Clear Sky Science · en
Multifunctional hierarchical Ni2CoS4 structures for combating drug-resistant infections and colorectal cancer via piezocatalytic ROS generation
Why this new approach to tough infections and cancer matters
Antibiotic‑resistant infections and colorectal cancer are two of the biggest threats to modern health, and both are becoming harder to treat with existing drugs. This study explores a very different kind of therapy: tiny nickel–cobalt sulfide particles that spring into action when jolted by gentle ultrasound. Instead of acting like a classic drug, these particles turn mechanical energy into bursts of reactive molecules that can tear apart bacteria, biofilms, and cancer cells—while largely sparing healthy cells at practical doses.
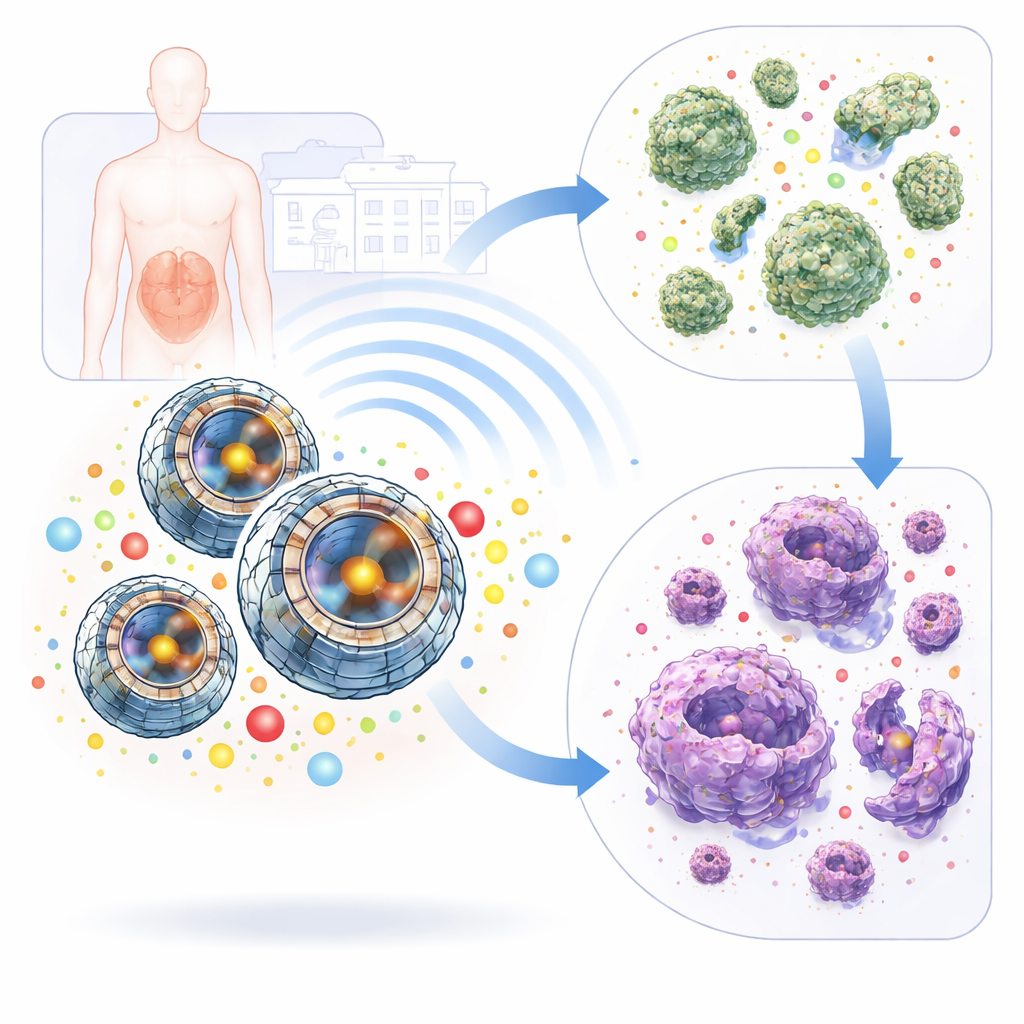
Small engineered spheres with a layered design
The researchers created a family of nickel–cobalt sulfide (Ni₂CoS₄) particles using a controlled heating process in liquid, then systematically varied time, temperature, and ingredient levels to tune their structure. Microscopy revealed that the best formulation, dubbed N3, forms nearly uniform hollow spheres made from thin, interlocking nanosheets, like a loosely woven ball. X‑ray and Raman measurements showed that N3 is also highly crystalline and chemically well‑ordered, and gas adsorption tests confirmed it has a relatively large internal surface area. Together, these traits mean that N3 can efficiently move electrical charges inside itself and expose many active sites to its surroundings—both crucial for its unusual mode of action.
