Clear Sky Science · en
Actinotrichia-independent developmental mechanisms of spiny rays facilitate the morphological diversification of Acanthomorpha fish fins
Why fin backbones matter for fish diversity
From flying fish that glide above the waves to anglerfish dangling glowing lures, many of the strangest fish tricks are built on one key structure: stiff fin spines. This study asks a deceptively simple question with big evolutionary consequences: what is different in the way these spines grow, compared with ordinary flexible fin rays? By uncovering that hidden construction plan, the authors show how small changes in cells and supporting materials can unlock a huge variety of forms.
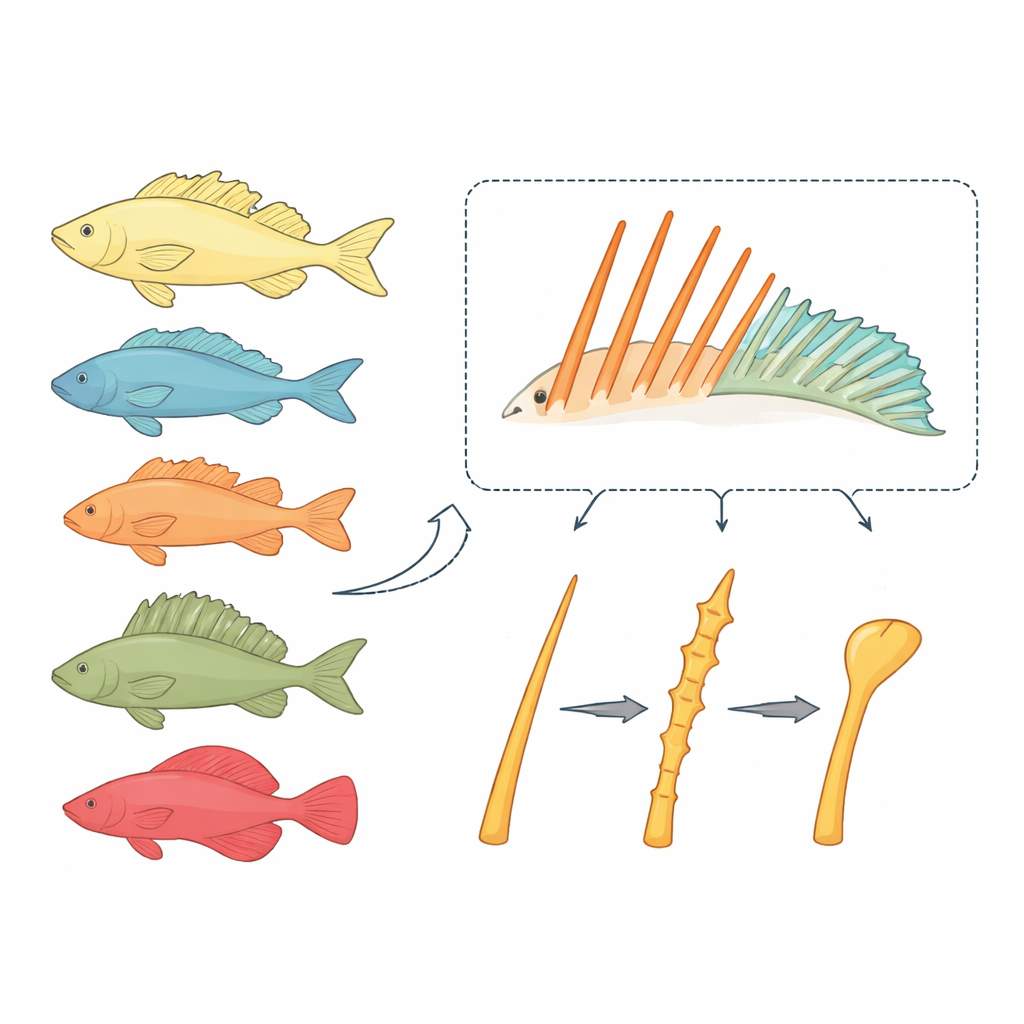
Two kinds of fin rods with very different fates
Bony fishes typically carry two types of fin supports: soft rays that bend and branch like tiny fingers, and spiny rays that are stiff, pointed rods. Soft rays rarely change their basic rod-like design, even when they grow very long. In contrast, spiny rays in the large fish group Acanthomorpha have repeatedly been reshaped into novel gadgets, including the suction disc of remoras and the fishing rod of anglerfish. The researchers suspected that the development of spiny rays follows its own rules, and that these rules might explain why spines are so evolutionarily flexible.
A new lab fish to watch spines grow
Traditional model species such as zebrafish and medaka are poorly suited for studying true spiny rays: zebrafish lack them, and medaka has only a rudimentary spine. The team instead turned to the dwarf neon rainbowfish, Melanotaenia praecox, a small freshwater species that develops clear sets of both spiny and soft rays and can be genetically manipulated. By staining growing bones at two time points, they showed that rainbowfish spiny rays elongate by adding new bone at their tips, much like soft rays. However, spines also thicken as the paired left and right halves fuse into a single rigid column, a hallmark of these structures.
