Clear Sky Science · en
Pulsed electrosynthesis orthogonally optimizes C‒N coupling and hydrogenation for amine production with a molecular catalyst
Turning Waste into Useful Ingredients
Carbon dioxide in the air and nitrate in water are usually seen as pollutants, linked to climate change and algal blooms. This study explores how to treat them instead as raw materials, converting them into amines – building-block chemicals used in fertilizers, medicines, and everyday products. By cleverly pulsing electricity through a special molecular catalyst, the researchers show that we can make these useful compounds faster and more selectively, while potentially reducing waste streams.
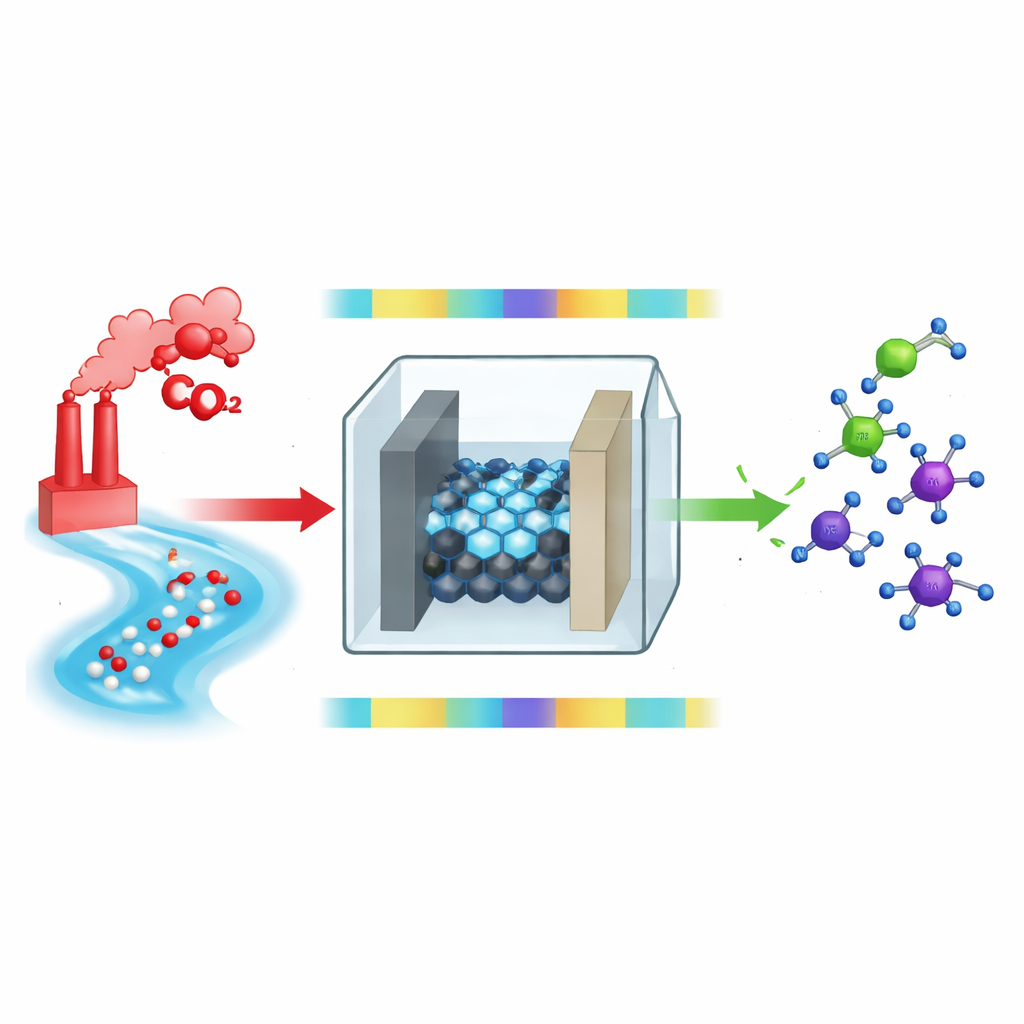
From Pollutants to Valuable Molecules
The core idea is to feed carbon dioxide from the air and nitrate from contaminated water into an electrochemical cell, where electricity drives them to form new bonds between carbon and nitrogen. These bonds create amines, which are central ingredients in many industrial and pharmaceutical processes. In a typical setup, carbon dioxide and nitrate are reduced – that is, they gain electrons and protons – and react to form intermediate compounds such as oximes, which then must be further “hydrogenated” to become amines. While the first bonding step is relatively quick, the later hydrogenation steps are slow and inefficient, creating a bottleneck that limits both yield and energy efficiency.
