Clear Sky Science · en
Efficacy and safety signals from early-phase studies of KRAS inhibition in pancreatic cancer
Why this matters for patients and families
Pancreatic cancer is one of the deadliest cancers, with most patients living only months after diagnosis once the disease has spread. For decades, doctors have had to rely almost entirely on harsh chemotherapy that offers limited benefit. This article looks at a new kind of medicine that directly targets a key faulty protein, called KRAS, which drives most pancreatic cancers. By pulling together results from early clinical trials, the researchers ask a simple but urgent question: do these new drugs work well enough, and are they safe enough, to change the outlook for people facing this disease?
A tough cancer and a long-standing roadblock
Pancreatic ductal adenocarcinoma, the most common type of pancreatic cancer, is aggressive and often diagnosed late, when surgery is no longer an option. Standard drug treatments combine multiple chemotherapies. While these can slightly extend survival, they frequently cause strong side effects that force dose cuts or early stoppage. Almost 90 percent of these tumors carry mutations in KRAS, a molecular on–off switch that controls growth signals in cells. For years, KRAS was labeled “undruggable” because its surface offered few places where a drug could latch on. As a result, attempts to block KRAS indirectly, by hitting other parts of its signaling network, largely failed to improve outcomes.
A new class of drugs takes aim at KRAS
This picture changed with the invention of small molecules that can bind directly to mutated KRAS and interfere with its activity. KRAS normally toggles between an inactive and an active form; cancer-causing mutations freeze it in the “on” position, constantly telling cells to grow. Different experimental drugs now being tested can lock KRAS in its off state, attack it while it is on, or form more complex three-part structures that disable its function. Because these drugs recognize different shapes of the same protein, they may have different strengths, weaknesses, and resistance patterns. The authors argue that their effects cannot be assumed to be identical and need to be judged drug by drug. 
What the early trials are showing