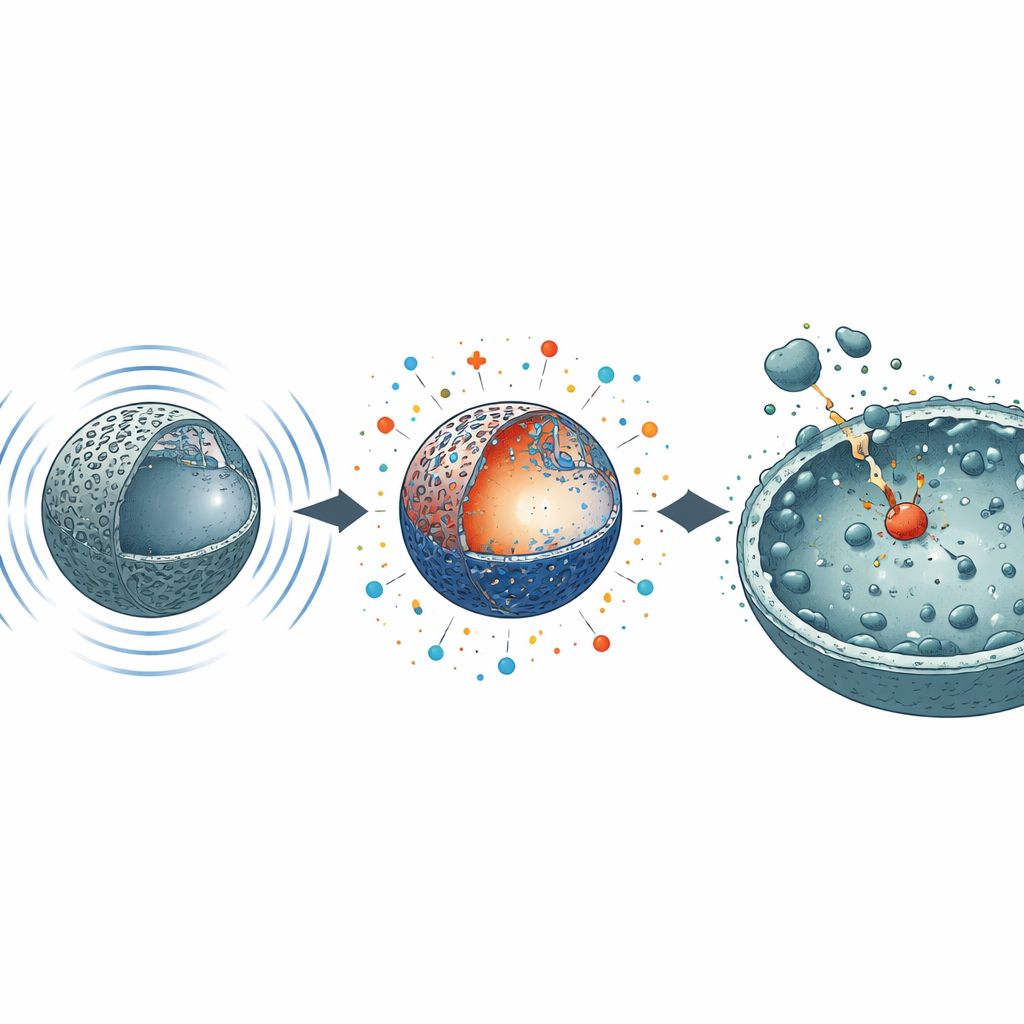
Turning sound into chemical attack
Unlike therapies that rely on light or added chemicals, this system is driven by mechanical energy. When the N3 spheres are exposed to ultrasound in liquid, the pressure waves squeeze and stretch their crystal lattice. This distortion separates positive and negative charges inside the material, which then react with dissolved oxygen to form several types of highly reactive oxygen species (ROS). In dye‑degradation tests used as a stand‑in for biological targets, sonicated N3 removed nearly 89% of a stubborn blue dye in just four minutes at 200 watts of ultrasound—far better than non‑sonicated material. Measurements with molecular probes showed that N3 boosts two long‑lived ROS, singlet oxygen and superoxide, more than thirteenfold compared with its inactive form, while also modestly increasing hydroxyl radicals. Once activated, N3 continues to show strong catalytic activity for days, indicating a “memory” effect where its reactive state persists long after the ultrasound is switched off.