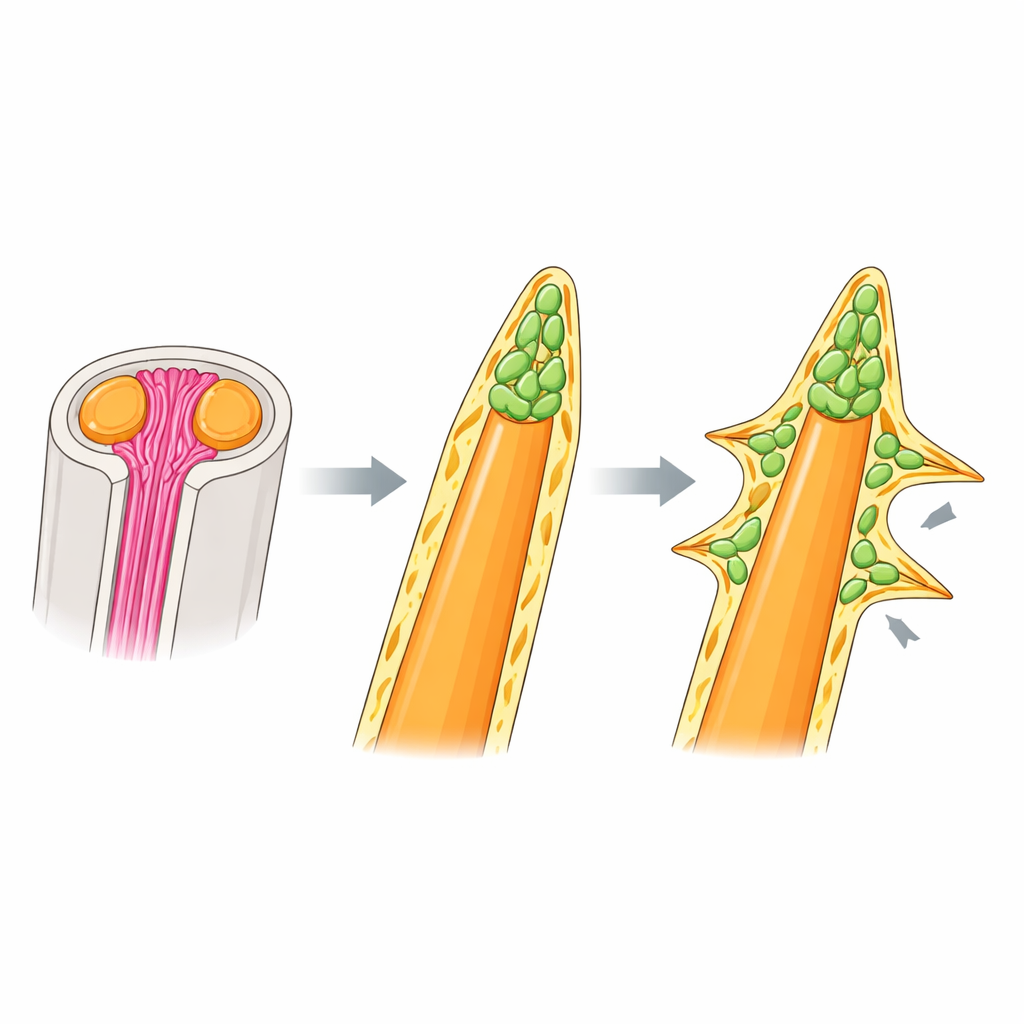
Letting go of collagen scaffolds and using cell caps instead
Soft rays in other fishes are known to rely on needle‑like collagen fibers, called actinotrichia, which bundle at the ray tips and guide bone-forming cells. Surprisingly, the authors found that rainbowfish spiny rays do not use this scaffold. Fluorescent collagen stains and expression of key actinotrichia genes appeared strongly at soft-ray tips but were weak or absent around spines, and mutants lacking actinotrichia showed curled, misshapen soft rays but perfectly normal spiny rays. Electron microscopy and 3D imaging revealed what spines use instead: a dense cap of mesenchymal cells at the tip of the bone, wrapped in a thick basement-membrane–like layer of extracellular matrix. This cap sits where actinotrichia would be in soft rays and appears to drive both elongation and thickening of the spine independently of collagen bundles.
Signaling that sculpts thorns and other spine shapes
Within the cell cap at rainbowfish spine tips, many cells carried markers of immature bone-forming cells (osteoblasts) and showed activity of BMP signaling, a pathway known to promote bone differentiation. When the team blocked BMP receptors with a drug, the spines did not turn into soft rays, but their tips became blunter and abnormally thick, with cells trapped inside the bone. This indicates that BMP signaling fine‑tunes how much bone is added and how neatly it is laid down, rather than determining whether a ray will be spiny or soft. To test whether tinkering with this same machinery could generate exotic spine shapes, the authors examined larvae of a filefish whose dorsal spine bears a row of thorny side projections. There too, clusters of immature osteoblasts with active BMP signaling appeared not only at the main tip but also at each lateral protrusion, suggesting that the basic “tip cap” program had been reused and repositioned to build thorns.
How flexible building rules fuel evolutionary invention
Taken together, the work paints spiny rays as structures built by mobile bone‑forming cells rather than tied to fixed collagen guides. Because their growth is not locked to straight actinotrichia bundles, the direction and location of spine expansion can change whenever osteoblast clusters and signaling hotspots shift along the bone. The authors argue that this developmental freedom made it easier for evolution to turn simple fin spines into the diverse, sometimes bizarre devices seen across acanthomorph fishes. More broadly, the study highlights how changes in where cells gather and which extracellular materials they use—without altering basic genetic toolkits—can drive major innovations in body shape across animals.
Citation: Miyamoto, K., Kuroda, J., Kamimura, S. et al. Actinotrichia-independent developmental mechanisms of spiny rays facilitate the morphological diversification of Acanthomorpha fish fins. Nat Commun 17, 2775 (2026). https://doi.org/10.1038/s41467-026-69180-y
Keywords: fish fin evolution, spiny rays, bone development, osteoblast signaling, morphological diversification