Clear Sky Science · en
The ISG Atlas: a loss-of-function analysis characterizes antiviral properties of interferon stimulated genes
How Our Cells Wage a Hidden War on Viruses
The human body is constantly bombarded by viruses, yet most infections never take hold. This paper uncovers how hundreds of protective genes inside our cells, switched on by immune molecules called interferons, work together to slow or even encourage different viruses. By mapping this complex network in detail, the researchers reveal new weak spots in viruses such as SARS-CoV-2 and point to fresh ideas for antiviral therapies.
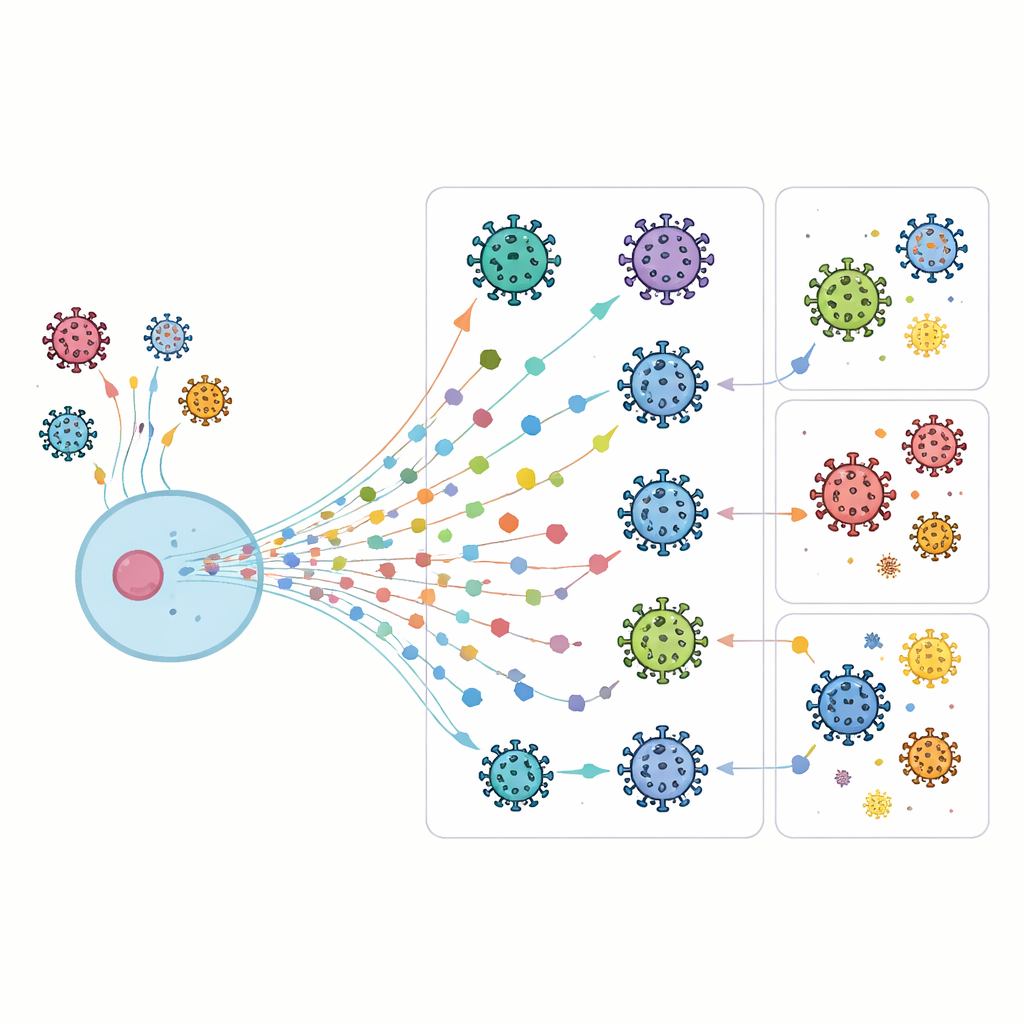
A Cellular Alarm System with Many Moving Parts
When cells sense a viral intruder, they release interferons, which in turn switch on thousands of interferon-stimulated genes (ISGs). These genes influence nearly every aspect of cell biology, from how proteins are made to how damaged material is removed. Some ISGs directly block a virus at specific stages of its life cycle, while others reshape the cell’s internal environment to make it less hospitable to infection. Because viruses have evolved many tricks to dodge these defenses, our cells maintain a large and overlapping toolkit of ISGs, creating a resilient but highly complex defense network.
