Clear Sky Science · en
ZFP148 is a transcriptional repressor of cytolytic effector CD8+ T cell differentiation
Helping the Body’s Assassins Do Their Job
Our immune system relies on specialized “killer” T cells to hunt down virus-infected and cancerous cells. Yet in long battles against chronic infections or tumors, these cells often tire out and lose their punch. This study reveals a molecular brake, a protein called ZFP148, that dampens the killing power of these T cells. By lifting this brake in mice and examining human cancers, the researchers show a way to boost the immune system’s natural ability to control disease and to improve responses to modern cancer immunotherapies.
When Killer Cells Grow Weary
In chronic infections and cancer, killer CD8 T cells face persistent exposure to their targets. Over time, many of them enter a state known as exhaustion, where they express inhibitory molecules and gradually lose their capacity to divide and kill. A smaller group of “progenitor” cells can still renew themselves and give rise either to exhausted cells or to highly destructive effector cells that excel at destroying infected or malignant cells. Understanding what tilts this fork in the road—toward powerful effectors or toward exhaustion—is a central question for improving vaccines and immunotherapy.
A New Molecular Brake Comes Into View
The team focused on ZFP148, a DNA-binding protein previously studied in other cell types but not well understood in killer T cells. They found that ZFP148 is most abundant in progenitor CD8 T cells and declines as cells become either short-lived effectors or exhausted. Using a mouse model of chronic viral infection, they selectively removed ZFP148 only from CD8 T cells. Without this molecule, the animals produced a larger pool of cytolytic effector cells—those rich in cell-destroying enzymes—and a smaller pool of exhausted cells. These effector cells also circulated more readily through the body and showed stronger tumor-cell killing in laboratory tests.
Rewiring the Genetic Control Panel
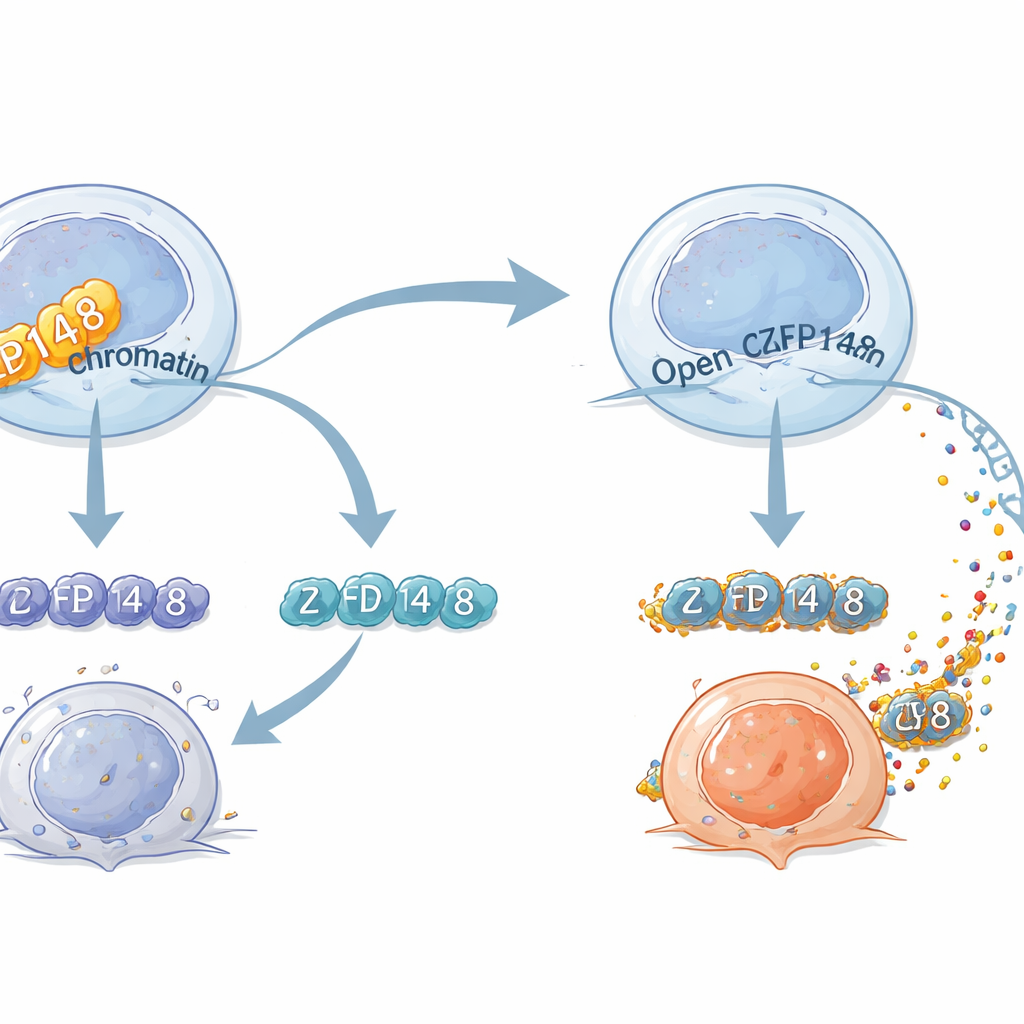
To see how ZFP148 reshapes T cell behavior, the researchers combined single-cell RNA sequencing (which genes are turned on) with single-cell chromatin profiling (which DNA regions are open for business). In normal mice, ZFP148 helped keep regions that drive effector programs relatively closed, while favoring patterns linked to stem-like or exhausted states. In the absence of ZFP148, DNA regions controlling effector molecules and natural-killer-like features became more accessible, and the cells’ gene activity shifted toward a highly cytotoxic identity. A key target emerged: another protein, KLF2, which supports the generation and movement of effector T cells. ZFP148 physically bound to a regulatory segment near the Klf2 gene and suppressed its activity. Removing either KLF2 or that specific DNA element erased the effector-boosting benefits of ZFP148 loss, showing that a ZFP148–KLF2 axis acts as a central switch for T cell fate.