Why Static Electricity Falls Short
Traditionally, chemists apply a single fixed voltage to the electrodes and hope to choose a compromise that works for all steps of the reaction. But the early and late stages prefer very different conditions. At steady, strongly negative voltages, the surface of the catalyst becomes crowded with hydrogen, which speeds up hydrogenation but also drives unwanted side reactions such as hydrogen gas and ammonia formation. At milder voltages, the crucial carbon–nitrogen couples form more cleanly, yet the intermediates cannot be fully converted into amines. This mismatch makes it hard to design a single, static operating point that gives high selectivity to the desired products.
Pulsing the Current to Split the Job
To solve this, the authors introduce a pulsed strategy: they rapidly alternate between a less negative and a more negative voltage. During the gentle phase, the surface of a cobalt phthalocyanine catalyst supported on carbon nanotubes accumulates carbon–nitrogen intermediates such as formaldoxime, without over-reducing them. Then, during the deeper, brief pulses, the extra driving force speeds up hydrogenation, pushing these intermediates toward methylamine and even higher amines like dimethylamine and trimethylamine. This time-sharing approach lets each reaction step experience its preferred conditions, rather than forcing all of them to happen under the same compromise voltage.
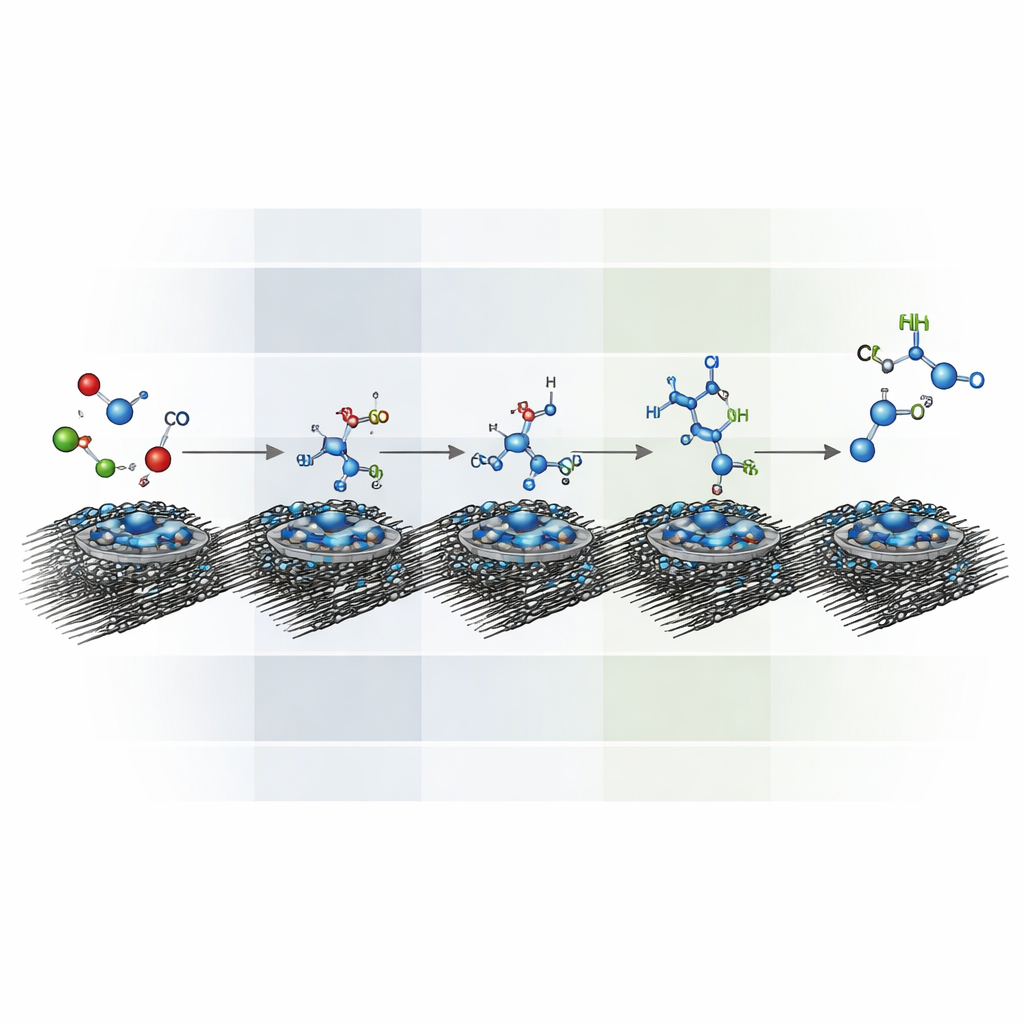
Peering Inside the Reaction Steps
The team used a combination of advanced tools to watch and compute what happens on the catalyst surface. Infrared spectroscopy detected specific vibrational fingerprints of intermediate species, showing how carbon–nitrogen double bonds give way to single bonds as hydrogenation proceeds at more negative pulses. Mass spectrometry monitored gaseous fragments such as carbon monoxide, formaldehyde-like units, and hydroxylamine, revealing how they appear and disappear as the voltage changes. Isotope-labeling experiments, where heavier versions of carbon and nitrogen were used, confirmed that all carbon and nitrogen atoms in the products truly originate from carbon dioxide and nitrate. Computer simulations based on quantum mechanics then mapped out the energy landscape, indicating that deeper pulses help key hydrogenation steps become more favorable while still needing careful control to avoid side reactions.
