Clear Sky Science · en
The nuclease EXO1 promotes genomic instability by degrading nascent DNA in BRCA-proficient cells
When DNA Repair Turns Risky
Our cells constantly copy and repair their DNA, using a fleet of enzymes that usually act as guardians against cancer. This study reveals that one of those guardians, an enzyme called EXO1, can become a troublemaker when present in excess. In many tumors, EXO1 is switched on too strongly, and instead of quietly helping with repairs, it chews into newly made DNA strands, leaving behind dangerous breaks that can fuel cancer development—but that may also make such tumors more vulnerable to certain drugs. 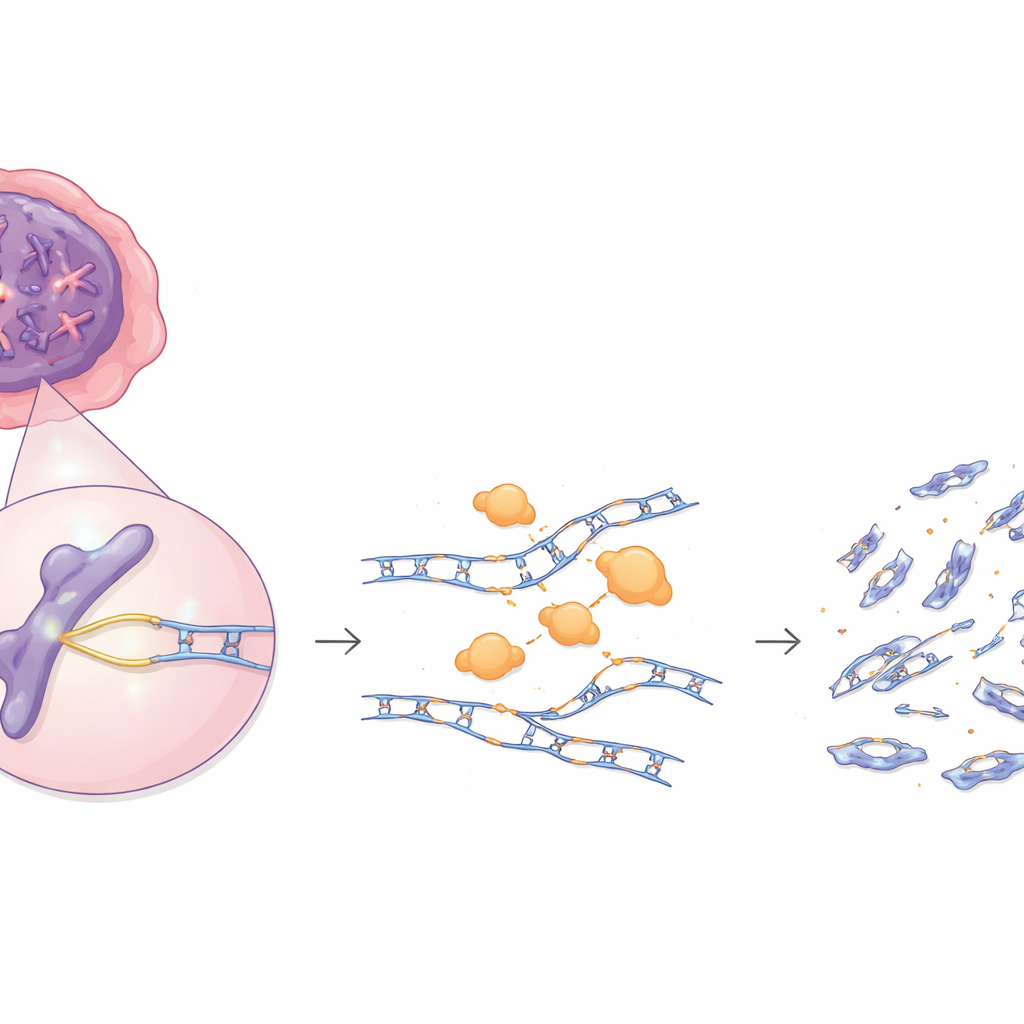
Keeping DNA Copying on Track
Every time a cell divides, it must duplicate its DNA. This process, known as replication, is carried out by a moving molecular machine that travels along the double helix, creating two new daughter strands. In cancer cells, this copying process is often stressed: building blocks for DNA may run low, or the machinery may hit chemical roadblocks caused by damage or chemotherapy. Normally, cells use backup strategies to cope—pausing and restarting the copying machine or skipping over obstacles and filling the gaps later—while repair systems patch up any vulnerable stretches of single-stranded DNA so they do not turn into full-blown breaks.
A Helpful Enzyme in Overdrive
EXO1 is one of the cleanup enzymes that trims DNA during repair. Under usual conditions, it nibbles back DNA ends so other proteins can accurately restore broken strands. The authors first looked at large cancer datasets and found that, in several tumor types—including breast, liver, skin, testicular, and cervical cancers—EXO1 is frequently present at unusually high levels. Instead of being lost, as many tumor-suppressing repair genes are, EXO1 is amplified or overexpressed. Tumors with more EXO1 tend to show more large-scale DNA alterations such as chromosome gains, losses, and rearrangements, a pattern that hints at increased genomic instability.
