Clear Sky Science · en
CD177⁺ neutrophil-platelet aggregates contribute to thromboinflammation via NETs in necrotizing enterocolitis
Why tiny babies face a big gut problem
Necrotizing enterocolitis is a sudden and often deadly disease of the intestine that strikes very premature babies. Doctors know that these infants develop fierce gut inflammation and sometimes dangerous blood clotting, but how these two processes connect has been a mystery. This study uncovers a hidden chain reaction in the blood vessels of the infant intestine that helps explain why the disease can worsen so quickly and suggests new ways to diagnose and possibly prevent it. 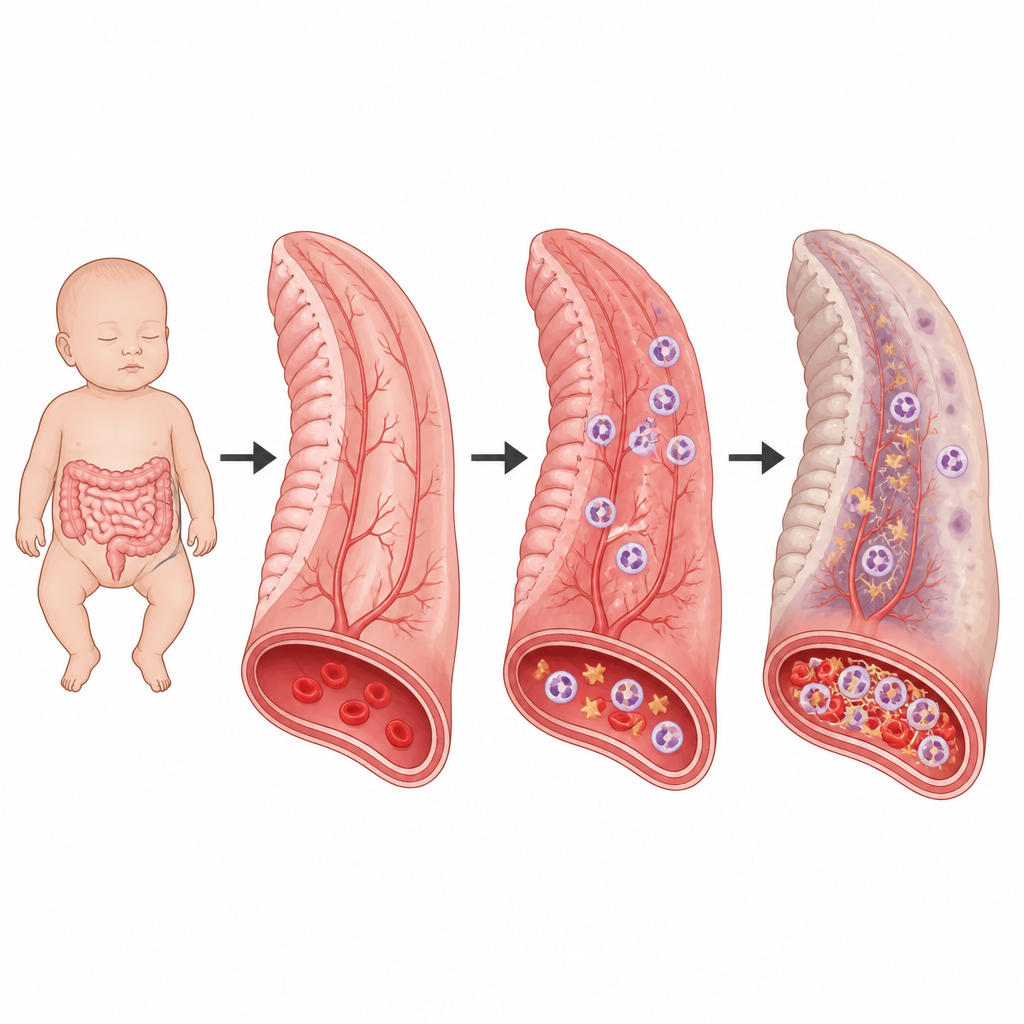
When defense and clotting team up
The body’s first responders to infection and injury are white blood cells called neutrophils and tiny cell fragments called platelets that help blood clot. Normally, they work together to trap germs in small clots and keep them from spreading. In necrotizing enterocolitis, however, this teamwork becomes overzealous. The authors analyzed blood tests and tissue from affected infants and found strong signs of both inflammation and clotting problems, including high clotting markers, low platelets, and many neutrophils. Under the microscope, small intestinal vessels were often plugged with clots rich in neutrophils, platelets, and fibrin, the protein strands that form a blood clot.
A special neutrophil subset takes center stage
Using single-cell gene reading technology, the researchers discovered that not all neutrophils behave the same way. In babies with necrotizing enterocolitis, a subgroup carrying a surface marker called CD177 was greatly expanded in both blood and gut tissue. These CD177-positive neutrophils were primed to stick to vessel walls, interact with platelets, and drive clotting pathways. In mouse models of the disease, similar CD177-marked neutrophils were enriched and showed gene activity linked to cell movement and clot tightening, suggesting that this subset is particularly good at building and remodeling clots inside tiny intestinal vessels.
