Clear Sky Science · en
A µ-opioid receptor superagonist analgesic with minimal adverse effects
Why a safer painkiller matters
Modern medicine leans heavily on opioid drugs to relieve severe pain, yet these same medicines can slow breathing, cause dependence, and drive addiction. This study describes a new laboratory-made opioid, called DFNZ, that powerfully eases pain in rodents while largely sidestepping the dangerous effects that make today’s opioids so risky. If its promise holds up in people, DFNZ could point the way toward pain treatments that do not fuel the opioid crisis.
A new twist on a dangerous drug family
DFNZ belongs to the “nitazene” family, a group of synthetic opioids that recently appeared in the illicit drug supply and are often even stronger than fentanyl. Earlier nitazenes were abandoned as medicines because tiny doses could kill by shutting down breathing. Here, chemists modified one such compound to create DFNZ, which still activates the same pain-relieving receptor in the brain—the µ-opioid receptor—but behaves very differently in the body. In test-tube experiments, DFNZ switched this receptor on more strongly than a standard laboratory opioid, earning it the label “superagonist.” Yet the real surprise came when the team examined how and where DFNZ acts in living animals.
Powerful pain relief without the usual toll
In rats and mice, DFNZ produced strong, dose-dependent pain relief in several models, including acute heat pain and persistent inflammatory pain. The effective doses were similar to or lower than those of common medical opioids. However, when the same animals were tested for hallmark side effects, DFNZ stood out. Repeated treatment did not produce tolerance (a fading of pain relief that pushes doses higher) or mechanical hypersensitivity (a heightened pain response), problems commonly seen with drugs like fentanyl. When the researchers forced withdrawal with an opioid blocker after a week of exposure, animals pretreated with DFNZ showed far milder signs of distress than morphine-treated animals, suggesting a weaker grip on the brain’s dependence circuits.
How the drug stays out of trouble in the brain
The key to DFNZ’s unusual safety appears to be how it moves through, and is kept out of, the brain. Imaging and chemical measurements showed that DFNZ has limited access to brain tissue because it is actively pumped out by two transporter proteins that guard the blood–brain barrier. As a result, even when blood levels are high enough to block pain, occupancy of µ-opioid receptors in key brain regions remains modest. At these therapeutic doses, DFNZ did not reduce oxygen levels in a deep brain structure linked to breathing and reward, whereas fentanyl and a related nitazene clearly induced hypoxia. Only when the researchers disabled the transporters or pushed DFNZ to very high doses did signs of brain oxygen loss emerge, underscoring the importance of this “safety valve” at the barrier between blood and brain.
Uncoupling pain relief from reward
Opioids are so addictive partly because they strongly boost dopamine signaling in the brain’s reward pathways and are readily self-administered by animals. Using optical sensors, the team found that DFNZ triggered only modest, delayed changes in dopamine activity, favoring slow, tonic signals over the sharp spikes thought to reinforce drug-taking. In self-administration experiments, rats would work to receive DFNZ, but far less vigorously than for heroin or for the related nitazene FNZ. When the drug was removed, lever pressing for DFNZ dropped off almost immediately, and re-exposure did not rekindle drug seeking. Molecular tests suggested one reason: DFNZ was less effective at a specific receptor partnership in the midbrain (a complex of the µ-opioid receptor with the galanin 1 receptor) that helps drive dopamine release. This receptor-level selectivity appears to let DFNZ blunt pain without strongly tapping into the circuitry that underlies craving and relapse.