Breaking down drug‑resistant bacteria and their biofilms
To test real‑world impact, the team challenged the particles with dangerous clinical strains of multidrug‑resistant Staphylococcus aureus and extensively drug‑resistant Pseudomonas aeruginosa. Among all formulations, sonicated N3 was consistently the most powerful, stopping bacterial growth at very low concentrations—down to 5–10 micrograms per milliliter—and fully clearing cultures within 24–48 hours. Disc tests showed that N3 produced larger kill zones than a commonly used antibiotic, especially against resistant strains. Microscopy images of treated bacteria revealed shredded cell walls, collapsed shapes, and leaking interiors, while a fluorescent dye that senses membrane damage showed sharp jumps in signal. N3 also attacked the slimy protective communities called biofilms: at tens of micrograms per milliliter it prevented new biofilms from forming and removed more than 99% of mature ones, outperforming non‑activated particles across the board.
Targeting colorectal cancer while sparing normal cells
Because the same ROS chemistry can injure tumor cells, the researchers next exposed human colorectal cancer (HCT‑116) cells to sonicated N3. After 24 hours, half the cancer cells were disabled at about 100 micrograms per milliliter, and most were dead at 200 micrograms per milliliter, with complete loss of viability at the highest tested dose. Follow‑up over four days showed that a single exposure kept killing cells, driving survival down to roughly 13% by 96 hours without any further ultrasound. Flow‑cytometry measurements confirmed widespread loss of membrane integrity at these doses. Normal human skin fibroblasts, by contrast, tolerated the same treatment much better: at the cancer‑cell half‑inhibiting dose, nearly 90% of fibroblasts remained viable, indicating a useful though not absolute safety window.
What this could mean for future treatments
Taken together, the results suggest that carefully engineered Ni₂CoS₄ spheres like N3 can act as miniature, rechargeable reactors inside the body: a brief ultrasound pulse switches them into a long‑lived state that continually generates ROS, which in turn perforate bacterial membranes, destabilize biofilms, and push cancer cells into lethal oxidative stress. Because this attack relies on broad physical and chemical damage rather than a single biochemical target, it may be much harder for microbes to evolve resistance. The work is still at a laboratory stage, and questions about long‑term safety, delivery, and behavior in living organisms remain. But it points toward a new class of “mechanically activated” therapies that could one day complement or rescue failing antibiotics and bolster cancer treatment.
Citation: Qurbani, K., Amiri, O. & Hamzah, H. Multifunctional hierarchical Ni2CoS4 structures for combating drug-resistant infections and colorectal cancer via piezocatalytic ROS generation. Sci Rep 16, 10294 (2026). https://doi.org/10.1038/s41598-026-41092-3
Keywords: piezocatalysis, antimicrobial resistance, biofilm disruption, nanoparticle therapy, colorectal cancer