Supercharging Immunotherapy
The researchers then moved to cancer models. In mice bearing colon tumors, CD8 T cells lacking ZFP148 inside the tumor showed stronger activation markers and more of the potent, granule-filled effector cells. On their own, these changes modestly improved tumor control. But when combined with an antibody that blocks the immune checkpoint PD-1—a widely used cancer immunotherapy—the benefits were striking. Tumors shrank more and mice lived longer when their CD8 T cells lacked ZFP148, compared with standard PD-1 blockade alone. The enhanced response was tied to an expanded population of rapidly dividing, highly cytotoxic T cells in the tumor microenvironment.
Clues from Human Tumors
To test whether the same principles apply to people, the authors reanalyzed large collections of single-cell data from human cancers and performed gene editing in human T cells in the lab. Tumor-infiltrating CD8 T cells with low levels of the human counterpart, ZNF148, expressed more effector molecules and natural-killer-like receptors, and scored higher on signatures of cytotoxic activity and T cell receptor signaling. Experimentally reducing ZNF148 in human CD8 T cells increased production of key inflammatory and killing factors. Across several cancer cohorts, patients whose tumors harbored lower ZNF148, or whose T cells bore a “ZFP148-loss-like” gene pattern, tended to live longer and respond better to checkpoint therapies such as PD-1 or CTLA-4 blockade.
Why This Matters for Future Treatments
This work positions ZFP148 as a conserved molecular checkpoint that steers killer T cells away from becoming full-fledged attackers. By suppressing KLF2 and keeping key DNA regions closed, ZFP148 limits the formation and expansion of the most effective cancer- and virus-fighting cells. In mice, disabling this brake amplifies the benefits of existing PD-1–based immunotherapies, and patterns seen in human tumors suggest the same strategy could help identify patients more likely to respond—or serve as a new target for drugs designed to boost immunity. In simple terms, dialing down ZFP148 could help our body’s own assassins stay energized longer and hit their targets harder.
Citation: Xiao, T., Chen, X., Song, NJ. et al. ZFP148 is a transcriptional repressor of cytolytic effector CD8+ T cell differentiation. Nat Immunol 27, 827–840 (2026). https://doi.org/10.1038/s41590-026-02461-2
Keywords: CD8 T cells, T cell exhaustion, cancer immunotherapy, transcription factors, chronic infection