Clear Sky Science · en
A single cell transcriptional profile of benign prostatic hyperplasia
Why this matters for men as they age
As men grow older, their prostate gland often enlarges, squeezing the tube that carries urine and causing frequent nighttime trips to the bathroom, weak stream, or difficulty starting to pee. This condition, called benign prostatic hyperplasia (BPH), affects roughly half of men by age 60, yet doctors still do not fully understand why the gland grows and stays inflamed. Current medicines can ease symptoms but do not work for everyone, leaving many men to undergo surgery. This study takes a cell-by-cell look inside enlarged prostates to discover which specific cells are driving both growth and inflammation, with the long-term goal of pointing to new, more targeted treatments.
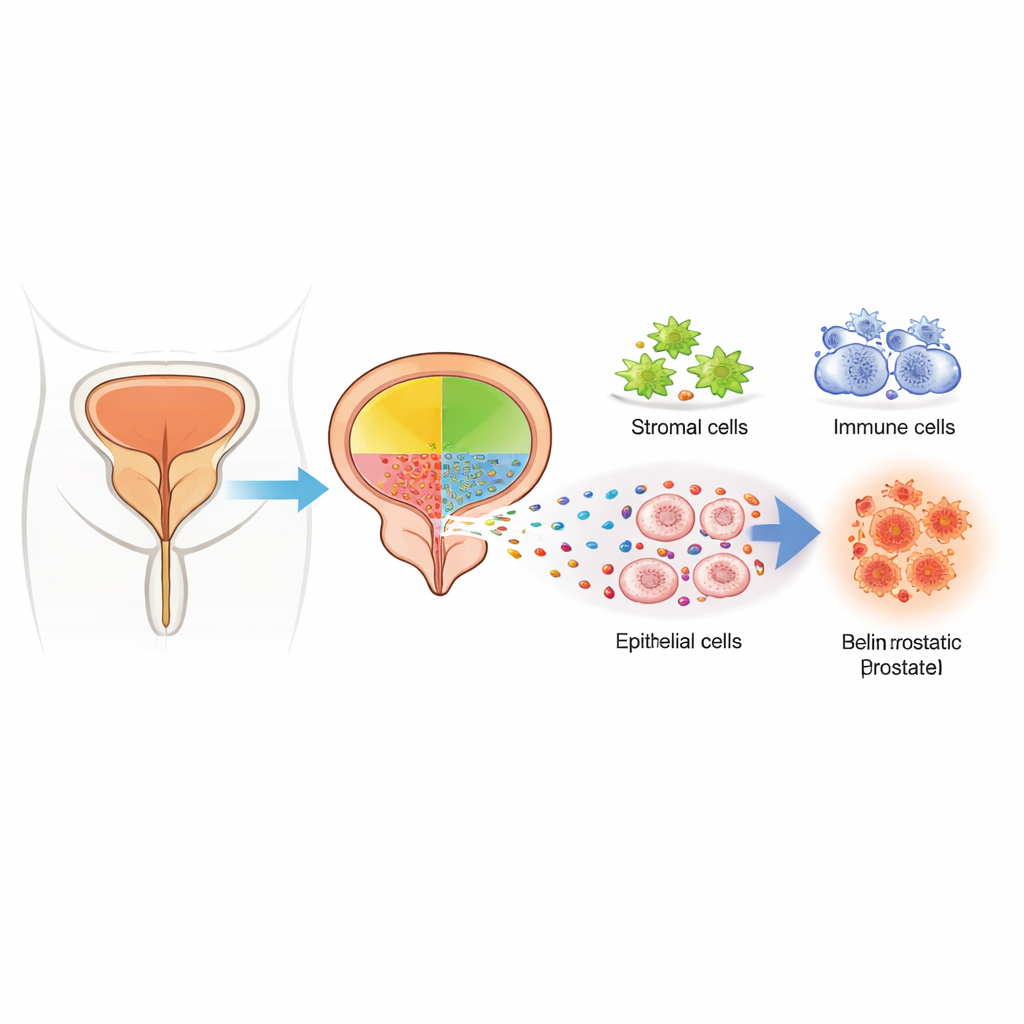
Looking at the prostate one cell at a time
The researchers obtained tissue from 15 men undergoing a modern surgical procedure (HoLEP) to remove overgrown prostate tissue. Instead of grinding the tissue into a composite sample, they separated it into more than 16,000 individual cells and measured which genes were switched on in each cell. Using computational methods to group cells with similar gene activity, they identified 14 major cell types, including gland-lining epithelial cells, supporting stromal cells, and a variety of immune cells such as T cells and macrophages. This fine-grained map allowed them to see not just which kinds of cells were present, but also how those cells differed in men with larger, more inflamed prostates.
Inflamed support cells around the glands
Among the supporting stromal cells, the team found distinct groups of fibroblasts, the connective tissue cells that surround and support the glands. One subgroup, called peri-epithelial fibroblasts because they sit right next to the glandular cells, showed particularly strong activation of gene programs linked to inflammation, including a pathway known for driving inflammatory signals (TNF/NF-κB). Men with very large prostates tended to have more of these peri-epithelial fibroblasts and higher expression of these inflammatory genes, suggesting that this cell type helps maintain the chronic irritation and tissue remodeling seen in BPH. Interestingly, patients taking one common class of BPH drugs (5‑alpha-reductase inhibitors) had fewer fibroblasts and lower inflammatory gene activity in these cells.
