Clear Sky Science · en
Rapid mid-Cretaceous diversification of squid and cuttlefish preceded radiation into coastal niches
Why squid history matters
Squid and cuttlefish are among the ocean’s most charismatic hunters, famous for their shifting colors and remarkable intelligence. Yet their own family history has been surprisingly murky. This study uses newly decoded genomes to redraw the squid and cuttlefish family tree and to explain how these animals survived ancient crises, diversified in the deep sea, and later exploded into today’s rich variety of coastal forms.
The big picture of squid kinships
For decades, biologists have debated how the many kinds of squid and cuttlefish are related—especially how their internal shells evolved and how they spread between deep and shallow seas. The authors combined three new, high-quality genomes from a pygmy squid, a Japanese flying squid, and the deep-sea ram’s horn squid with existing genomic and transcriptomic data from other cephalopods. By comparing thousands of genes shared across species, they built a robust evolutionary tree that clarifies which lineages truly share a common ancestry and which similarities arose independently.

Two major squid worlds
The new tree reveals a clean split between two main branches. One, which the authors call Acorneata, contains open-ocean dwellers such as large oceanic squids and the ram’s horn squid. The other, called Corneata, includes coastal and shallow-water groups such as cuttlefish, neritic squids, bobtail squids, and pygmy squids. Members of Corneata share two features absent from their open-ocean cousins: a transparent covering over the eye (a cornea) and small pockets that store their shooting tentacles. These shared traits, together with the gene-based tree, support the idea that coastal squids and cuttlefish form a natural group that split off from deep-water ancestors.
A burst of change in the age of dinosaurs
Using a molecular clock approach, which estimates ages from the accumulation of genetic changes and key fossils, the team dated the origin of modern squid and cuttlefish orders to about 101 million years ago, during the mid-Cretaceous. At that time, global sea levels were high and shallow coastal waters were often low in oxygen, making them harsh places to live. The authors argue that the main lineages of modern squid and cuttlefish therefore arose in the deeper open ocean, where stable, cooler, and more oxygenated “refugia” existed. This early diversification set a “long fuse”: the major branches split in the Cretaceous, but most of today’s coastal diversity appeared only tens of millions of years later, after the dinosaurs died out.
From heavy shells to light skeletons
Another puzzle is how the internal supports of these animals—the cuttlebone of cuttlefish, the thin gladius of many squids, and the coiled shell of the ram’s horn squid—are related. Fossils and genetic evidence together suggest a stepwise trend: an ancestral chambered shell was progressively simplified and lightened. In the open-ocean lineages, one branch kept and elaborated a mineralized, chambered shell (as in the ram’s horn squid), while the oceanic squids reduced it to a mostly organic blade-like gladius with only faint remnants of chambers. In coastal groups, cuttlefish reworked all three ancestral shell parts into the buoyant cuttlebone, while many bobtail and pygmy squids drastically shrank or even lost their internal shells altogether. The study also traces the gains and losses of key shell-building genes, showing, for instance, that some coastal squids abandoned certain rigid-shell proteins, possibly favoring lighter, more flexible supports in acidifying, unstable post–mass extinction seas.
