Clear Sky Science · en
Single-cell analysis reveals that the hormone T3 affects colon epithelial differentiation and induces a mixed progenitor-like cell population
Why a gut hormone story matters
Every day, the lining of your intestine quietly renews itself, replacing millions of cells to digest food, absorb nutrients, and protect against germs. This constant turnover depends on stem cells buried deep in tiny pockets called crypts. The hormone T3, made by the thyroid, is known to influence growth and metabolism throughout the body, yet its fine‑scale impact on the colon’s renewing cells has been unclear. This study uses single‑cell genetic profiling to zoom in on individual cells in the mouse colon and reveals how T3 can nudge them into an unusual, mixed state that may matter for gut health and possibly cancer risk.
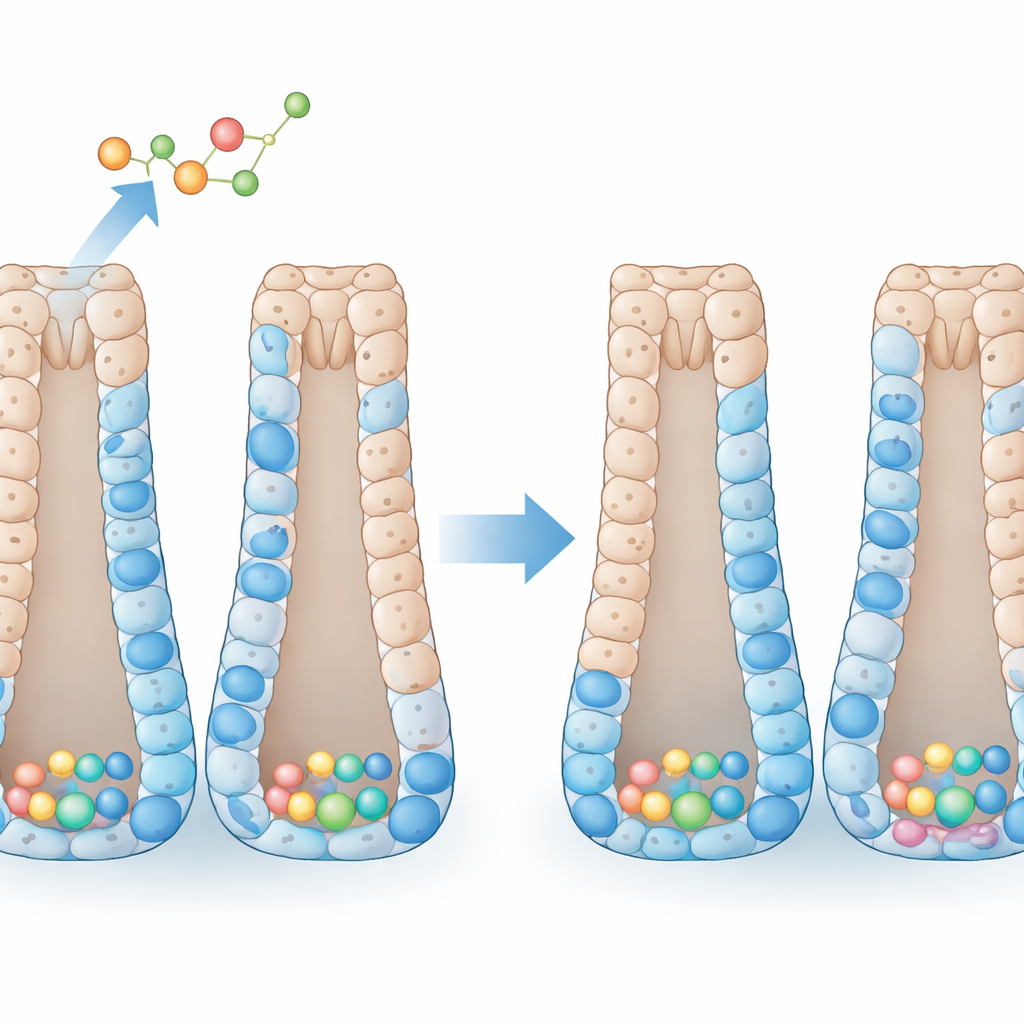
How the colon keeps itself in shape
The colon’s inner lining is organized like a series of test tubes turned upside down. At the bottom of each tube sit stem cells, which divide to make progenitor cells. As these progenitors climb upward, they specialize into several mature types: absorptive cells that take up water and nutrients, goblet cells that secrete protective mucus, hormone‑producing enteroendocrine cells, and tuft cells that help sense the environment. Under normal conditions, a tight balance between self‑renewal and specialization keeps this system stable. Many internal signals are known to guide this process, but less is understood about how whole‑body hormones, such as thyroid hormones, fine‑tune it, especially in the colon, which is a frequent site of tumors in humans.
