Clear Sky Science · en
Hh and EGFR-Ras signaling promote distinct steps of tumor progression in the Drosophila follicle epithelium
How Cells Keep Tissues in Balance
Our bodies, like those of fruit flies, constantly renew their tissues. To stay healthy, stem cells must divide and mature into the right kinds of cells at the right time, while avoiding the runaway behavior that leads to cancer. This study uses the Drosophila ovary as a powerful model to uncover how two major communication systems between cells cooperate to maintain normal growth—and how their malfunction can drive tumor-like overgrowth.
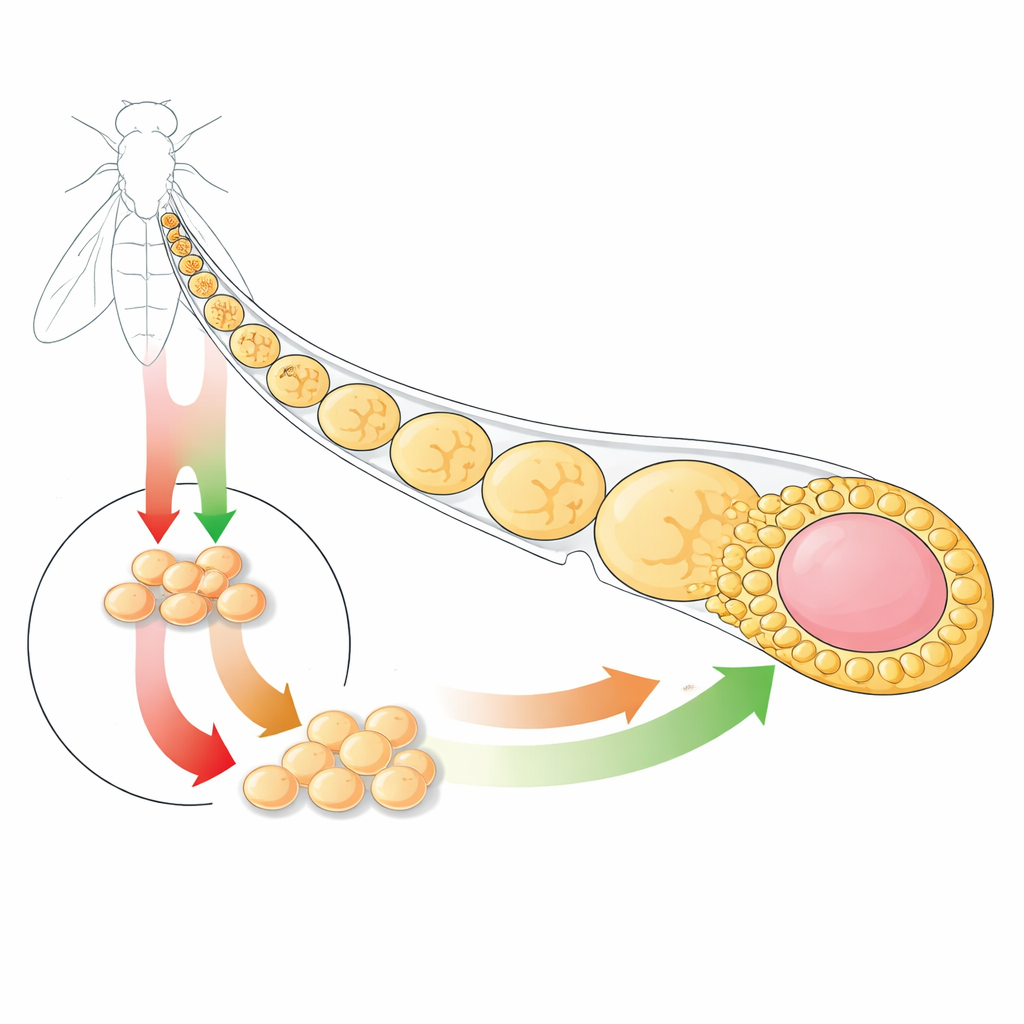
A Tiny Organ with Big Lessons
The researchers focus on the follicle epithelium in the fruit fly ovary, a simple sheet of cells that wraps developing egg chambers. This tissue is maintained by follicle stem cells that sit in a defined niche and produce daughter cells which then specialize into several roles, such as main body cells that cover the egg, stalk cells that link egg chambers, and polar cells that help organize the structure. Because all of these events occur within a very small region and are repeated many times, the system is ideal for studying how signals control both proliferation and differentiation in a living epithelium.
Two Key Messages: Growth and Change
The team examines two major signaling pathways: Hedgehog (Hh) and EGFR-Ras. Both are notorious in human cancer and are active in the fly follicle stem cell region. Using fluorescent reporters and genetic tools, the authors show that in normal conditions these pathways are turned on in overlapping but distinct patterns. Importantly, each pathway controls its own set of target genes and does not simply switch the other on or off. This independence means the cell integrates two separate messages about when to proliferate and when to move toward a mature fate.
When Hedgehog or EGFR-Ras Go Awry
To see what happens when these signals are too strong, the researchers artificially boost either Hh or EGFR-Ras in adult ovaries and then apply single-cell RNA sequencing to tens of thousands of individual cells. Overactive EGFR-Ras mainly pushes cells to keep cycling instead of exiting the usual division program, delaying a key switch to a more relaxed endocycle in main body cells. Overactive Hh has a different effect: it traps many cells in a mixed identity. They retain features of immature cells while also turning on genes typical of stalk cells and markers linked to epithelial–mesenchymal transition (EMT), a process associated with increased motility and cancer spread. In these Hh-boosted ovaries, cells lose their usual polarity, crawl abnormally, and sometimes invade regions they normally do not occupy.
