Clear Sky Science · en
Brain-derived neurotrophic factor and the derived dodecapeptide function as Toll-like receptor 4 antagonists in acute lung injury
Why calming lung inflammation matters
Acute lung injury and its severe form, acute respiratory distress syndrome, can turn a routine infection or trauma into a life‑threatening crisis within hours. In these conditions, the lungs fill with fluid and inflammatory cells, making it hard or impossible to breathe. Current treatments mainly support breathing but do little to tame the underlying storm of inflammation. This study uncovers a surprising natural brake inside the lungs themselves—a nerve‑related protein and a tiny fragment of it—that can dial down runaway immune reactions and may inspire new medicines for both lung failure and sepsis.
A nerve helper with an unexpected lung role
Brain‑derived neurotrophic factor (BDNF) is best known as a molecule that helps nerve cells grow, survive, and form new connections. The authors asked whether BDNF and related factors also matter in sudden inflammatory diseases outside the brain. Using multiple mouse models of acute lung injury and sepsis, as well as public human and animal datasets, they examined nine members of this family. Among them, BDNF stood out: its gene activity dropped consistently in injured lungs, and protein levels fell specifically in the fluid that bathes the air sacs, but not in the bloodstream. Single‑cell analyses showed that lung epithelial cells—the cells lining the air sacs where gas exchange occurs—are the main local source of BDNF and that their BDNF production shrinks during severe inflammation.
More BDNF, less damage
To test whether this decline in BDNF simply accompanies disease or actually influences it, the team boosted BDNF in different ways. They engineered mice so that lung epithelial cells produced extra BDNF, and they also treated normal mice with injected recombinant BDNF protein. In models where bacterial toxins triggered acute lung injury, added BDNF reduced fluid buildup, leakage of proteins into the air spaces, and thickening of the delicate alveolar walls. Fewer immune cells, particularly neutrophils and macrophages, flooded into the lungs, and animals were more likely to survive experimental sepsis. Measurements of inflammatory messengers such as TNF‑α and IL‑6 fell in both lung tissue and lung fluid, linking higher BDNF to a quieter immune response.
Macrophages and a key danger sensor
