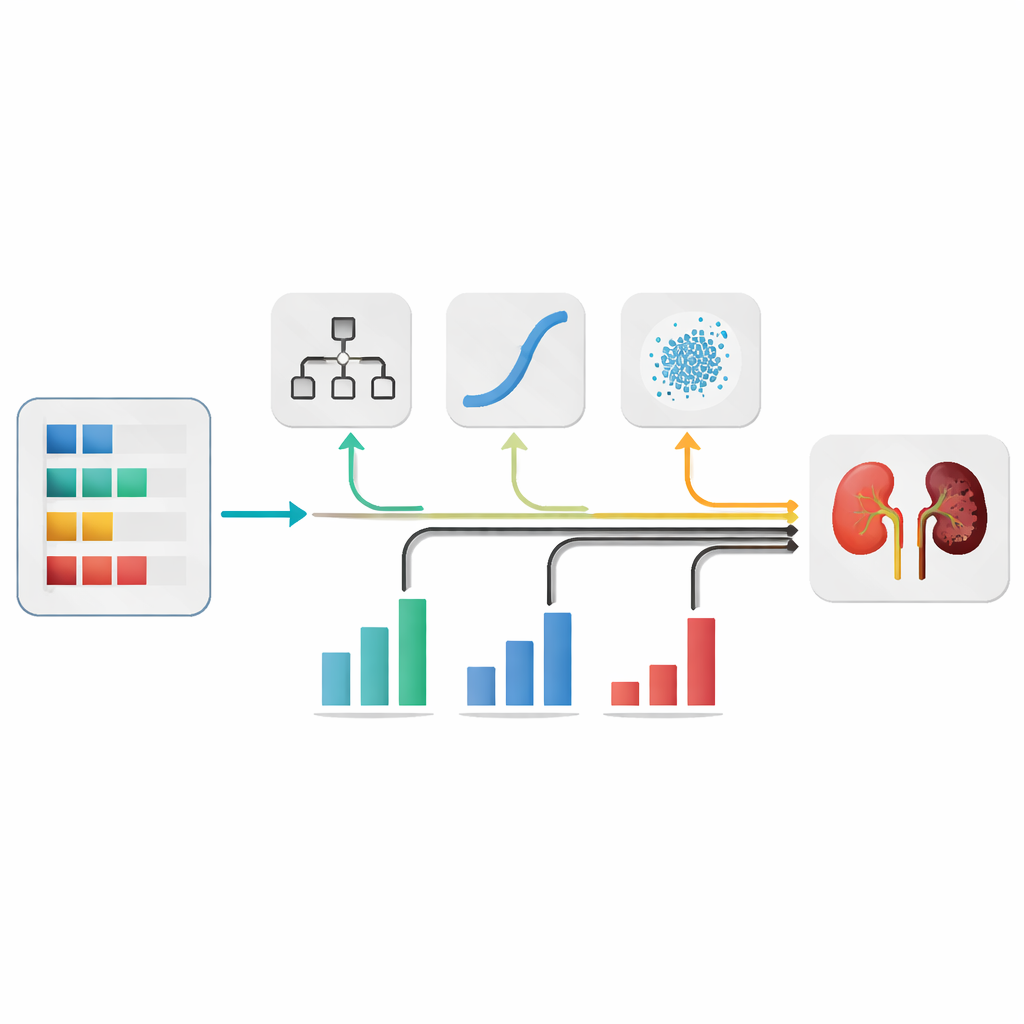
Instead of betting on a single type of algorithm, the authors assembled several familiar machine-learning models that each look at the same patient data in different ways. They evaluated five contenders and chose three that performed best: a decision tree, a logistic model, and a simple probabilistic classifier. These models were then combined into an “ensemble,” where each gives its own opinion about whether a patient likely has kidney disease. The final decision is a weighted blend of their outputs, similar to consulting multiple doctors whose opinions are not counted equally but according to how reliable they are.
Letting a digital wolf pack pick the best mix
Choosing how much to trust each model in the ensemble is crucial. Rather than guessing, the authors used an optimization technique inspired by the hunting behavior of grey wolves. This algorithm explores many combinations of weights and gradually moves toward the mix that yields the highest accuracy on held-out data. With this tuned combination, the system correctly classified nearly 99 out of 100 cases in cross-validation and maintained a very low rate of missed kidney disease patients, an especially important goal in screening.
Opening the black box for clinicians
A major concern with AI in medicine is that its decisions can seem opaque. To address this, the researchers applied explainability tools that show which lab features push a prediction toward or away from kidney disease for each patient. They found that measures like albumin in urine, red blood cell counts, blood pressure, diabetes status, and kidney-related blood markers strongly influenced the model’s judgments. These patterns align with medical knowledge, suggesting the system is learning clinically sensible rules rather than obscure statistical quirks.
What this could mean for patients
In plain terms, this work demonstrates that a carefully prepared and explained AI assistant can turn routine lab data into a highly reliable early detector of chronic kidney disease. By cleaning missing information, correcting imbalances in the data, blending several simple models, and then shining a light on how decisions are made, the framework achieves high accuracy without becoming a mysterious black box. While it still needs to be tested on larger and more diverse patient groups before use at the bedside, it points toward a future in which inexpensive tests, combined with transparent AI, help doctors catch kidney trouble earlier and tailor care more confidently.
Citation: Gupta, R., Gambhir, S., Krejcar, O. et al. Data-driven explainable chronic kidney disease detection using RF based data imputation and meta-ensemble learning. Sci Rep 16, 12679 (2026). https://doi.org/10.1038/s41598-026-41425-2
Keywords: chronic kidney disease, medical AI, ensemble learning, health data preprocessing, explainable AI