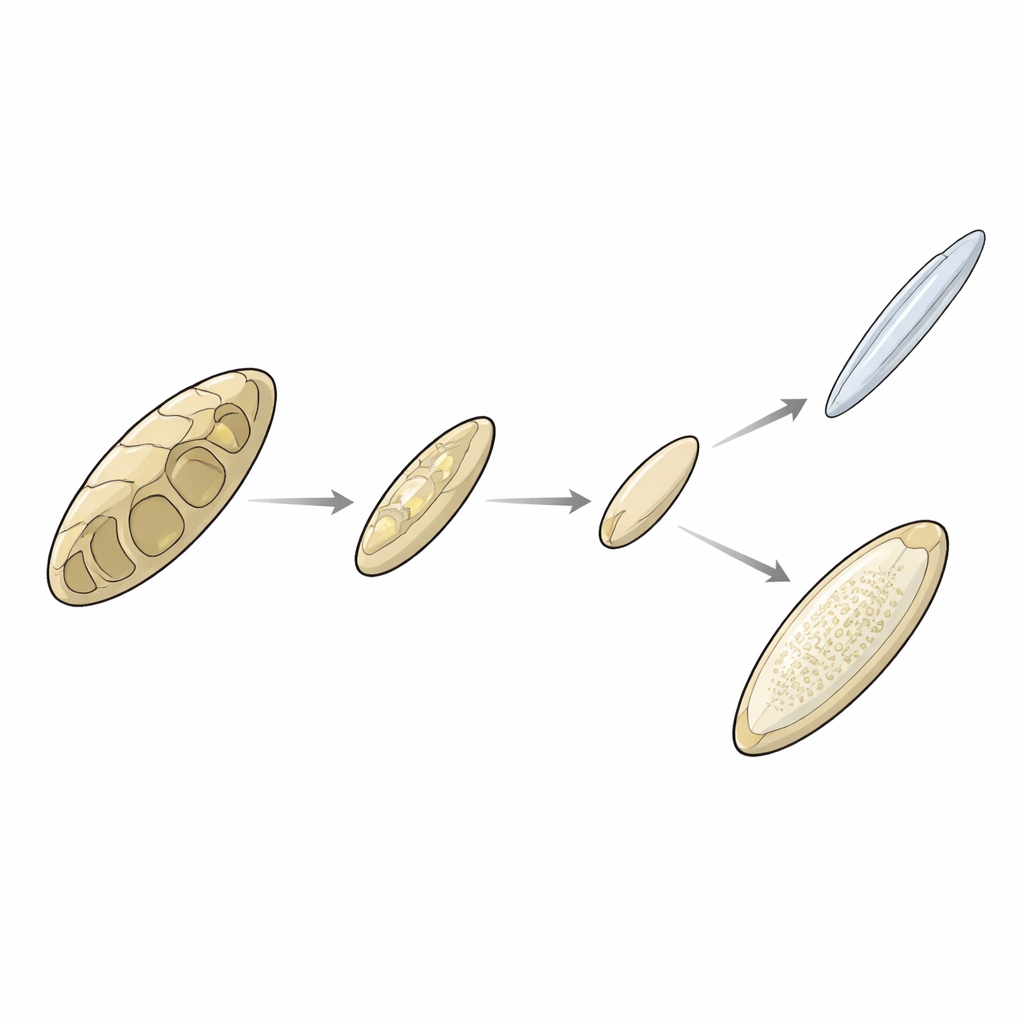
Hidden stories in chromosomes and genes
By aligning chromosomes across species, the authors find that most squid and cuttlefish share a remarkably stable set of 46 chromosome pairs, hinting at a conserved genomic blueprint dating back to early coleoids. Only the rapidly evolving pygmy and bobtail squids show major rearrangements, which may have arisen when their coastal populations were small and fragmented. The team also detects signs of natural selection in genes tied to vision in shallow-water squids, consistent with adaptation to brighter, more variable light, and in energy-producing enzymes of fast-swimming open-ocean squids. In the ram’s horn squid, immune-related gene families have expanded, possibly reflecting the demands of life in the deep sea and complex interactions with microbes.
What this means for understanding squid evolution
Altogether, the study paints squid and cuttlefish as deep-sea survivors that diversified quietly in the open ocean during the Cretaceous, then later surged into recovering coastal ecosystems after the end-Cretaceous mass extinction. Their rich modern diversity—the many shapes of their internal shells, their flexible genomes, and their varied lifestyles—emerges as the outcome of this long fuse: ancient splits followed by much later ecological opportunity. For non-specialists, the key message is that today’s familiar coastal squids and cuttlefish are not primitive shoreline relics, but the descendants of deep-water ancestors that repeatedly reinvented their bodies and genomes to exploit new corners of the sea.
Citation: Sanchez, G., Fernández-Álvarez, F.Á., Bernal, A. et al. Rapid mid-Cretaceous diversification of squid and cuttlefish preceded radiation into coastal niches. Nat Ecol Evol 10, 662–676 (2026). https://doi.org/10.1038/s41559-026-03009-1
Keywords: squid evolution, cuttlefish, Cretaceous oceans, cephalopod genomes, marine diversification