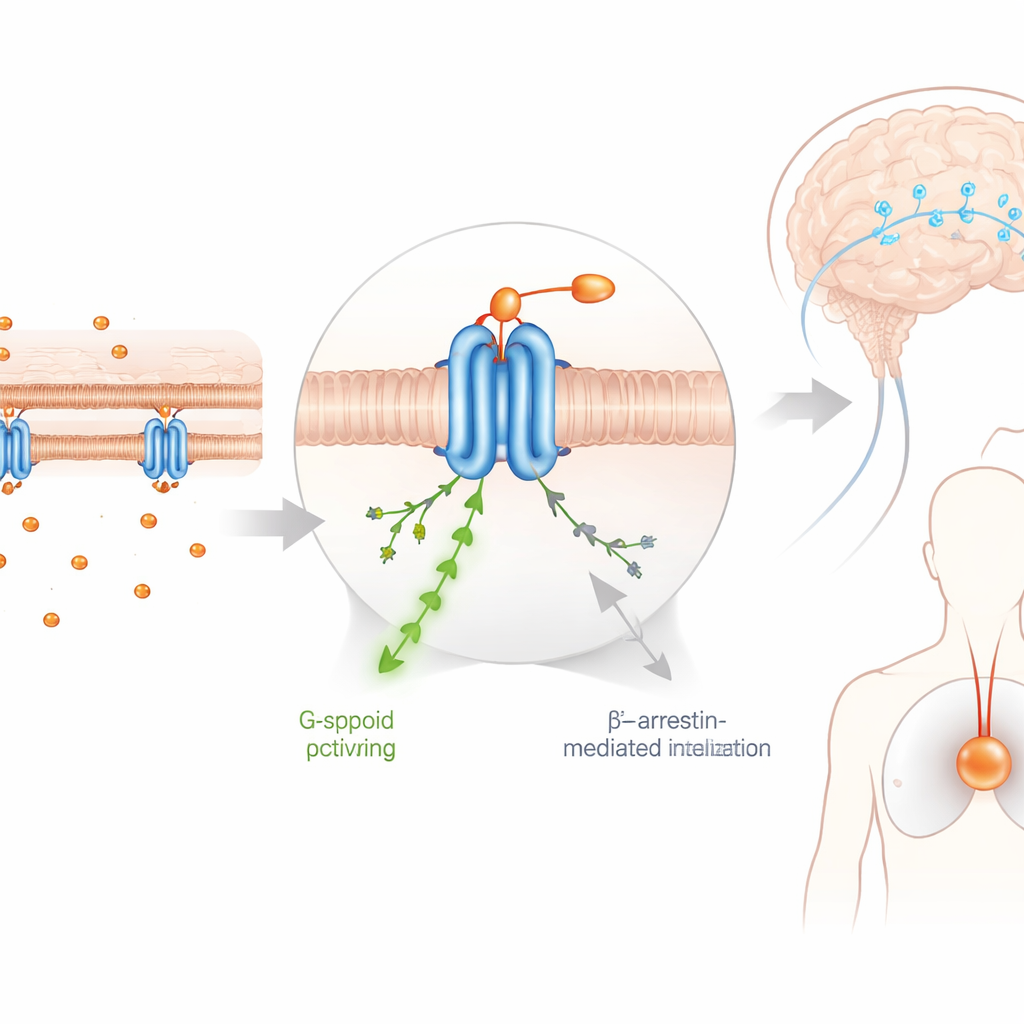
What this could mean for future treatments
Taken together, the findings challenge a long-standing belief that highly powerful µ-opioid drugs are inevitably dangerous. In rodents, DFNZ combines strong analgesia with minimal respiratory depression, little tolerance or withdrawal, and reduced reinforcement, all while dampening activity in reward circuits. Because it still reaches some brain receptors and may act partly outside the brain, DFNZ embodies a new design strategy: shaping how, when, and where an opioid engages its target, rather than simply dialing its strength up or down. The authors argue that DFNZ—and similar compounds yet to be created—deserve further study as potential pain relievers and even as long-acting maintenance therapies for opioid use disorder, offering the benefits of drugs like methadone with a lower risk of overdose.
Citation: Gomez, J.L., Ventriglia, E.N., Frangos, Z.J. et al. A µ-opioid receptor superagonist analgesic with minimal adverse effects. Nature 652, 1393–1404 (2026). https://doi.org/10.1038/s41586-026-10299-9
Keywords: opioid analgesics, mu-opioid receptor, nitazenes, addiction risk, dopamine signaling