Clear Sky Science · en
Deep learning for vessel segmentation and flow analysis to identify clusters associated with adverse outcomes in a fontan patient registry
Why studying heart flow in children matters
Children born with only one working heart pump often undergo a complex surgery called the Fontan procedure so they can survive into adulthood. These patients can look stable for years, yet some later develop serious problems affecting the heart and liver. Doctors already collect detailed MRI scans that capture how blood actually moves through their hearts and vessels over time, but most of this rich motion data is never used. This study shows how modern artificial intelligence can unlock that hidden information at scale, revealing patterns of blood flow that are linked to better or worse long‑term health.
Turning thousands of scans into usable data
The researchers worked with the FORCE registry, a large international collection of cardiac MRI exams from more than 3,000 people with Fontan circulation. Each exam includes specialized movies that track how fast blood flows through key blood vessels with every heartbeat. To learn from these scans, the team first needed to outline the vessels on each image frame and measure flow over time—a tedious manual task that would have taken an estimated year of nonstop expert work. Instead, they built a deep learning model that can both recognize which vessel is being imaged and automatically draw the vessel boundary for five major pathways: the aorta and four big veins and arteries leading to and from the lungs.
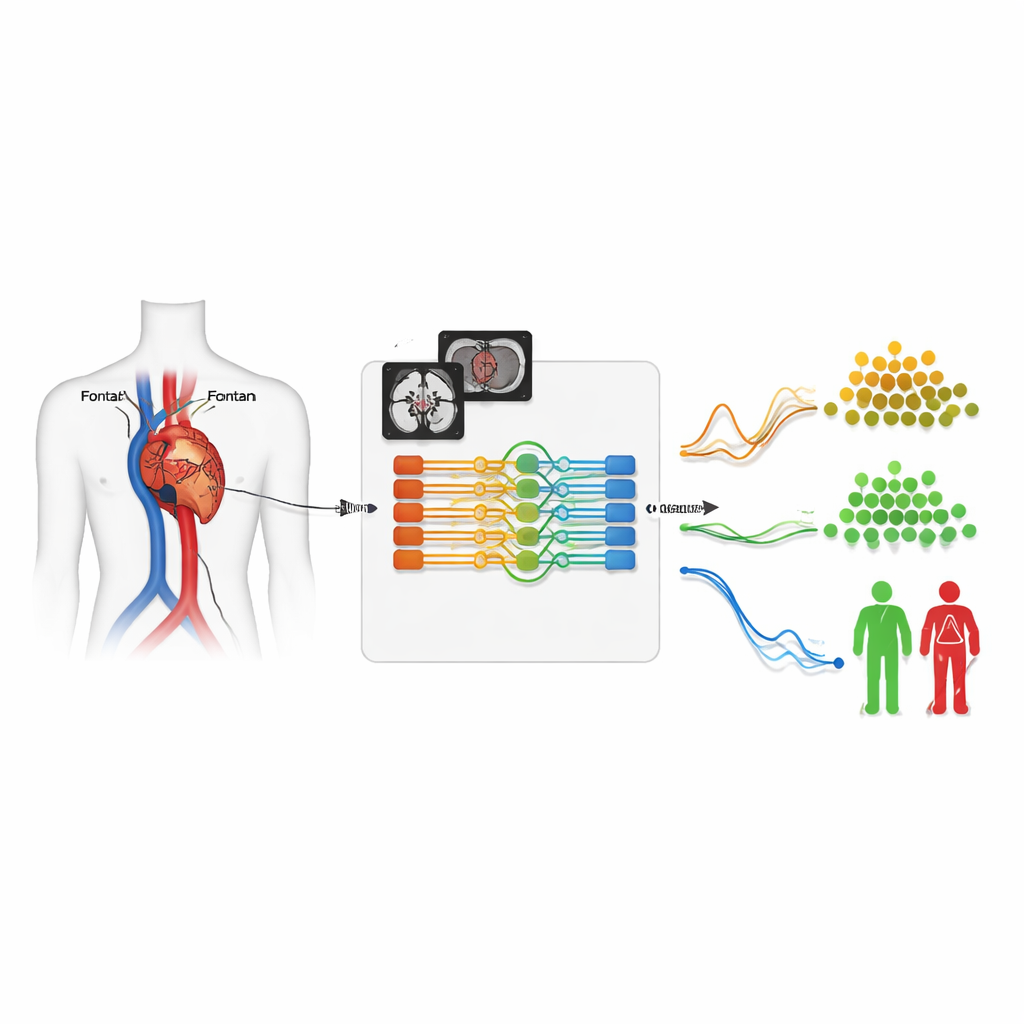
How the smart segmentation system works
The core of the system is a neural network that analyzes each movie as a short 3D block: two spatial dimensions plus time. It takes in two versions of the MRI signal—one showing anatomy, the other highlighting moving blood—and has been trained on 260 exams where experts had already traced the vessels by hand. The model doesn’t just outline structures; it also learns to identify which vessel is which, aided by information the scanner operator typed in at the time of imaging. With this combined classification-and-segmentation design, the system achieved very high overlap with human tracings and correctly identified the vessel type in nearly all test cases, despite the unusually twisted and variable anatomy common in Fontan patients.
Scaling up to an entire international registry
Once validated, the model was placed into an automated cloud pipeline and unleashed on more than 4,500 registry exams. For each scan, it sifted out the flow movies, segmented the five target vessels, and generated a flow‑versus‑time curve for every heartbeat, all without human intervention. Experts later checked these results and found that around 90% of the segmentations were good enough for clinical use when all five vessels were present. The system performed slightly less well in patients with especially unusual anatomies, such as duplicated veins, but still delivered a massive, previously unattainable dataset of clean, time‑varying blood‑flow measurements.

Finding hidden flow types linked to outcomes
With thousands of flow curves in hand, the team’s second deep learning model searched for natural groupings of patients based solely on how blood moved through their vessels over time. This model compressed each pair of curves—either from the lung arteries or the large veins—into a compact internal representation and then clustered patients into distinct flow “phenotypes.” Some groups had balanced, normal‑sized flows; others showed reduced overall flow, dominant flow to one lung, or flow that was shifted toward the relaxation phase of the heartbeat. The researchers then followed patients over time and discovered that certain flow patterns were associated with higher risks of death, heart transplant, or liver disease, even after accounting for age, heart pumping strength, and other standard measures.
What this could mean for future care
In simple terms, the study demonstrates that how blood moves through the Fontan circulation—which vessels carry more or less flow, and at what point in the heartbeat—contains vital clues about which patients are likely to face serious complications. The authors show that artificial intelligence can turn large archives of raw MRI movies into meaningful flow profiles and risk groups, without manual tracing or hand‑crafted rules. In the future, a system like this could run automatically right after a child’s MRI, flagging those whose flow patterns resemble higher‑risk groups and helping doctors tailor follow‑up, testing, or even surgical planning. Beyond Fontan patients, the same approach could be adapted to other heart conditions, motion‑tracking scans, or even everyday signals like ECGs, making fuller use of information that hospitals already collect but rarely mine in depth.
Citation: Yao, T., Clair, N.S., Gong, M. et al. Deep learning for vessel segmentation and flow analysis to identify clusters associated with adverse outcomes in a fontan patient registry. Sci Rep 16, 11956 (2026). https://doi.org/10.1038/s41598-026-40738-6
Keywords: Fontan circulation, cardiac MRI, deep learning, blood flow patterns, patient risk stratification