To understand what these new medicines are achieving so far, the researchers performed a systematic review and meta-analysis of seven early-phase studies that included 463 patients with advanced or metastatic pancreatic cancer. All patients had already received at least one prior line of treatment, making their disease particularly hard to control. Only 293 patients could be evaluated for tumor shrinkage because many lacked complete scans or dropped out for various reasons. Pooling the available data, about 29 percent of evaluable patients saw their tumors shrink by a meaningful amount. The results were strikingly consistent between studies, suggesting that future trials using similar drugs in similar patients are likely to see comparable response rates. For pancreatic cancer—where meaningful shrinkage has historically been rare—this level of activity is considered encouraging, though still far from a cure.
Side effects and study caveats
Effectiveness is only part of the story; safety matters just as much. Across the trials, stomach and gut problems were among the most frequent side effects. Around four in ten patients experienced diarrhea or nausea. When the authors focused only on patients with pancreatic cancer in studies that reported disease-specific safety data, the rates were similar or slightly higher. The exact pattern of side effects varied between drugs, and some broader studies that mixed different tumor types did not break out pancreatic cancer separately, making it harder to spot clear trends. The authors also found that the overall risk of bias in the included studies was moderate to high. These were small, early-phase, single-arm trials without comparison groups, which makes it impossible to say for sure how much better patients did than they would have with standard care alone. 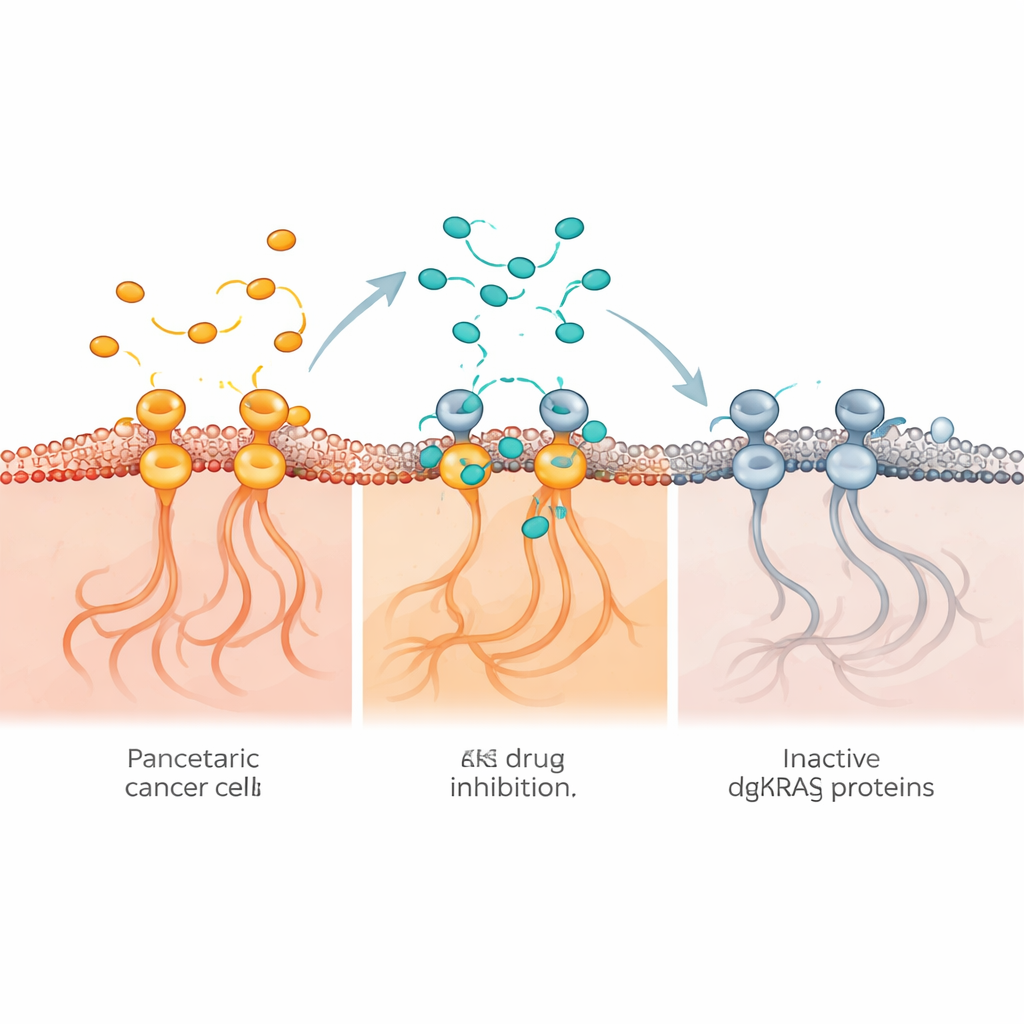
Where the field goes from here
Despite these cautions, the combined evidence supports a key conclusion: it is now genuinely possible to drug KRAS in pancreatic cancer, and doing so can produce real, if modest, tumor shrinkage in heavily pretreated patients. The authors see this as a landmark shift after decades of frustration. At the same time, responses are not yet durable for most patients, and side effects remain an issue. The paper calls for larger, more rigorous, and more uniform trials that match specific KRAS mutations to specific drugs, combine these agents wisely with immunotherapies and other targeted treatments, and embed modern biomarkers such as blood-based tumor DNA tracking. For patients and families, the message is cautious but hopeful: KRAS inhibitors are opening an important new door in pancreatic cancer care, but much work remains to turn early signals into long-lasting benefit.
Citation: Tiede, K.O.M., Teixeira, M.F., Moura, M. et al. Efficacy and safety signals from early-phase studies of KRAS inhibition in pancreatic cancer. Sci Rep 16, 13189 (2026). https://doi.org/10.1038/s41598-026-40757-3
Keywords: pancreatic cancer, KRAS inhibitors, targeted therapy, clinical trials, drug safety