Clear Sky Science · en
MDM2 Inhibition with Alrizomadlin (APG-115) in TP53 wild-type salivary gland cancers: a phase I clinical trial
New hope for a rare head and neck cancer
Salivary gland cancers are uncommon tumors that often strike without warning and can return or spread years after initial treatment. For people with advanced disease, there are few proven drug options and most therapies focus on easing symptoms rather than controlling the cancer long term. This study tests a pill called alrizomadlin that aims to switch back on one of the body’s natural tumor-fighting safeguards, offering a potential new avenue for patients who currently have limited choices.
Why these cancers are so hard to treat
Malignant salivary gland cancers account for only a small fraction of head and neck tumors, but they are remarkably diverse. The most common subtype in this trial was adenoid cystic carcinoma, a slow-burning cancer that can stay quiet for years before suddenly accelerating. Once the disease has spread or come back in places where surgery and radiation are no longer possible, doctors turn to systemic drugs. Traditional chemotherapies and newer blood vessel–targeting pills have produced modest tumor shrinkage and often cause substantial side effects, and no drug is formally approved for these patients. Researchers therefore look closely not just at whether tumors shrink, but also at how long growth can be held in check.
Turning a natural guardian back on
Many solid tumors carry damage in a key gene called TP53, which encodes a protein often described as a guardian of the genome. Salivary gland cancers are unusual because TP53 is usually intact, but another protein called MDM2 can overact and keep this guardian switched off. Alrizomadlin is an oral drug designed to block MDM2 so that the body’s own defenses can once again slow or stop cancer cell growth. Earlier work in mice with patient-derived adenoid cystic carcinoma tumors suggested that this strategy could shrink tumors, especially when combined with platinum chemotherapy, leading researchers to test the approach in people.
How the trial was carried out
This phase I study enrolled adults with salivary gland cancers whose tumors were confirmed to have normal TP53 and had clearly grown by at least 20 percent over the previous year. Forty patients received at least one dose of study treatment: 36 took alrizomadlin alone and 4 took alrizomadlin plus the chemotherapy drug carboplatin. A flexible dosing design helped the team identify a dose that balanced safety and potential benefit. The main early goal was to find serious side effects that would limit dosing, while also tracking tumor responses, time until tumor growth resumed, and survival.
Safety signals and side effects