Sticky cell clusters and lethal molecular webs
The team then looked at how these specialized neutrophils behave together with platelets. They found that CD177-positive neutrophils frequently bound to platelets to form mixed cell clusters called neutrophil–platelet aggregates. These clusters were far more common in sick infants and in diseased mouse intestines and became more abundant as the disease grew more severe. Inside these clusters, neutrophils released webs of DNA and proteins known as neutrophil extracellular traps, or NETs. While NETs can help capture microbes, in this setting they intertwined with platelets and fibrin inside vessels, thickening clots, blocking blood flow, and worsening oxygen starvation in the gut wall. 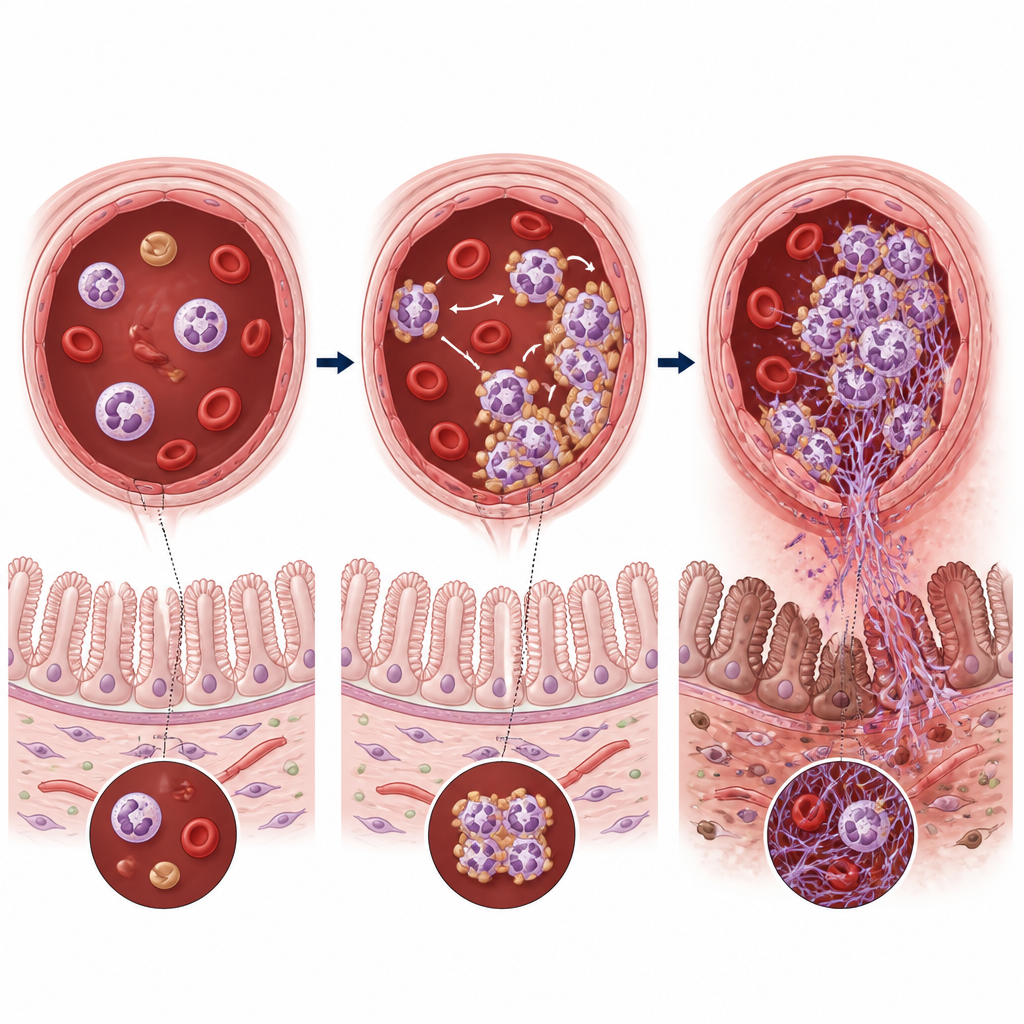
Turning down the harmful cycle
To test whether these processes actually drive damage rather than just accompany it, the researchers deliberately altered them in mice. Removing neutrophils or specifically eliminating CD177-positive neutrophils reduced the number of aggregates, lowered NET formation, eased intestinal injury, and improved survival. Drugs that directly break down NETs or block their formation had similar protective effects. The blood thinner low-molecular-weight heparin, already used in many hospitals, also disrupted neutrophil–platelet aggregates, reduced NET burden, calmed inflammation, and helped preserve gut tissue when given preventively in animal models.
What this could mean for fragile newborns
In a small group of infants, levels of CD177-positive neutrophils, their aggregates with platelets, and NET-related markers rose at diagnosis and fell as the babies improved, suggesting that these measurements could help doctors track disease activity in real time. Overall, the study paints necrotizing enterocolitis not just as an intestinal inflammation, but as a disorder where immune cells and clotting spiral together into a damaging loop inside the gut’s microvessels. Breaking this loop by targeting CD177-positive neutrophils, their sticky aggregates, or the NET webs they spin may one day offer safer ways to predict, prevent, or lessen this devastating disease in the most vulnerable newborns.
Citation: Lan, C., Tian, B., Shi, Y. et al. CD177⁺ neutrophil-platelet aggregates contribute to thromboinflammation via NETs in necrotizing enterocolitis. Nat Commun 17, 4167 (2026). https://doi.org/10.1038/s41467-026-70717-4
Keywords: necrotizing enterocolitis, neutrophil platelet aggregates, neutrophil extracellular traps, thromboinflammation, low molecular weight heparin