A Systematic "Atlas" of Antiviral Genes
Instead of artificially boosting single genes one at a time, the team took the opposite approach: they selectively switched off 285 different ISGs in lung-like cells and then watched how eight distinct viruses behaved. Using live-cell microscopy over several days, they measured how quickly each virus spread and how strongly it took over the cell population. By fitting these infection patterns to growth curves, they could estimate both the strength and speed of viral expansion for every gene-virus combination, building a time-resolved atlas of how each ISG shapes infection.
Shared Defenders, Hidden Helpers, and Virus-Specific Effects
The atlas confirmed that well-known players in interferon signaling, such as STAT1, STAT2, IRF9, and the sensor DDX58, broadly restrain many viruses. It also highlighted unexpected defenders that had not been linked to antiviral action before, including proteins involved in protein degradation, RNA processing, and basic cell maintenance. Surprisingly, a substantial fraction of ISGs actually helped viruses replicate in this system. Some genes promoted the growth of nearly all tested viruses, while others showed "split personalities," blocking one virus but aiding another. These mixed roles suggest that many ISGs fine-tune essential cell processes that viruses can either exploit or be hindered by, depending on their strategy.
Zooming In on SARS-CoV-2 and a New Gatekeeper
Because of its global impact, the researchers paid special attention to SARS-CoV-2. By combining their loss-of-function atlas with protein interaction maps and proteomic analysis, they pinpointed a set of ISGs with especially strong influence on coronavirus growth. As expected, genes that control interferon signaling and known entry blockers such as LY6E emerged as key defenders. Among several new candidates, one stood out: BORCS8, a little-studied component of a complex that controls the movement and function of internal vesicles and lysosomes. When BORCS8 was removed, SARS-CoV-2 entered cells more efficiently, produced more infectious particles, and showed similar advantages across multiple virus variants.
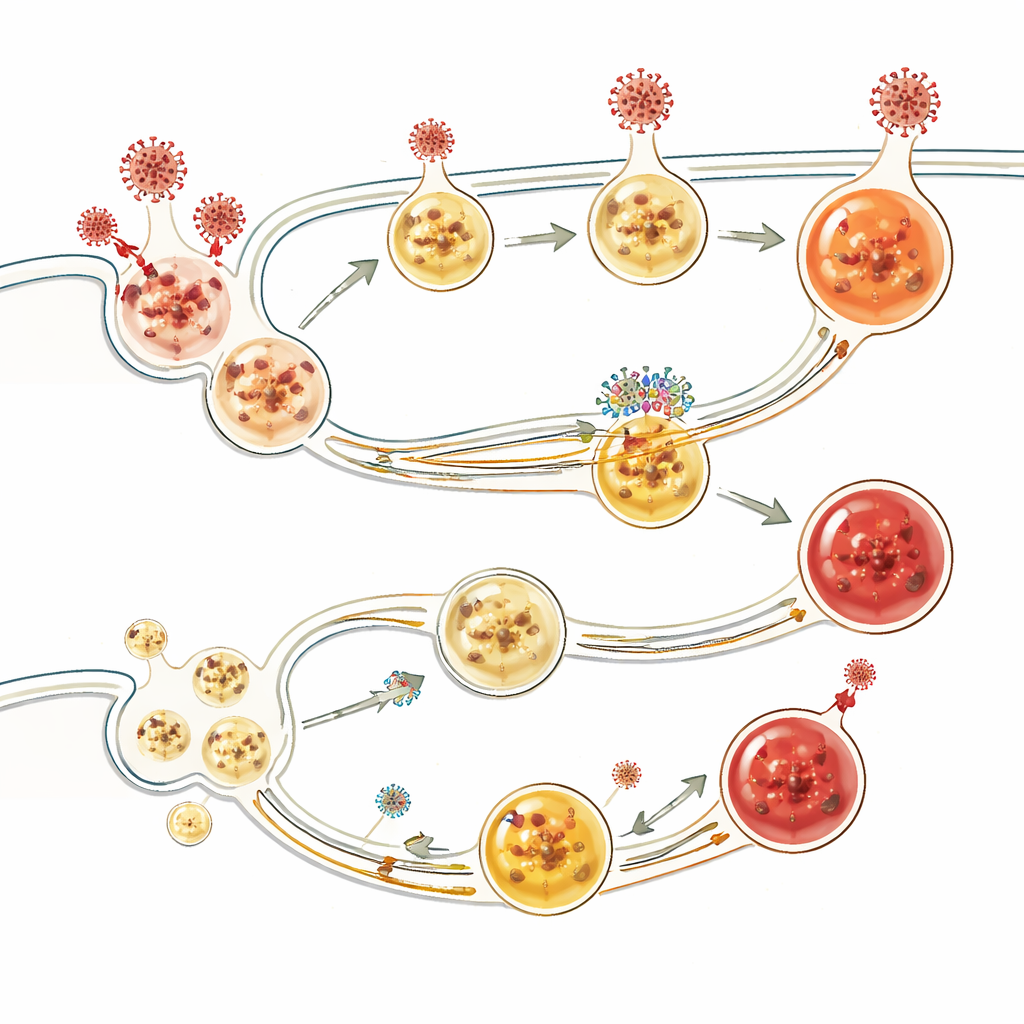
How BORCS8 Shapes Viral Entry Routes