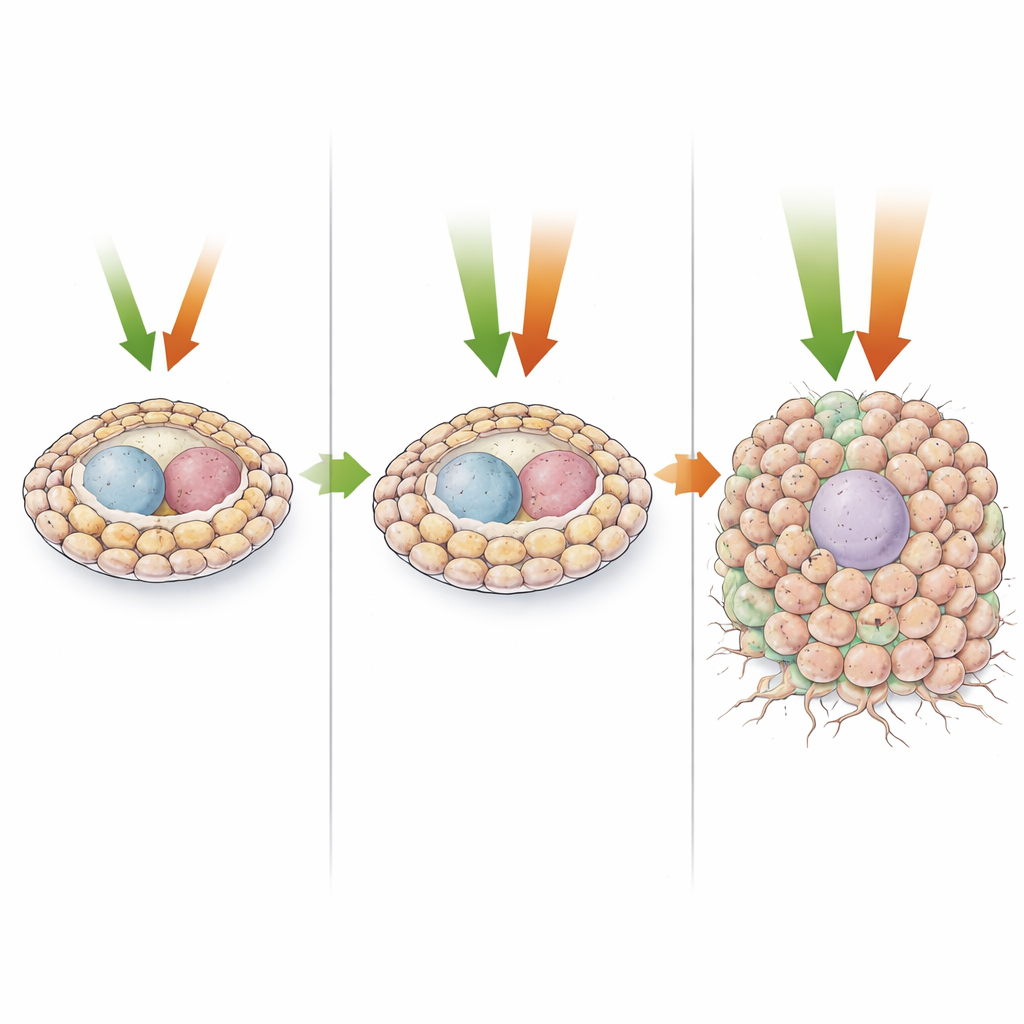
When Both Signals Fire at Once
The most dramatic changes appear when both pathways are overactivated together. Here, stem-cell-like descendants expand massively without maturing, forming large, disorganized cell masses with few remaining germ cells. Single-cell analyses reveal that most of these cells resemble early progenitors rather than fully differentiated types and sit at the very start of developmental trajectories. These overgrown tissues display many hallmarks of malignancy: sustained cell division with shortened resting phases, scrambled tissue architecture, loss of polarity, altered metabolism, and a severe cost to the host's survival. The study also pinpoints two transcription factors, Zfh1 (related to human ZEB1/2) and Pointed (related to ETS1/2), as key downstream players in driving EMT-like behavior and excessive proliferation, respectively.
What This Means for Cancer Biology
In simple terms, this work shows that normal tissue health in the ovary depends on a careful balance between two independent yet converging signal systems. Hedgehog on its own nudges cells toward a partially mobile, stalk-like state, while EGFR-Ras alone keeps them dividing. When both are ectopically turned up, cells become stuck in an undifferentiated, highly proliferative, and invasive state that closely resembles early steps of tumor development. Because the core components of these pathways are conserved from flies to humans, the findings provide a genetically tractable model for understanding how combinations of growth signals and identity switches can cooperate to drive cancer, and why targeting both Hedgehog and EGFR-Ras signaling may be especially effective in treating some epithelial tumors.
Citation: Anschütz, S., Müller, H., Schubert, A. et al. Hh and EGFR-Ras signaling promote distinct steps of tumor progression in the Drosophila follicle epithelium. Nat Commun 17, 2790 (2026). https://doi.org/10.1038/s41467-026-70844-y
Keywords: Drosophila ovary, Hedgehog signaling, EGFR-Ras pathway, epithelial tumor progression, epithelial-mesenchymal transition