Clear Sky Science · en
Using electronic health records to assess the relationship between colonization pressure and nosocomial pathogen acquisition
Why germs from other patients matter
Anyone who has stayed in a hospital has probably wondered: can I catch something from the person in the next bed? This study tackles that question using the digital footprints of care left in electronic health records. The authors ask whether the number of people on a ward recently carrying dangerous bacteria and other microbes influences how likely a new patient is to pick up those same germs, even before they cause full-blown infection.
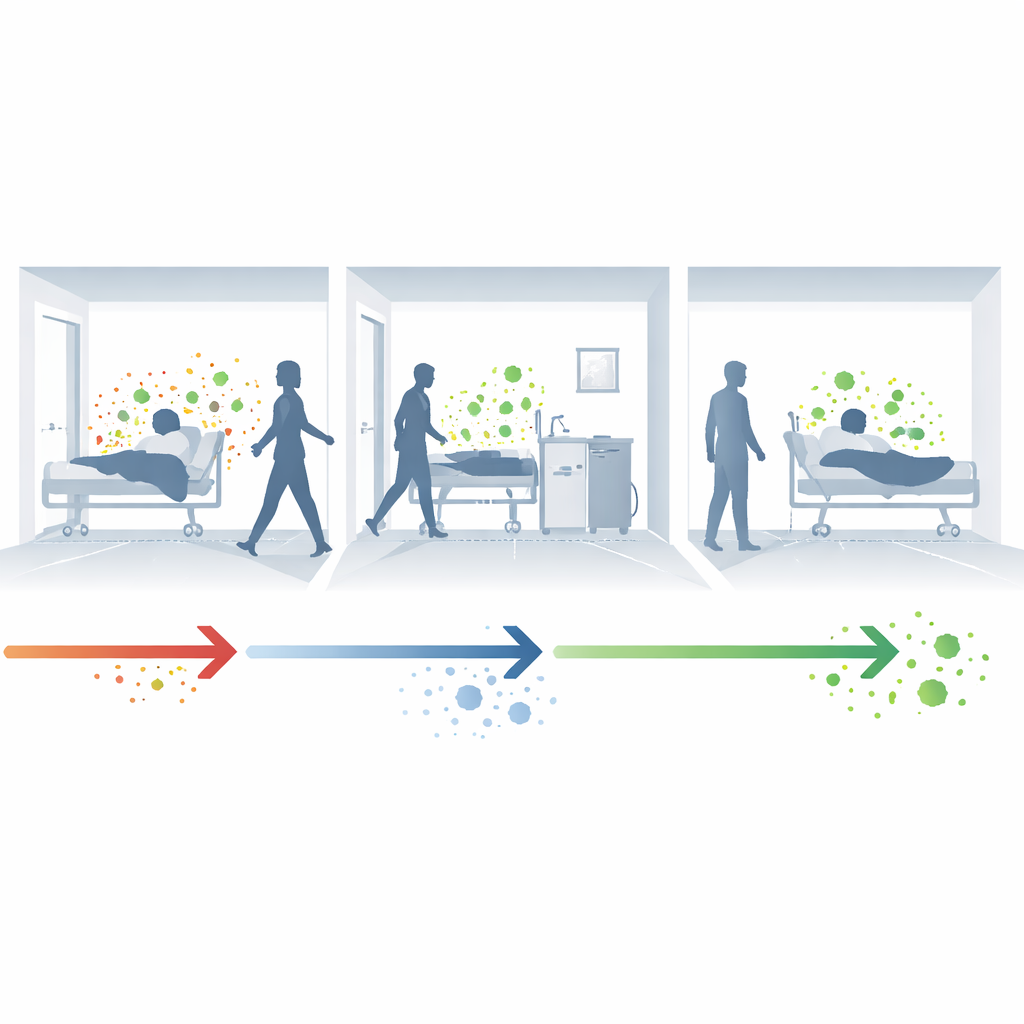
How germs quietly move around hospitals
Hospital-acquired infections often start with a hidden step: colonization. A patient carrying a microbe, such as resistant bacteria in the gut or on the skin, may not feel sick but constantly sheds those organisms into the room and onto equipment. When staff move between patients, or when surfaces are not perfectly cleaned, these organisms can reach other vulnerable patients. If the new host is weakened or exposed to antibiotics, the colonizing microbe can tip over into a serious infection, completing a cycle of person-to-person and room-to-person spread.
