Clear Sky Science · en
Computational modeling of seizure reconstruction and attenuation via model reference control
Why calming seizures with smart computers matters
Epileptic seizures can arrive suddenly, disrupting daily life and, in severe cases, threatening safety. Doctors already use electrical stimulation to help some patients, but the exact settings—how strong, how often, and in what pattern to stimulate—are still chosen largely by trial and error. This paper explores a new way to use computers and real brain recordings to "practice" on virtual seizures first, learning how to weaken them on screen before any current is delivered to a patient.
Seeing patterns in stormy brain waves
The authors start from the idea that seizure and non-seizure brain activity each have their own recognizable electrical signature. Using scalp EEG and intracranial EEG from people with generalized tonic–clonic seizures, they compare brain waves recorded during quiet periods and during full seizures. Even without heavy filtering, seizures show much larger swings and more regular, low-frequency bursts than the more irregular, lower-amplitude background activity. In the frequency domain, seizures carry extra power at low frequencies and recruit broader bands over time, suggesting that a few dominant rhythms help drive the abnormal activity.
Building simple stand-ins for complex brains
Instead of trying to reproduce every detail of the brain, the team builds very simple “dummy” models that behave like a single vibrating system—similar in spirit to a mass on a spring or a basic electrical circuit. They identify these models directly from recorded EEG and iEEG signals using mathematical tools based on orthogonal functions (in practice, Fourier-like building blocks). One model is fit to non-seizure data and another to seizure data. On their own, these models are crude, but when they are placed in a feedback loop that continuously nudges the model’s output toward the real recording, the simulated activity closely tracks the true seizure or non-seizure signal. A key tuning knob, called the decay rate, sets how quickly the model is forced to follow the data—and how strong the required input must be.
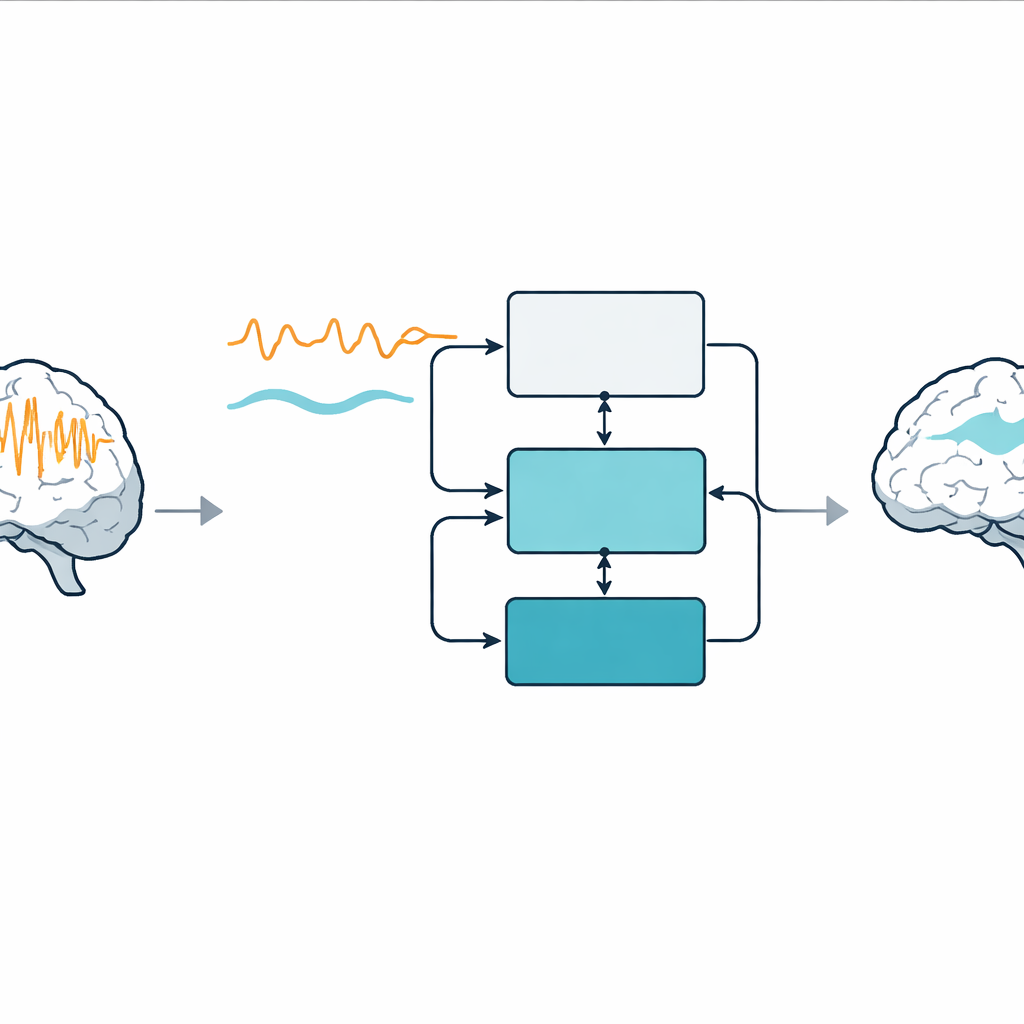
Teaching the model to push seizures back toward normal
Once a seizure model can faithfully replay a patient’s seizure, the authors add a second feedback loop whose job is to make the seizure-like activity resemble the non-seizure pattern. This is known as model reference control: the non-seizure model defines the desired behavior, and the controller computes an electrical stimulus that drives the seizure model toward that reference. Two versions are tested. In the adaptive version, the controller’s gains change over time based on how well it is doing, producing burst-like pulses that weaken the seizure across the whole recording by roughly one third in power. In the fixed-gain version, the controller reuses a gain originally designed to reconstruct non-seizure activity; with suitable tuning, this simpler option can cut seizure power by more than 80% while using relatively modest stimulus strengths.
Finding realistic stimulation recipes
The study also examines how design choices affect both effectiveness and safety. Higher decay rates make the models match the data better but demand stronger stimulation and can actually reduce the percentage of seizure suppression. There is a clear trade-off: better description of the seizure calls for more intense pulses to tame it. By adjusting parameters and deliberately limiting the maximum output, the authors keep simulated voltages mostly within about 5 to 18 volts and frequencies from roughly 2 to 80 hertz—ranges that overlap with those already used in clinical brain stimulation for epilepsy and other disorders. They show that methods such as saturating the output or switching the controller on and off can further cap intensity while still achieving meaningful attenuation.
From virtual control loops to bedside tools
Although these experiments are entirely computational, they outline a practical roadmap. In everyday monitoring, long stretches of non-seizure EEG could be used to keep updating a simple reference model and its controller. When a seizure is detected, the same controller—designed without needing to see that seizure in advance—could drive an open-loop stimulator that delivers pulses with shapes, frequencies, and amplitudes informed by the virtual experiments. A separate monitoring system would judge whether the real seizure is subsiding and adjust or limit the input accordingly. In plain terms, this work suggests that computers can rehearse how best to calm seizures using past recordings, offering physicians clearer guidance on “how to stimulate” rather than relying purely on trial and error.
Citation: Brogin, J.A.F., Faber, J., Ryvlin, P. et al. Computational modeling of seizure reconstruction and attenuation via model reference control. Sci Rep 16, 9951 (2026). https://doi.org/10.1038/s41598-026-39645-7
Keywords: epilepsy, seizure control, brain stimulation, computational modeling, EEG