The investigators next asked which cells BDNF acts on. They found that when macrophages—the immune cells that first sense invading bacteria in the lungs—were removed from mice, BDNF treatment no longer protected against lung injury or sepsis. In cultured macrophages, BDNF sharply reduced the activity of dozens of inflammation‑related genes and the release of multiple cytokines after exposure to bacterial components. Using proteomic screens and binding assays, the authors discovered that BDNF attaches directly to Toll‑like receptor 4 (TLR4), a major danger sensor on macrophages that recognizes bacterial endotoxin and launches powerful inflammatory cascades. BDNF competes with part of the usual TLR4 helper complex for access to the receptor’s outer domain, blocking the chain of signals that would normally amplify inflammation.
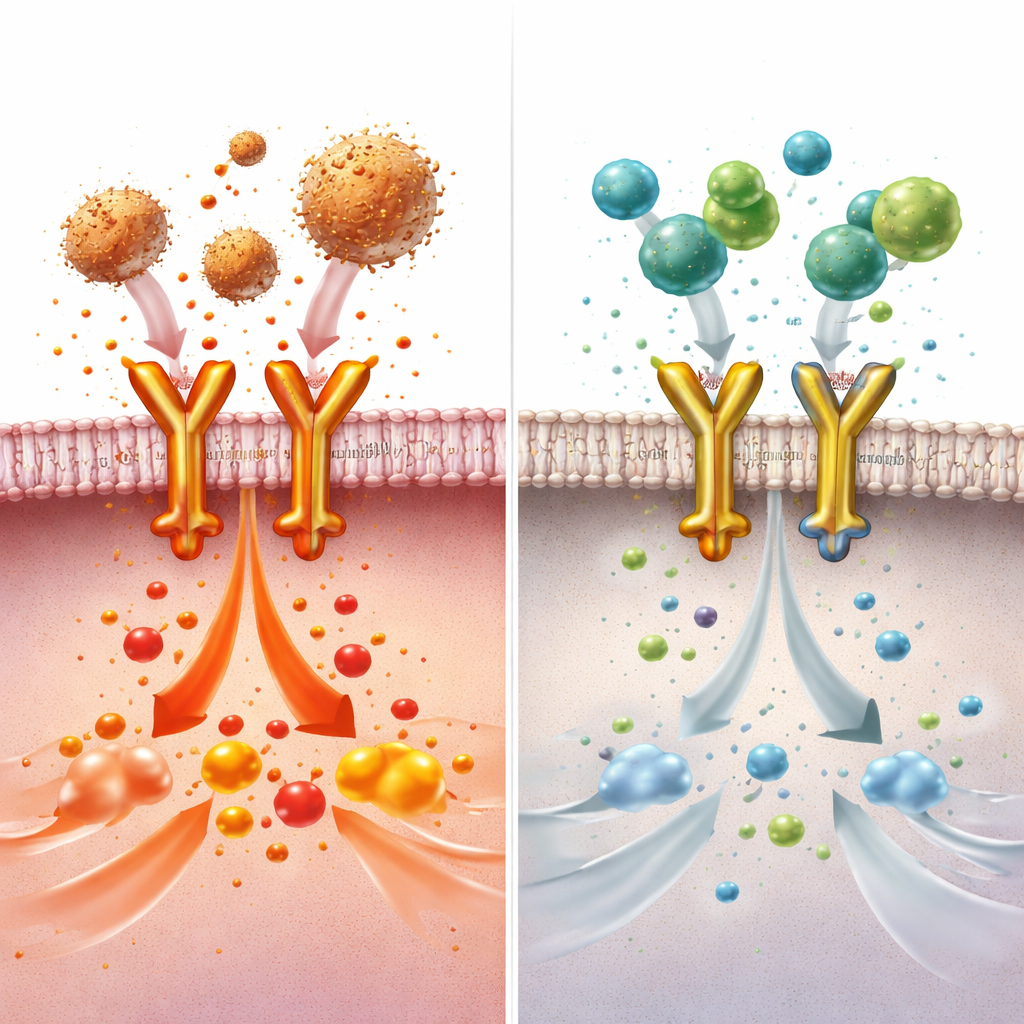
A tiny peptide with big potential
Because full‑length BDNF can stimulate cell growth through its classical nerve receptor and has been linked to cancer in some settings, the researchers searched for a smaller, safer fragment that preserves the anti‑inflammatory effect. By digesting BDNF and mapping which pieces bind to the outer portion of TLR4, they pinpointed a 12‑amino‑acid stretch, dubbed BDP‑12, that attaches strongly to TLR4 but does not activate growth‑promoting pathways. In cell tests, BDP‑12 blocked formation of the TLR4 helper complex and suppressed inflammatory gene activity more potently than full BDNF, and its action disappeared when TLR4 was removed, indicating high specificity. In mice with toxin‑induced lung injury or with polymicrobial sepsis, BDP‑12 reduced tissue damage, lung swelling, and immune‑cell infiltration in the lungs, heart, and kidneys, while improving survival. Importantly, repeated dosing did not spur extra cell proliferation in lung tissue, suggesting a lower risk of tumor‑like side effects.
What this could mean for future treatments
This work reveals a previously unrecognized conversation between lung surface cells and immune cells: epithelial BDNF normally helps hold macrophage‑driven inflammation in check by directly blocking a key danger receptor. When BDNF levels fall during severe infection, this brake is released and damage accelerates. By distilling BDNF down to the short BDP‑12 peptide that still antagonizes TLR4 but lacks growth‑stimulating activity, the study offers a promising blueprint for drugs that could calm overwhelming lung and whole‑body inflammation in acute lung injury, sepsis, and possibly other inflammatory diseases.
Citation: Zhu, W., Jin, L., Zhang, Q. et al. Brain-derived neurotrophic factor and the derived dodecapeptide function as Toll-like receptor 4 antagonists in acute lung injury. Nat Commun 17, 2786 (2026). https://doi.org/10.1038/s41467-026-69541-7
Keywords: acute lung injury, BDNF, TLR4, macrophage inflammation, therapeutic peptide