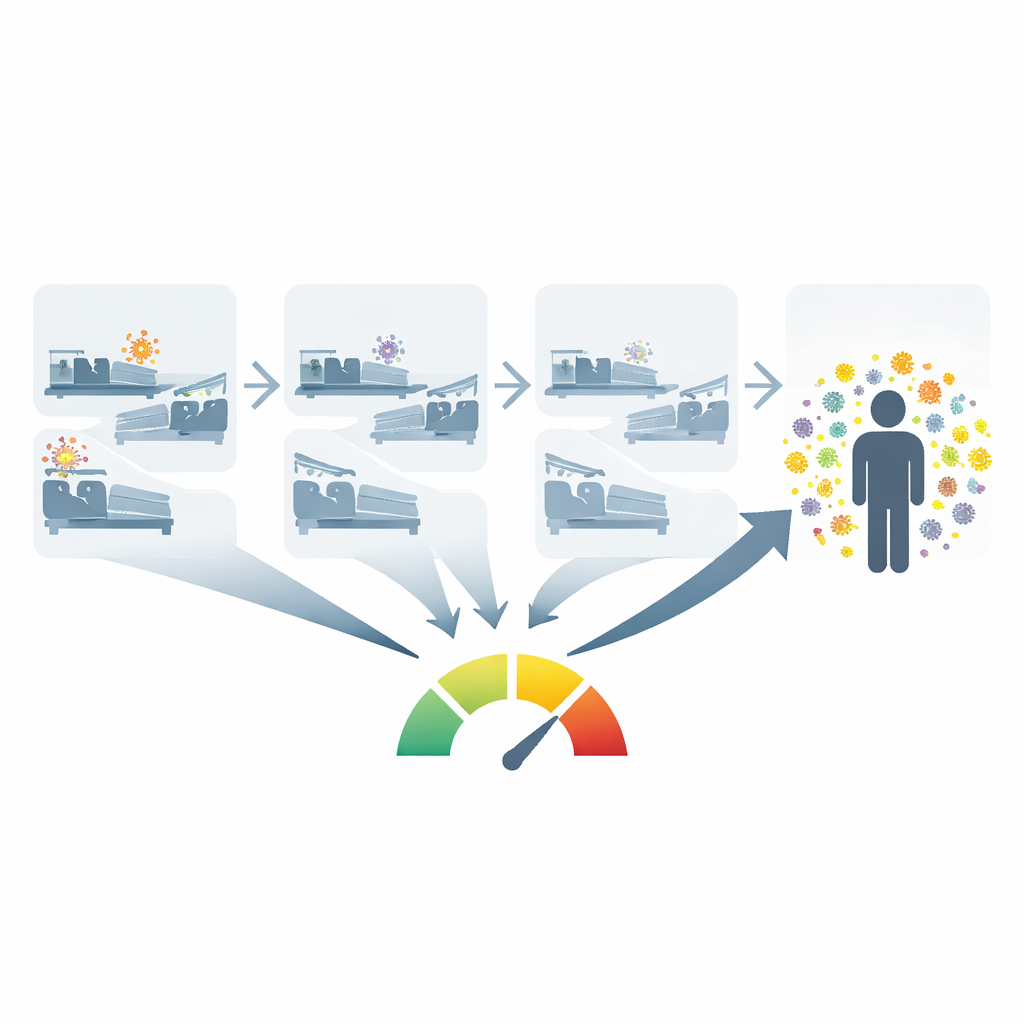
Turning hospital data into a measure of crowding by germs
Directly swabbing every patient to see who is carrying which microbe is expensive and usually limited to a few high-risk organisms in intensive care units. Instead, the researchers created an open-source tool that uses existing electronic health record data to estimate something they call colonization pressure: essentially, how many people sharing a ward recently had a given pathogen, and how recently it was detected. For each new patient, the tool looks back over the previous months at other patients who stayed on that ward, checks their lab results, and gives more weight to microbes identified in the recent past than long ago. This yields a single number for each organism that summarizes how saturated the ward has been with that germ.
What the large health system study found
Using data from more than 43,000 adult inpatients across ten hospitals in one health system over nine years, the team compared patients who newly acquired a microbe during their stay with carefully matched controls who did not. They studied 11 different organisms, including both drug-susceptible and drug-resistant strains that live in the gut, on the skin, or in the wider environment. Across this wide range, the pattern was striking: when the colonization pressure for a particular organism was higher, patients were more likely to acquire that same organism. This held true for classic hospital threats such as Clostridioides difficile, methicillin-resistant Staphylococcus aureus, and resistant Pseudomonas, as well as for common, usually more treatable strains.
Unexpected push and pull between different microbes
The analysis also revealed more complex ecological effects. In many cases, high colonization pressure from one organism was linked to a lower chance of picking up another. For example, wards with more drug-resistant Pseudomonas seemed to have fewer new cases of a particular gut bacterium. These inverse patterns were most common when the microbes tended to live in different body sites, hinting that competition between species—or differences in room layout, cleaning practices, or patient types—might shape which germs take hold. The authors emphasize that their strict matching of patients on age, underlying illness, and antibiotic exposure reduces, but cannot fully remove, such confounding.
Promise and limits of prediction
Because colonization pressure is simple to compute from routine records, the team tested whether it could power a practical risk-prediction system. They trained modern machine-learning models that used colonization pressure and a summary of patients’ chronic conditions to predict who would acquire each organism. Although the statistical association between colonization pressure and acquisition was clear, the models’ ability to correctly flag future cases was modest. This suggests that colonization pressure is just one of many ingredients in infection risk, alongside factors such as devices, procedures, and individual immune defenses that were not fully captured here.
What this means for patients and hospitals
For lay readers, the key message is that germs do not just follow individual patients—they also accumulate in wards and rooms, shaping the invisible "microbial weather" that every new patient walks into. This study shows that the recent history of a unit, in terms of which organisms have been present there, measurably changes the odds that newcomers will pick up those same microbes. It also highlights that ordinary, drug-susceptible bacteria, which are often not tracked closely, account for far more hospital-acquired cases than the highly resistant strains that dominate headlines. By releasing both their software and de-identified data, the authors hope other hospitals will refine these methods and eventually build real-time warning systems that signal when a ward’s colonization pressure is climbing, prompting extra cleaning, protective measures, or closer monitoring to keep patients safer.
Citation: Sagers, L., Wei, Z., McKenna, C. et al. Using electronic health records to assess the relationship between colonization pressure and nosocomial pathogen acquisition. Nat Commun 17, 3134 (2026). https://doi.org/10.1038/s41467-026-69873-4
Keywords: hospital-acquired infection, colonization pressure, electronic health records, antibiotic-resistant bacteria, infection control