Building Up to More Complex Amines
By feeding some of the products back as starting materials, the researchers traced how higher amines form. Their tests suggest a stepwise pathway: carbon dioxide and nitrate first produce hydroxylamine and formaldehyde, which join to make formaldoxime; this is then hydrogenated to methylhydroxylamine and onward to methylamine. Methylhydroxylamine and methylamine can then react with more formaldehyde and undergo further hydrogenation to yield dimethylamine and finally trimethylamine. Pulsed operation not only speeds up the reaction but also improves selectivity, roughly tripling the rate and doubling the fraction of current that ends up as methylamine compared with static conditions, while keeping the cobalt-based molecular catalyst largely intact.
What This Means for a Cleaner Future
To a non-specialist, the main message is that carefully timed bursts of electricity can steer complex chemical reactions much like a conductor guides an orchestra. Instead of fighting the natural differences between fast and slow steps, this pulsed approach gives each stage the conditions it needs, turning troublesome pollutants into useful chemicals more efficiently. Although further advances are required before large-scale use, the work outlines a general blueprint: dynamic control of voltage can unlock new pathways for transforming waste carbon and nitrogen into valuable products, bringing greener chemical manufacturing a step closer.
Citation: Yan, S., Wang, Y., Chen, S. et al. Pulsed electrosynthesis orthogonally optimizes C‒N coupling and hydrogenation for amine production with a molecular catalyst. Nat Commun 17, 4027 (2026). https://doi.org/10.1038/s41467-026-72678-0
Keywords: electrochemical amine synthesis, CO2 upcycling, nitrate reduction, pulsed electrolysis, C–N bond formation