Clear Sky Science · en
Concise synthesis of bufogargarizin B by a conformation-controlled skeletal reorganization approach
Why chemists care about toad venom
Toad venoms have long been used in traditional medicine for heart and cancer-related ailments, but many of the active molecules are rare and structurally complex. This paper describes a streamlined way to build one such molecule, bufogargarizin B, in the lab from an inexpensive starting material, opening the door to better studies of its biological effects and to new chemistry for shaping steroid frameworks.
A rare steroid with a twisted backbone
Bufogargarizin B belongs to a family of steroid-like compounds called bufadienolides, found in plants and animals and especially rich in some toads used in Chinese medicine. Unlike typical steroids, which have a regular four-ring “6/6/6/5” backbone, bufogargarizins carry a rearranged “5/7/6/5” ring pattern and a heavily decorated final ring. These unusual shapes are thought to influence how the molecules interact with biological targets, but their scarcity in nature and structural complexity have made them hard to study.
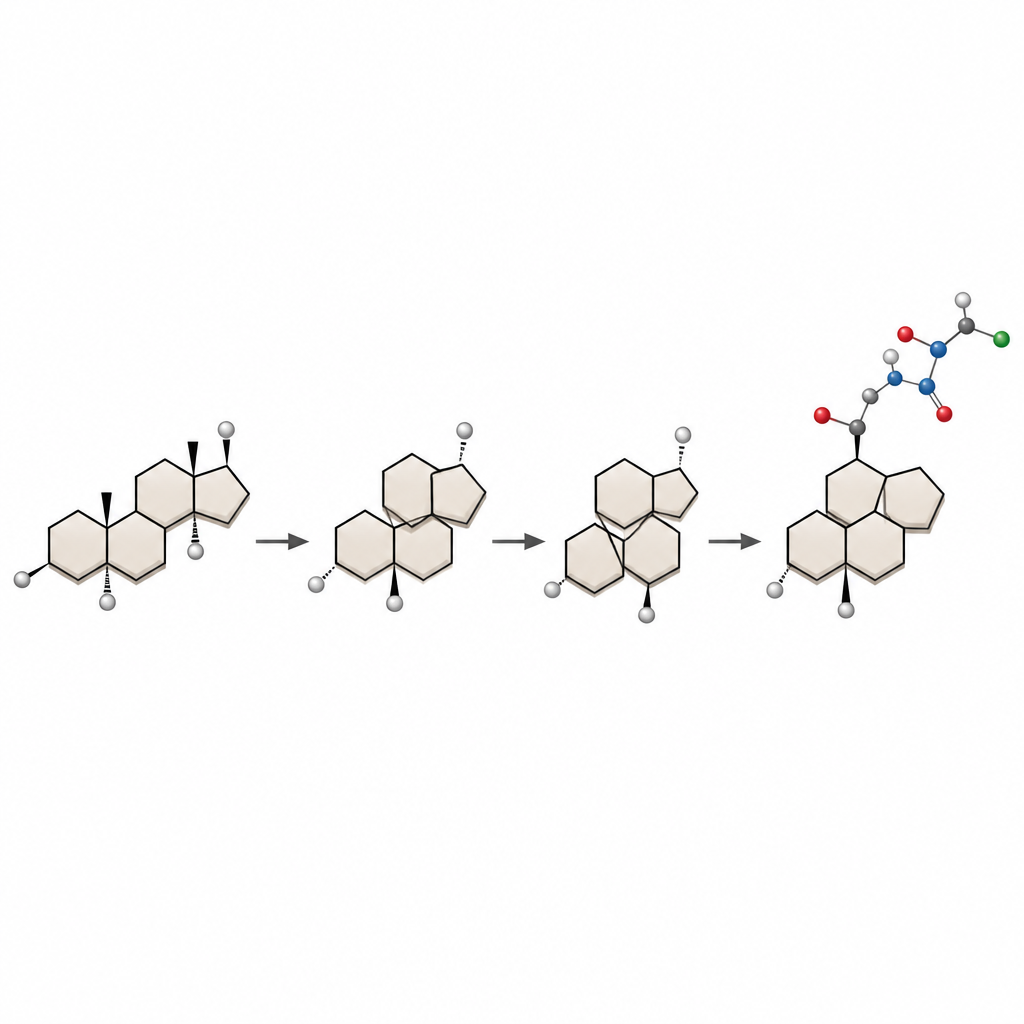
Rebuilding the steroid framework from the inside
The authors start from dehydroepiandrosterone, a cheap and readily available steroid, and redesign its core. They first convert part of the rigid steroid into a flexible ten-membered ring. Then, using a samarium-based reagent, they trigger a transannular reaction, where two distant points across the ring connect to form a new fused system. By carefully controlling how this ring folds in three dimensions and the arrangement of a key carbon–carbon double bond, they steer the reaction to give the desired 5/7/6/5 core of bufogargarizin B with the correct three-dimensional orientation of atoms.
