Clear Sky Science · en
Transmission of Salmonella clones between different animal species in a horse and cattle breeding region in Japan
Why germs that jump between barn animals matter
Food poisoning from Salmonella is a global problem that can start long before food reaches our kitchens. It often begins on farms, where the same germs can move quietly between different animals and then into the human food chain. This study looks closely at a busy horse and cattle breeding region in northern Japan to see how a particularly successful type of Salmonella spreads between species, and whether it is part of a local problem, a global wave of infection, or both.
A hardy germ with a taste for many hosts
The researchers focused on a form of Salmonella, called sequence type 34 (ST34), that is now one of the most common causes of diarrhea in both people and animals around the world. Unlike some Salmonella strains that specialize in one host, ST34 freely infects many mammals and birds. It often carries multiple genes that make it resistant to important antibiotics and able to tolerate metals such as copper or zinc—traits that help it survive on modern farms. Japan has seen repeated waves of different Salmonella strains over the past four decades, and ST34 has become the dominant type in cattle since the 2010s.
Tracing infections across farms and years
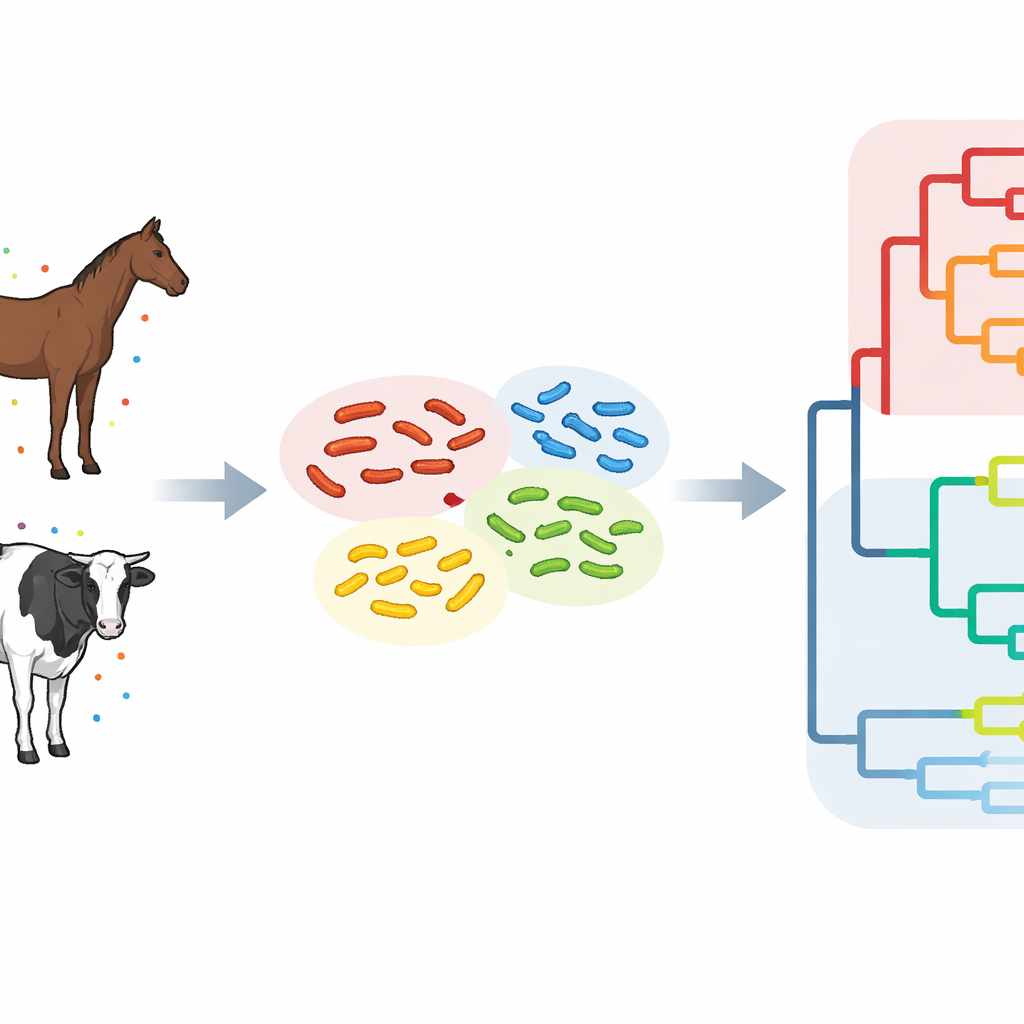
To understand how ST34 behaves where horses and cattle live side by side, the team analyzed 42 Salmonella samples collected from animals in one district of Hokkaido between 1981 and 2023. Using whole-genome sequencing, they read nearly the entire DNA code of each bacterial isolate and compared tiny differences, known as single-nucleotide changes, across their core genomes. Strains that differed by fewer than 25 such changes were grouped into five closely related "clusters," indicating they had separated from a common ancestor fairly recently. In most clusters, nearly identical bacteria were found in both horses and cattle on different farms, sometimes years apart and tens of kilometers away, strongly pointing to cross-species and between‑farm transmission.
Local outbreaks within a global family tree
The scientists then placed the Hokkaido strains into a global context by comparing them with nearly 500 ST34 genomes from 28 countries. This broader analysis revealed three major branches, or clades, on the Salmonella family tree. Two branches consisted largely of strains that had circulated within Japan and were labeled Japanese epidemic lineages, while the third branch contained a mix of strains from Europe, Asia, the Americas, Africa, and Oceania and was considered the global lineage. Three of the Hokkaido clusters fell squarely within one Japanese epidemic lineage, showing that local horse and cattle infections were part of a wider, long‑running national problem. Another cluster grouped instead with strains from East Asian countries such as China and South Korea, and yet another showed genetic ties to European strains, hinting at repeated introductions from abroad.
Hidden travel routes and rising drug resistance
By estimating when different branches split from one another, the team concluded that Japan’s main ST34 lineage separated from the global lineage in the late 1990s or early 2000s and then adapted to food animals within the country. At the same time, at least one East Asian sublineage carrying extra drug-resistance genes appears to have entered Japan separately. While the exact paths are unclear, the patterns suggest that animal movement, contaminated feed, vehicles, or even wild animals such as birds and raccoons could be shuttling these bacteria between farms and species. Nearly all of the strains carried a core set of resistance genes to common antibiotics, and some carried additional genes that could make treatment more difficult if these bacteria reach people.
What this means for farms and food safety
For a general reader, the key message is that dangerous germs do not respect species boundaries or farm fences. In this Japanese district, the same Salmonella clones were found in both horses and cattle and could be genetically linked to larger national and international waves of infection. The study shows that careful DNA tracking can reveal how quietly these bacteria move across animals, regions, and borders. To reduce the risk of food poisoning and animal disease, disease control programs cannot focus on a single species or a single country: they must watch for cross-species jumps, limit the spread of drug-resistant strains, and treat farms, wildlife, and global trade as parts of one connected system.
Citation: Arai, N., Niwa, H., Uchida-Fujii, E. et al. Transmission of Salmonella clones between different animal species in a horse and cattle breeding region in Japan. Sci Rep 16, 12412 (2026). https://doi.org/10.1038/s41598-026-39311-y
Keywords: Salmonella, cross-species transmission, antibiotic resistance, livestock, genome sequencing