Taking a single‑cell census after hormone exposure
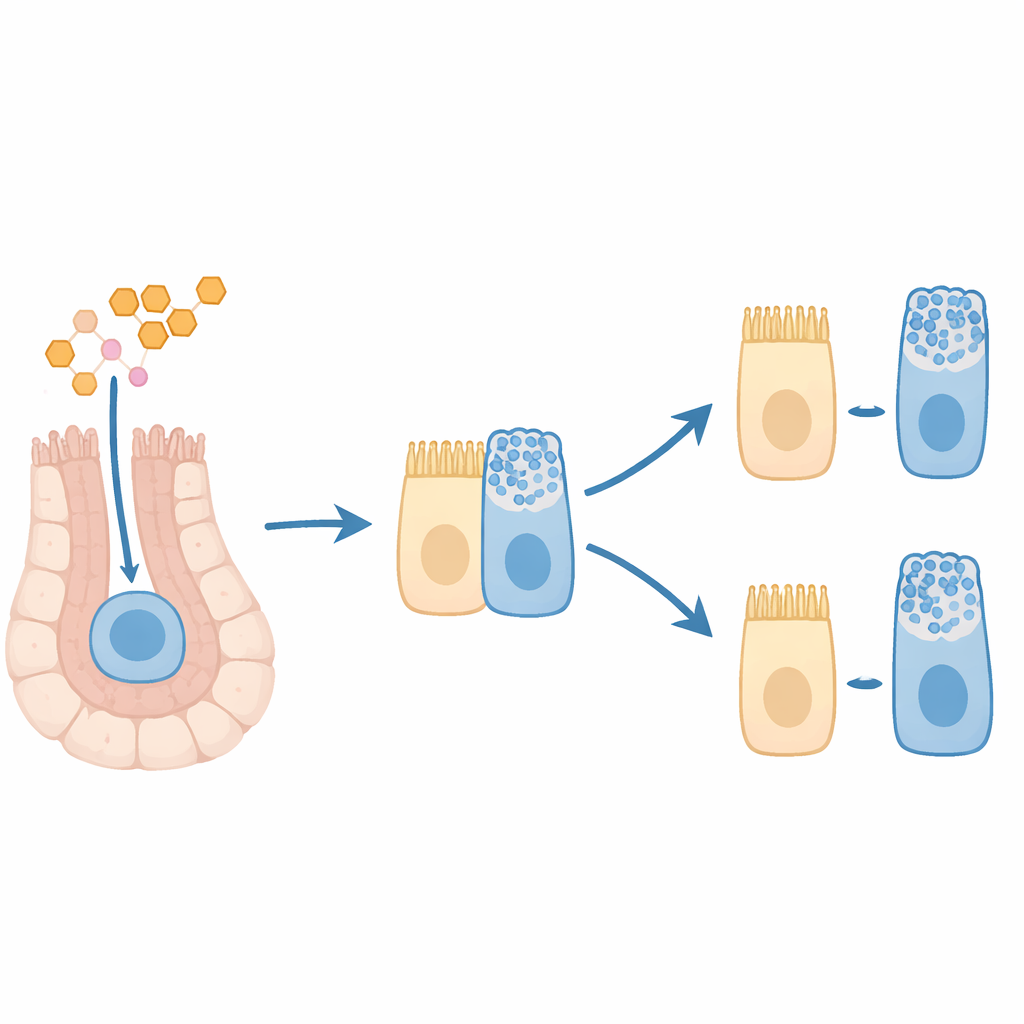
To uncover T3’s role, the researchers injected adult mice with either T3 or a harmless salt solution for two days, then isolated colon epithelial cells for single‑cell RNA sequencing. This technique reads out which genes are active in thousands of individual cells, allowing the team to group cells into distinct types based on their expression patterns. After quality control, they analyzed more than 15,000 cells and identified the expected families: stem/progenitor cells, secretory cells (including goblet and related types), proximal and distal absorptive cells, hormone‑producing cells, and tuft cells. Each cluster showed the gene signatures expected from its known function, confirming that the single‑cell map faithfully captured the colon’s cellular landscape.