How Excess EXO1 Damages New DNA
To see what EXO1 overabundance actually does inside cells, the researchers forced cancer cell lines that still have an intact BRCA repair pathway to make extra EXO1. When they then put these cells under replication stress using drugs that slow DNA copying or create chemical adducts on DNA, they saw more DNA lesions specifically in recently replicated regions. Detailed single-molecule assays showed that extra EXO1 causes gaps in the freshly made strands and also gnaws back DNA at replication forks that have folded into a four-way “reversed” shape during stalling. Crucially, this destructive behavior required EXO1’s cutting activity; a version of the enzyme that cannot cut DNA did not produce these effects. 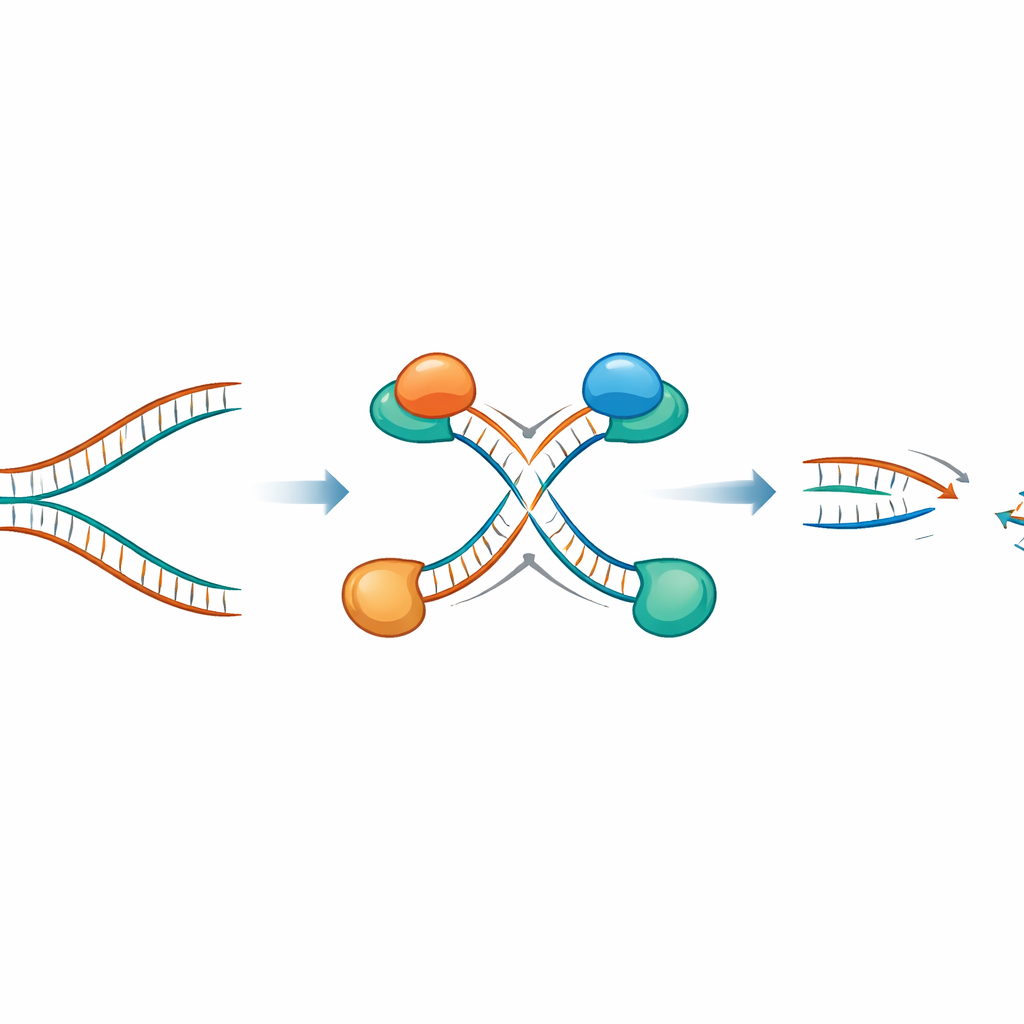
A Destructive Partnership and Broken Chromosomes
The team then probed how EXO1 cooperates with another nuclease, MRE11. Using microscopic proximity assays, they found that excess EXO1 draws MRE11 onto newly made DNA at both gaps and reversed forks, forming paired cutting machines that attack the two strands from opposite directions. Even though the cells still possessed working BRCA proteins, which normally protect stalled forks, the combined action of EXO1 and MRE11 overwhelmed this safeguard. The result was widespread degradation of nascent DNA, conversion of gaps and stalled forks into double-strand breaks, and a marked increase in structural damage to chromosomes.
From Genomic Chaos to Drug Sensitivity
Although this damage promotes the kind of genomic chaos that can drive cancer evolution, it also creates an Achilles’ heel. Cells overloaded with EXO1 were hypersensitive to cisplatin, a chemotherapy drug that creates bulky lesions in DNA, and to PARP inhibitors, drugs that already work well against tumors lacking BRCA function. In fact, the drug responses of EXO1-rich cells resembled those of cells with direct BRCA defects, a state often referred to as “BRCAness.” To a layperson, this means that even without mutations in the famous BRCA genes, tumors that crank up EXO1 behave as if they were BRCA-deficient: their DNA is fragile and easily pushed over the edge by treatments that stress replication, suggesting EXO1 levels might serve as a biomarker to guide therapy choices.
Citation: Nusawardhana, A., Nicolae, C.M. & Moldovan, GL. The nuclease EXO1 promotes genomic instability by degrading nascent DNA in BRCA-proficient cells. Nat Commun 17, 3169 (2026). https://doi.org/10.1038/s41467-026-69981-1
Keywords: DNA repair, genomic instability, BRCA pathway, cancer chemotherapy, EXO1 nuclease