Further experiments revealed that BORCS8 helps coordinate the transport and acidification of endosomes and lysosomes—the small compartments that receive incoming cargo and can destroy it. In cells lacking BORCS8, lysosomes clustered abnormally near the nucleus, endocytic vesicles became overly acidic yet failed to mature properly, and viral cargo was not routed efficiently toward degradation. Viruses that rely heavily on endosomal entry, including SARS-CoV-2 in certain cell types, could therefore slip through more easily. The study also found that BORCS8 works in concert with other vesicle-sorting complexes, and that it affects the localization of SARS-CoV-2’s ORF3a protein, which is involved in viral release from cells.
Working Together for Stronger Antiviral Defense
Because interferon turns on many ISGs at once, the authors tested how pairs of genes behave when disabled together. By comparing observed outcomes to mathematical expectations, they uncovered synergistic combinations where double loss produced a much larger boost in viral growth than either gene alone. Notably, removing both entry blockers (like LY6E or BORCS8) and interferon signaling components (like STAT2 or IRF9) greatly amplified SARS-CoV-2 replication, underscoring how entry control and immune signaling reinforce each other. These findings echo clinical successes where drugs that block viral entry are combined with interferon-based therapies.
What This Means for Future Antiviral Strategies
This work delivers a rich, quantitative map of how hundreds of interferon-triggered genes shape the fate of diverse viruses. For non-specialists, the key message is that our antiviral defense is not driven by a handful of "magic bullets" but by a coordinated community of genes, some of which inadvertently assist the enemy. By revealing broad defenders, virus-specific weak points, and powerful gene combinations, the ISG Atlas offers a blueprint for designing smarter antiviral strategies—particularly those that pair entry blockers like BORCS8-related pathways with boosters of interferon signaling to keep future viral threats in check.
Citation: Krey, K., Risso-Ballester, J., Hamad, S. et al. The ISG Atlas: a loss-of-function analysis characterizes antiviral properties of interferon stimulated genes. Nat Commun 17, 4206 (2026). https://doi.org/10.1038/s41467-026-72732-x
Keywords: innate immunity, interferon-stimulated genes, antiviral defense, SARS-CoV-2 entry, lysosome trafficking