Alrizomadlin on its own was generally tolerable. Among 37 patients treated with the pill, three experienced serious early side effects that met the study’s strict definition of dose-limiting, including short-lived drops in white blood cells and dizziness. Most patients had some treatment-related fatigue, nausea, or blood count changes, and about two thirds had at least one severe (grade 3 or higher) side effect, but only a small fraction stopped the drug because of toxicity. In contrast, when alrizomadlin was paired with carboplatin in four patients, every patient had severe side effects and half experienced the most extreme blood count drops, leading the team to halt the combination arm.
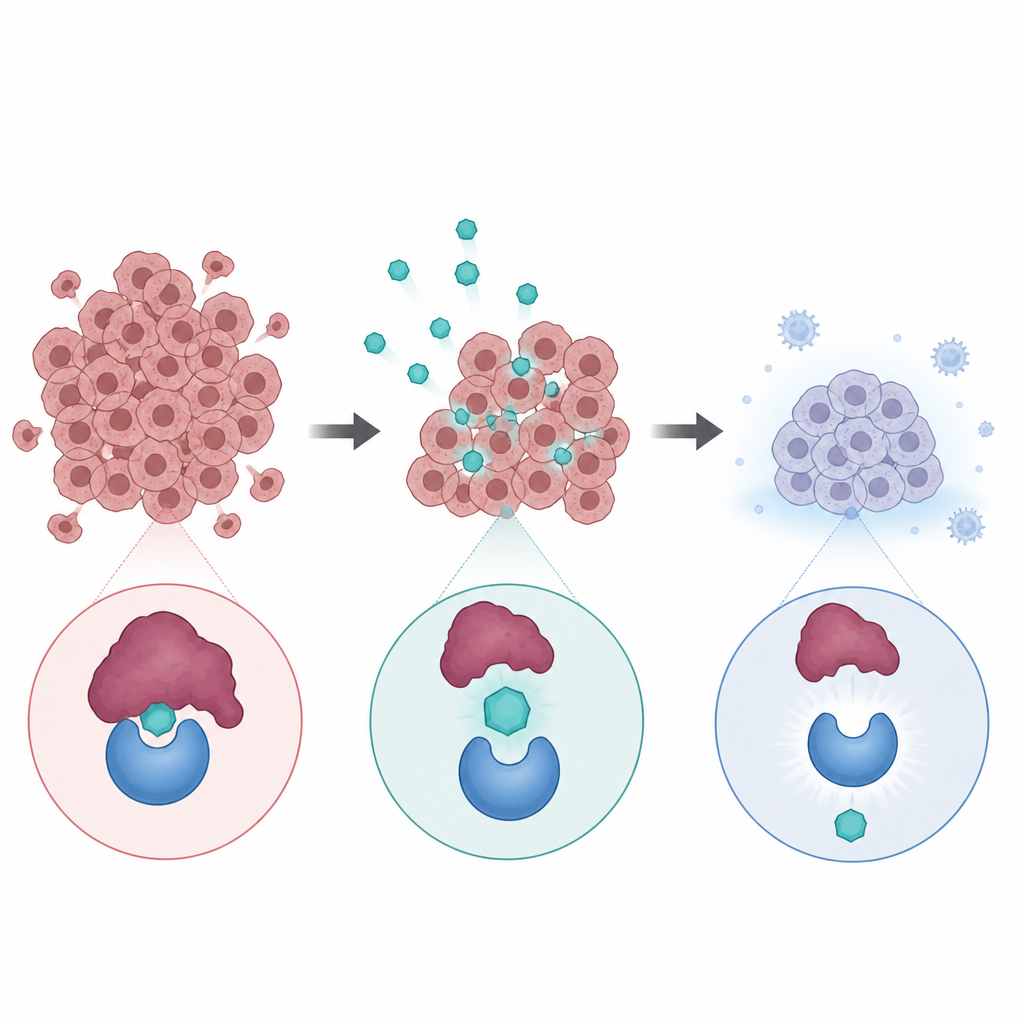
What the drug did to the tumors
Among 34 patients who took alrizomadlin alone long enough to measure a response, 15 percent had clear tumor shrinkage and nearly 80 percent had stable disease, for a disease control rate of over 94 percent. Tumors got smaller in about three quarters of patients at some point during follow up. The typical time before the cancer started growing again was around nine to ten months, and several patients stayed on treatment for more than a year. People with adenoid cystic carcinoma, who made up most of the participants, appeared to fare particularly well, with a similar response rate but slightly longer median time without progression compared to other salivary gland cancer types.
Clues from tumor DNA
The researchers also examined stored tumor samples from a subset of patients. They confirmed that none of these tumors carried TP53 mutations, aligning with the trial’s design. Overall, the cancers showed relatively few genetic changes, and there were no alterations in MDM2 or its close partner MDM4. Staining tests for other markers, such as MYB and ALDH1A1, did not clearly separate patients who benefited from those who did not, suggesting that more work is needed to find simple lab tests that can predict who will respond best to alrizomadlin.
What this means for patients
For people with advanced salivary gland cancers, especially adenoid cystic carcinoma, this early trial suggests that alrizomadlin as a single pill can keep tumors in check for many months with manageable side effects. The drug did not work for everyone and combining it with standard chemotherapy proved too harsh at the doses tested, but the results strengthen the idea that reawakening the body’s own tumor-protective pathways can be clinically useful. Larger, carefully controlled studies will be needed to confirm whether alrizomadlin meaningfully extends life or quality of life, yet this work marks a concrete step toward more targeted options for a rare group of patients who have long faced limited treatment choices.
Citation: Pearson, A.T., Muzaffar, J., Kirtane, K. et al. MDM2 Inhibition with Alrizomadlin (APG-115) in TP53 wild-type salivary gland cancers: a phase I clinical trial. Nat Commun 17, 4240 (2026). https://doi.org/10.1038/s41467-026-70653-3
Keywords: salivary gland cancer, adenoid cystic carcinoma, MDM2 inhibitor, phase I trial, targeted therapy