Hormone T3 bends cell fates without changing cell types
Comparing T3‑treated and control samples, the authors found that T3 did not dramatically change how many cells fell into each broad category, but it did alter what many of those cells were doing. In several groups—especially stem/progenitor, secretory, and absorptive cells—T3 boosted the activity of genes typically associated with goblet cells, such as those involved in mucus production. Strikingly, this happened even in cells that were not classic goblet cells. At the same time, genes involved in detoxifying chemicals and managing oxidative stress were generally dialed down, while genes linked to defense and inflammation were turned up. Yet markers of the core stem cell identity, such as Lgr5, changed little overall, suggesting that T3 mostly reshapes the behavior of precursor cells rather than fully redefining cell types.
A mixed, stuck‑in‑between cell state
The team then used computational tools to reconstruct likely “trajectories” of differentiation, tracing how cells progress from stem/progenitor states toward mature fates. In control colons, these paths formed clear branches leading to secretory and absorptive lineages. After T3 exposure, the paths blurred: cell branches were less distinct, and certain lineages, such as hormone‑producing and tuft cells, appeared less fully separated from their progenitor origins. A closer look revealed a “mixed” population in the T3‑treated tissue whose cells co‑expressed markers of early decision genes for both absorptive and secretory fates, alongside late goblet‑related genes. Importantly, many of these cells also carried high levels of proliferation markers, indicating that they were still actively dividing.
Seeing early goblet‑bound cells in the tissue
To test whether this mixed state shows up in actual colon tissue architecture, the researchers stained colon sections from control and T3‑treated mice for proteins marking proliferation and specific cell types. They observed that T3 did not greatly change the total number of mature goblet cells along the crypts. Instead, T3 caused an accumulation of goblet‑marker‑positive cells at the very bottom of the crypts—the zone normally occupied by stem and early progenitor cells. Many of these mucus‑marker‑positive cells were also positive for a proliferation marker, reinforcing the idea that T3 generates dividing progenitors already leaning toward a goblet fate but not yet fully mature.
What this means for gut health
To a lay observer, the study’s central message is that the thyroid hormone T3 can push colon precursor cells into a hybrid state: they divide like stem‑like cells but already carry features of mucus‑secreting goblet cells, and they appear to stall before fully maturing. Because proper timing of cell fate decisions is essential for a healthy colon lining, such mixed, immature populations could, under prolonged or excessive hormone exposure, become more vulnerable to stress or malignant change. While this work is in mice and focuses on short‑term treatment, it highlights a previously unrecognized way that thyroid hormone shapes colon renewal and raises new questions about how altered thyroid states might influence colon disease and cancer risk in humans.
Citation: Bidoli, C., Reslinger, M., Sieffert, C. et al. Single-cell analysis reveals that the hormone T3 affects colon epithelial differentiation and induces a mixed progenitor-like cell population. Sci Rep 16, 10369 (2026). https://doi.org/10.1038/s41598-026-40397-7
Keywords: colon epithelium, thyroid hormone T3, single-cell RNA sequencing, goblet cell differentiation, intestinal stem cells