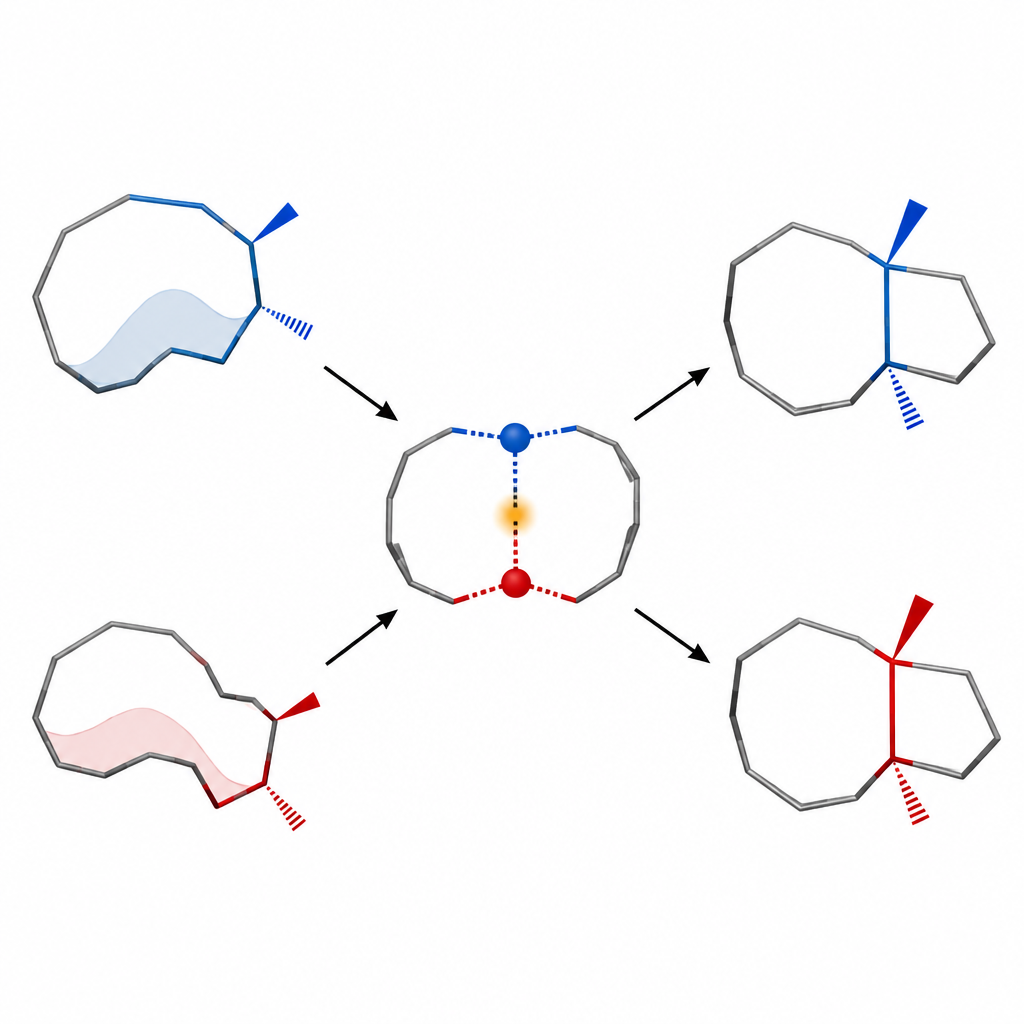
Controlling molecular posture to choose the outcome
A central theme of the work is conformational control: the idea that how a molecule bends and twists can dictate what product a reaction gives. The team shows that their ten-membered ring can adopt different shapes, and that each shape prefers to lead to a different fused-ring outcome. By tuning conditions such as temperature and substituents, they enrich the conformation that delivers the correct pair of neighboring stereocenters in the final framework. This approach contrasts with more traditional reactions that often yield messy mixtures of stereoisomers when used on such medium-sized rings.
Building the highly decorated side ring
Once the core framework is in place, the researchers still need to install the densely functionalized final ring that carries an α-pyrone side segment characteristic of bufadienolides. They develop a practical, gram-scale synthesis of a small building block called 2-pyrone-5-boronate from the simple chemical furfuryl alcohol. Using this fragment in a cross-coupling reaction, followed by a sequence of carefully chosen oxidation and reduction steps, they construct the all-cis “D” ring of bufogargarizin B, including its multiple oxygen-containing groups, without disturbing the rest of the molecule.
Switching between related ring systems
The authors also explore a biomimetic reaction that interconverts different fused-ring layouts similar to what may happen in nature. They design intermediates that can undergo a retro-aldol step, breaking a carbon–carbon bond, followed by a transannular aldol reaction that re-forms a different connection across the ring. This cascade allows access to both 5/7/6/5 and 7/5/6/5 core skeletons and supports the proposed relationship between bufogargarizin B and its close relative bufogargarizin A. Along the way they map which stereochemical arrangements are favored, highlighting again how molecular conformation guides which structures are thermodynamically preferred.
What this means for future molecules
In the end, the team achieves an 18-step synthesis of bufogargarizin B from a common steroid and outlines a route to the core of bufogargarizin A. For a non-specialist, the key message is that they have learned to “pose” a flexible ring so that a single, well-defined product forms, rather than a confusing mixture. This strategy of conformation-controlled skeletal reorganization, combined with a practical route to the 2-pyrone building block, should be useful for making other rare toad-derived and plant-derived bufadienolides, enabling more rigorous studies of their biological roles and potential therapeutic value.
Citation: Yang, P., Shen, Y. & Gui, J. Concise synthesis of bufogargarizin B by a conformation-controlled skeletal reorganization approach. Nat Commun 17, 4156 (2026). https://doi.org/10.1038/s41467-026-70735-2
Keywords: bufogargarizin B, steroid synthesis, conformational control, bufadienolides, transannular cyclization