Clear Sky Science · en
A general purposed machine learning interatomic potential for Mg-Al-Si-O system suitable for Earth materials at high pressure and temperature conditions
Peering into Earth’s Hidden Heart
Deep beneath our feet, rocks made of magnesium, aluminum, silicon, and oxygen shape everything from volcanic eruptions to plate tectonics. Yet the extreme pressures and temperatures of Earth’s interior make these materials nearly impossible to study directly in the lab. This paper presents a new computer-based way to mimic how these minerals behave, using modern artificial intelligence to bridge the gap between costly quantum calculations and oversimplified traditional models.
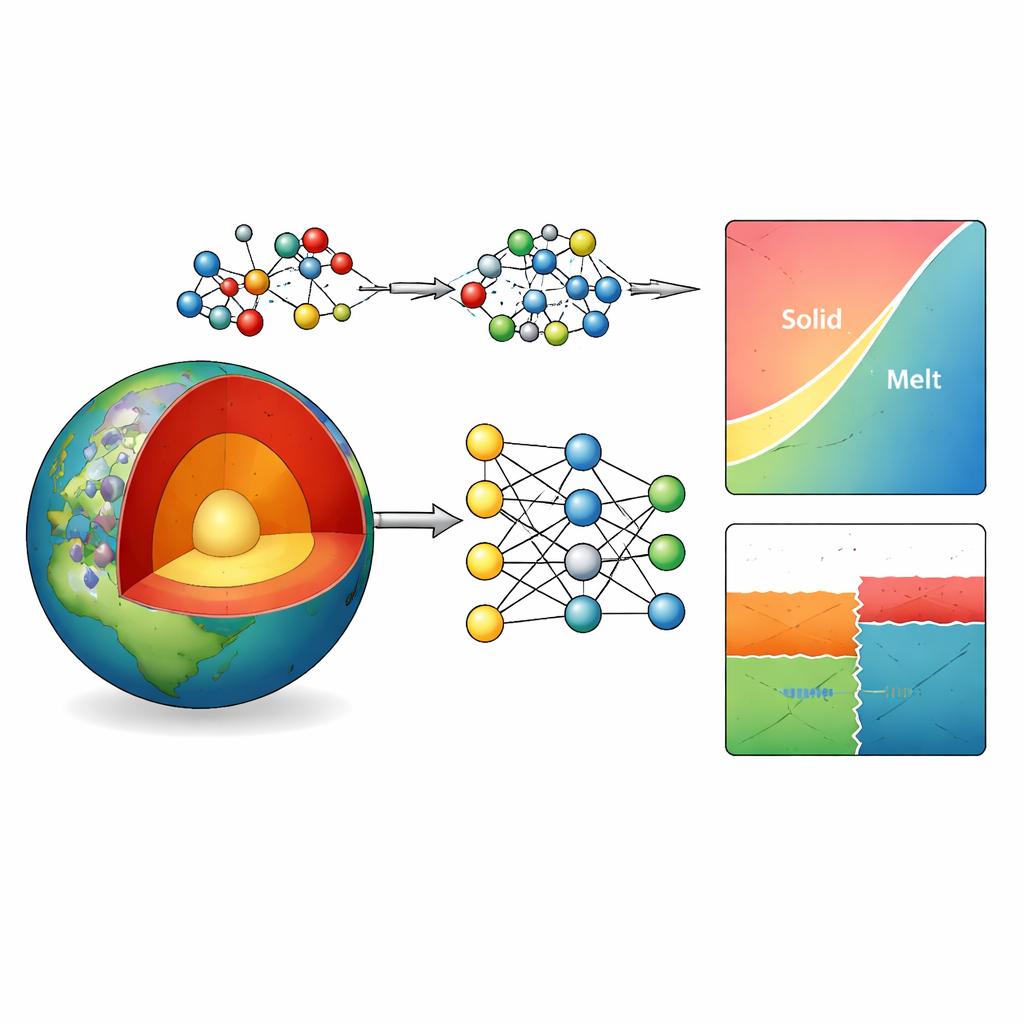
Why Simulating Deep Rocks Is So Hard
Earth’s mantle is dominated by minerals built from magnesium, aluminum, silicon, and oxygen. Their stability and melting behavior control how stiff or runny rocks are, how plates dive into the mantle, and where earthquakes and seismic speed jumps occur. Researchers rely on phase diagrams—maps that show which minerals are stable at a given depth and temperature—to understand these processes. Experiments at hundreds of kilometers depth are extremely challenging, so scientists turn to computer simulations. Classical models run fast but often get the wrong mineral transitions, while more accurate quantum-mechanical methods are far too slow for the large, complex systems geoscientists care about.
Teaching a Neural Network the Rules of Atoms
The authors tackle this bottleneck by building a machine-learning “interatomic potential” for the Mg–Al–Si–O system. In essence, they train a deep neural network to predict how atoms push and pull on each other, based on a large library of example structures. These examples are generated using advanced quantum calculations with a particularly reliable method (called r2SCAN) over a wide range of pressures, temperatures, and mineral and melt types. To sharpen the accuracy even further where it matters most—tiny energy differences that decide which mineral is stable—they add a simple, pairwise Gaussian correction tuned against a trusted thermodynamic database. This hybrid approach cuts the average energy error for 20 common mantle minerals from about 5 to just over 1 kilojoule per mole, without significantly disturbing their volumes.
