Clear Sky Science · en
AI-powered ultrasound radiofrequency analysis for non-invasive pediatric liver fat quantification
Why children’s liver fat matters
More children than ever are developing excess fat in their livers, a condition linked to obesity and future diabetes and heart disease. Detecting and tracking this liver fat early is essential, but today’s most accurate test—an MRI scan—is expensive, not widely available, and can be difficult for kids to tolerate. This study explores whether ordinary ultrasound machines, assisted by artificial intelligence (AI), can measure liver fat in children as accurately as MRI without needles, radiation, or long hospital visits.
A growing problem in young bodies
Metabolic dysfunction–associated steatotic liver disease (MASLD), formerly grouped under nonalcoholic fatty liver disease, has become the leading chronic liver condition in children. It affects up to one in ten kids overall and as many as half of those with obesity. Unlike adults, children may struggle with breath-holding, staying still, or undergoing invasive tests like liver biopsy, which limits how often doctors can safely and comfortably check their livers. MRI-based fat measurements are highly reliable but costly and sometimes require sedation. Standard ultrasound is far easier to use but mainly gives a grainy gray picture that depends heavily on the operator’s eye and misses mild disease. The central question is whether deeper information hidden in the raw ultrasound signals can close this gap.
Listening to raw sound instead of just looking at the image
Modern ultrasound machines record not only the familiar gray image but also the underlying radiofrequency (RF) echoes—rich sound wave data that are usually discarded. The researchers prospectively enrolled 40 children and teens being evaluated for fatty liver. On the same day, each child had a specialized ultrasound exam that stored RF data, a standard ultrasound-based measure of sound weakening in the liver (called an attenuation parameter), basic blood tests, and an MRI scan that provided a precise liver fat percentage. Using these data, the team built two types of AI systems. One group of models analyzed handcrafted numeric patterns, or “radiomics,” extracted from the RF signals. The other used deep learning, in which neural networks learned patterns directly from RF-based images, with options to add the ultrasound attenuation measure and a key blood enzyme level.
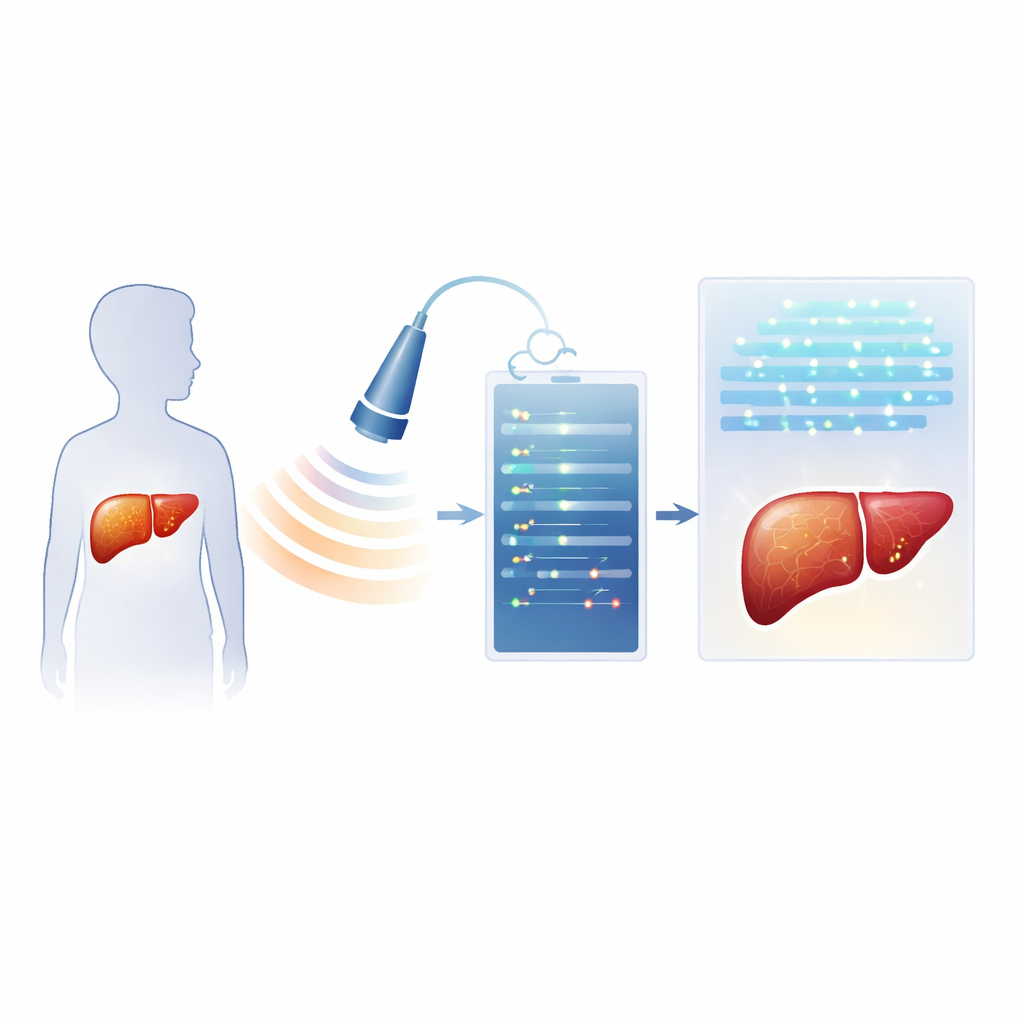
What the AI systems learned from children’s livers
The researchers compared multiple input combinations: RF data alone; RF plus the conventional gray ultrasound image; RF plus the attenuation measure; and a full blend of RF, attenuation, and a liver enzyme from blood tests called ALT. They found that RF data were crucial: models that relied only on standard images performed noticeably worse. Adding the gray image to RF did not help and sometimes hurt accuracy, suggesting that compressing rich sound information into a simple picture throws away useful detail. In contrast, combining RF data with the quantitative attenuation measure sharply improved performance, and adding ALT provided a further boost, especially for the deep learning model. This reflects how real-world liver assessment benefits from mixing imaging signs with chemical clues from the bloodstream.