A rare growth-prone gland cell with a loud voice
Within the gland-lining epithelial cells, the scientists uncovered a rare luminal subgroup that looked different from the bulk of the gland cells. These cells showed a gene pattern resembling stem or progenitor cells, which are capable of giving rise to other cell types, and analysis of their progression suggested they sit at an early point in the luminal cell family tree. They also expressed high levels of genes tied to cell growth and protein production, hinting that they may be primed for proliferation. Crucially, this subgroup produced large amounts of a signaling molecule called MIF, previously linked to inflammation and cell growth in the prostate. The authors detected this same progenitor-like luminal signature, although very rarely, in other prostate datasets, including cancer and noncancer samples, implying it may represent a shared growth-prone state.
Immune cells fed by inflammatory signals
On the immune side, the study identified several flavors of macrophages—immune cells that can either fuel or calm inflammation. Two macrophage groups in particular were abundant in BPH and carried strong inflammatory gene signatures. When the researchers modeled signaling between cell types, they found that the progenitor-like luminal cells were predicted to communicate intensely with both fibroblasts and macrophages through MIF and its partner receptors on the surface of those cells. This network of signals could attract and activate immune cells and further stimulate fibroblasts, creating a self-reinforcing loop of inflammation and tissue growth in the prostate.
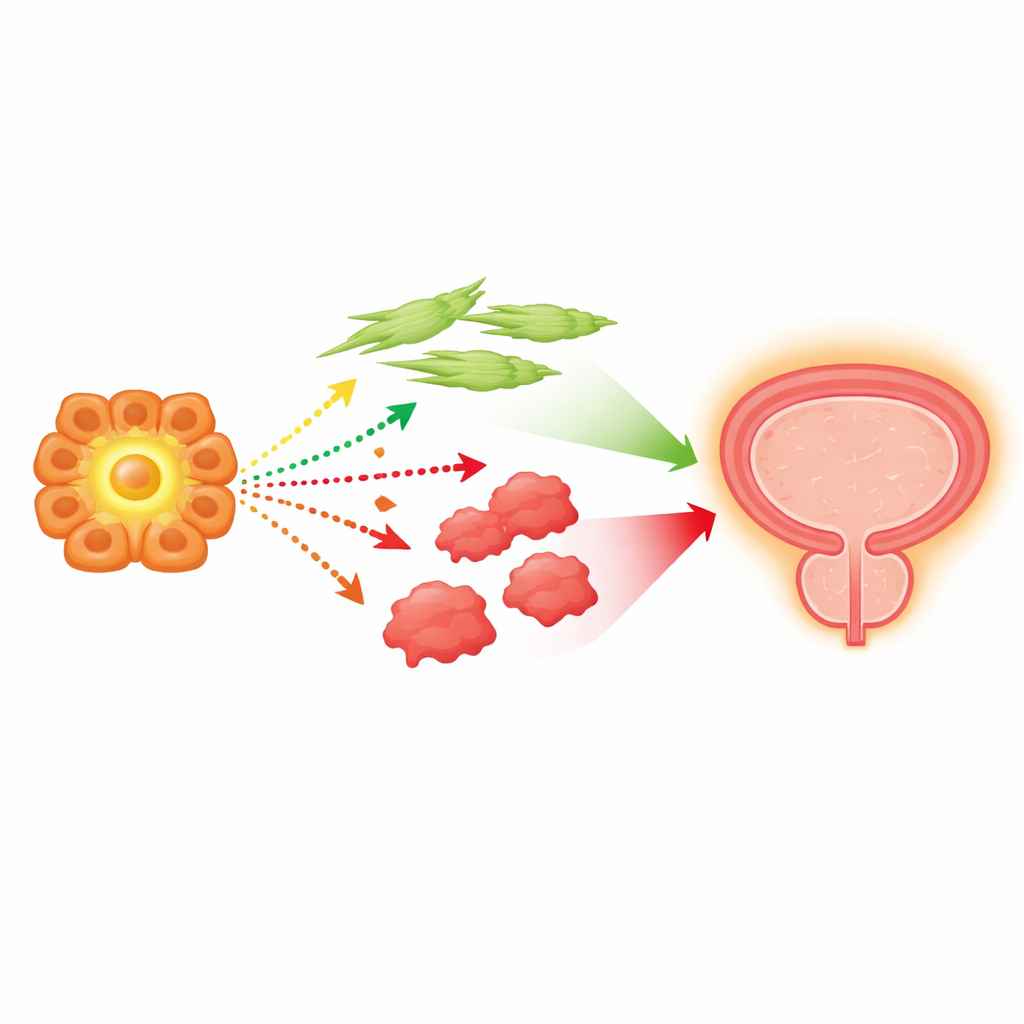
What this could mean for future treatments
Taken together, the findings paint a picture in which a rare, growth-prone gland cell population sends out powerful chemical messages that activate nearby support and immune cells, which in turn help sustain chronic inflammation and enlargement of the prostate. Because many of these signals funnel through MIF and its main receptor, CD74, the study highlights this pathway as a promising target for new drugs—especially for men whose BPH remains highly inflamed despite current therapies. Experimental MIF‑blocking compounds already being tested in cancer and autoimmune diseases could, one day, be adapted to quiet this cellular conversation in the prostate, offering more precise relief for bothersome urinary symptoms in aging men.
Citation: Unno, R., Akutagawa, J., Song, H. et al. A single cell transcriptional profile of benign prostatic hyperplasia. Sci Rep 16, 9556 (2026). https://doi.org/10.1038/s41598-025-02417-w
Keywords: benign prostatic hyperplasia, single-cell RNA sequencing, prostate inflammation, fibroblasts and macrophages, MIF signaling