Checking the Model Against Nature’s Phase Maps
Armed with this refined potential, the team computes phase diagrams for key building-block systems, including pure silica, aluminosilicates, and magnesium silicate compositions that span major mantle minerals like forsterite, wadsleyite, and ringwoodite. Using thermodynamic integration and standard relations from physical chemistry, they trace out the boundaries where one phase gives way to another or to melt. The predicted diagrams closely match experimental results, even capturing high-pressure transitions in silica that were not explicitly included in the training data. Where there are differences—such as a modest shift in the boundary between forsterite and wadsleyite—they can be traced to remaining energy uncertainties of only a few kilojoules per mole, equivalent to errors of a few hundred degrees or a fraction of a gigapascal.
New Views of Interfaces and Atomic Disorder
Because the machine-learned model is both accurate and fast enough for large systems, the authors can explore properties that are nearly inaccessible in the lab. One example is the way aluminum and silicon atoms rearrange within the mineral sillimanite as temperature rises, which they study by combining their potential with Monte Carlo simulations. Another is the free energy of the boundary between solid crystals and molten rock for two representative minerals: periclase (MgO) and forsterite (a form of olivine). Using sophisticated sampling methods, they show that these interfaces have relatively low anisotropy—the difference in surface energy between crystal faces—about 6 percent for periclase and 12 percent for forsterite. They also investigate how uneven stress affects the well-known transformation of low-temperature quartz to its high-temperature form, finding that typical geological levels of differential stress shift the transition only slightly.
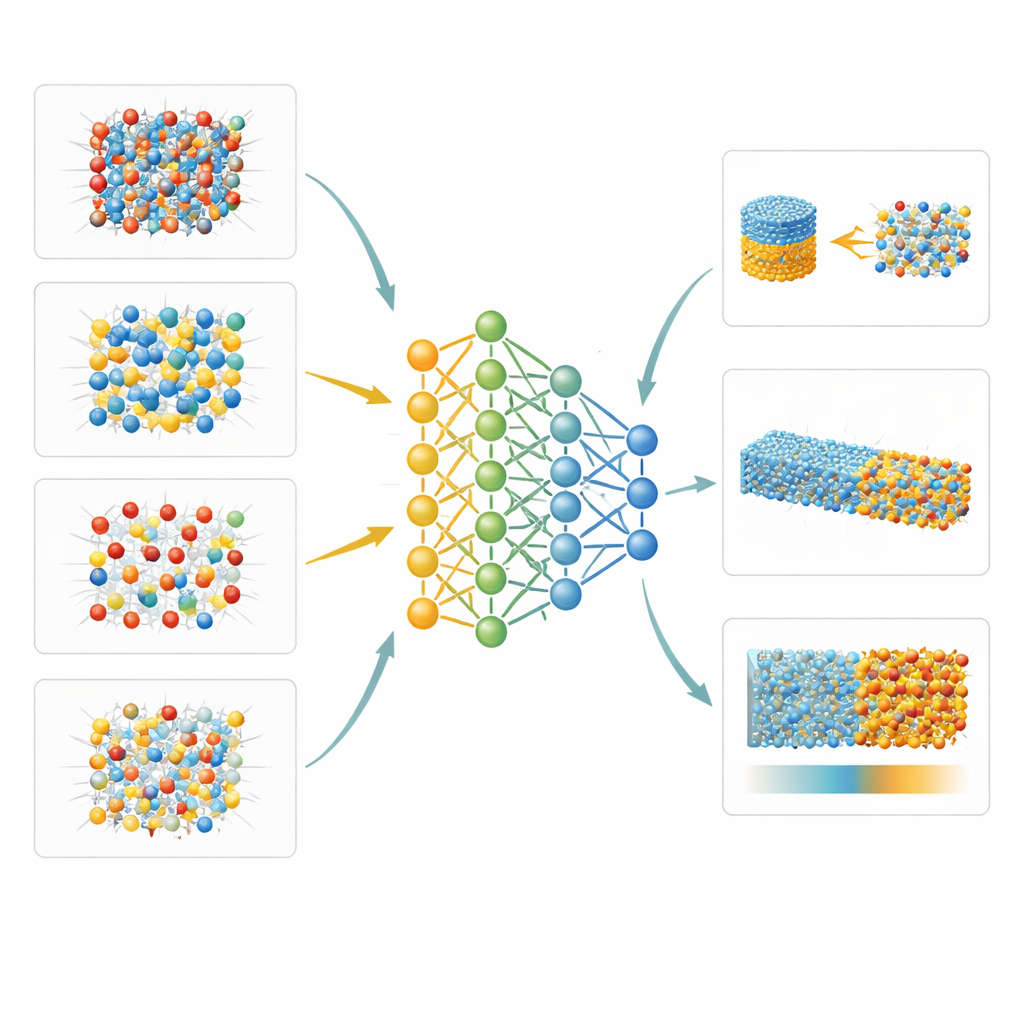
What This Means for Understanding Our Planet
To a non-specialist, the bottom line is that the authors have created a powerful new “digital laboratory” for deep Earth materials. Their machine-learning model can reproduce known mineral boundaries and then go beyond current experiments to estimate subtle properties of melts, interfaces, and atomic disorder under extreme conditions. This opens the door to more realistic simulations of mantle flow, melting, and seismic structures, helping scientists connect the behavior of atoms to the large-scale workings of our planet’s interior.
Citation: Zhong, X., Li, Y. & John, T. A general purposed machine learning interatomic potential for Mg-Al-Si-O system suitable for Earth materials at high pressure and temperature conditions. npj Comput Mater 12, 141 (2026). https://doi.org/10.1038/s41524-026-02056-3
Keywords: Earth mantle materials, machine learning potential, molecular dynamics, phase diagrams, solid–melt interfaces