How close to MRI did ultrasound plus AI get?
The best radiomics model, using a method called gradient boosting, explained about four-fifths of the variation in MRI-measured liver fat and typically missed the true value by less than 2 percentage points. The top deep learning network showed similar though slightly lower accuracy. An agreement analysis comparing each AI estimate to MRI for many image slices showed only tiny average differences—well under half a percentage point—with most errors falling within a few percentage points. In practical terms, this means that the AI-enhanced ultrasound system could track liver fat levels in children closely enough to mirror MRI results, without requiring the MRI itself.
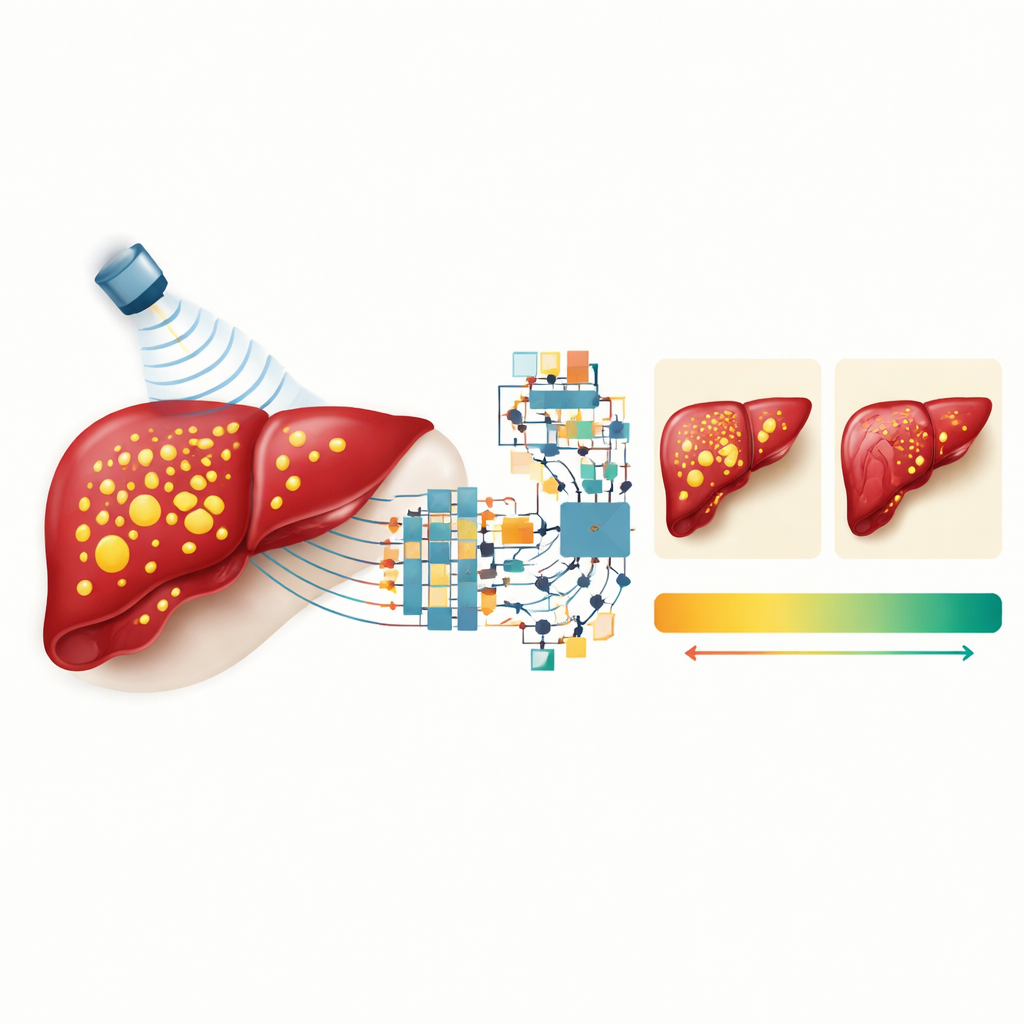
What this could mean for children and clinics
The study suggests that ordinary ultrasound machines, upgraded to save raw sound data and paired with AI software, could become a powerful, child-friendly tool for measuring liver fat. Such systems are cheaper, more accessible, and free of radiation, making them well suited for repeated monitoring as children grow or begin lifestyle and medical treatments. While the work was done in a single hospital with a modest number of patients and one type of scanner, and still needs confirmation in larger, multi-center studies, it points toward a future where pediatric liver fat can be tracked in regular clinic visits. For families, this may mean earlier detection of trouble, more frequent checks without added risk, and better chances to protect children from long-term liver damage.
Citation: Choi, G., Ham, S., Je, BK. et al. AI-powered ultrasound radiofrequency analysis for non-invasive pediatric liver fat quantification. Sci Rep 16, 13798 (2026). https://doi.org/10.1038/s41598-026-37862-8
Keywords: pediatric fatty liver, ultrasound imaging, artificial intelligence, liver fat quantification, childhood obesity