Clear Sky Science · en
Epitope mapping of vaccine antigens Tc24 and TSA1 with antibodies from Trypanosoma cruzi-infected patients
Why this research matters for heart health
Chagas disease, a parasitic infection common in Latin America and increasingly seen worldwide, can quietly damage the heart for years before causing serious problems such as heart failure and sudden death. Drugs exist but work best early and often cause strong side effects. Scientists are therefore developing a therapeutic vaccine to help the immune system control the parasite in already infected people. This study asks a simple but crucial question: do real patients naturally make antibodies that recognize the same vaccine ingredients now headed toward clinical trials, and are those targets stable across the many strains of the parasite?
The parasite, the threat, and the vaccine idea
Chagas disease is caused by the protozoan parasite Trypanosoma cruzi, which circulates in animals, insects, and humans. Many infected people feel fine for years, even though the parasite persists in their bodies. About one-third eventually develop chronic heart disease or digestive problems. Current drugs are most effective soon after infection and become less useful in the chronic stage, where they can also be hard to tolerate. Researchers have spent years testing a vaccine strategy based on two parasite proteins, Tc24 and TSA1, engineered into versions called Tc24-C4 and TSA1-C4 that are easier to produce but keep their key immune features. In mice, non-human primates, and even naturally infected dogs, these vaccine components have helped lower parasite levels and protect the heart.
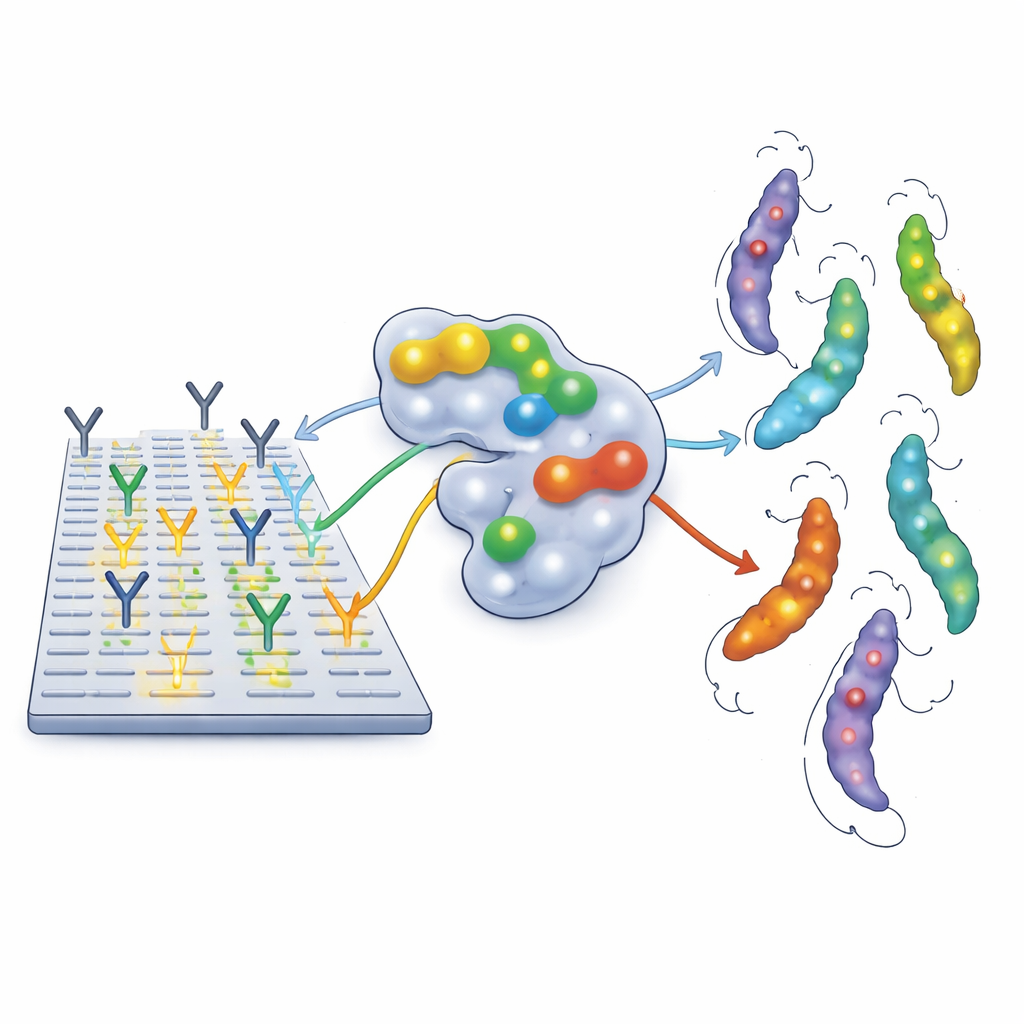
How the team read the immune “fingerprints”
To see exactly where human antibodies latch onto these vaccine proteins, the authors used a high-resolution technique called peptide microarrays. They broke each vaccine protein into a series of short, overlapping fragments and fixed these tiny pieces on a glass slide. Blood samples, collected from 27 pregnant women with documented T. cruzi infection in Argentina, Honduras, and Mexico, were processed to purify IgG antibodies and flowed across the slides. Wherever antibodies bound strongly, the spot lit up, revealing an “epitope” – a small region of the protein that the immune system recognizes. The researchers also checked how these recognized regions map onto the three-dimensional shapes of Tc24 and TSA1 and whether the same sequences appear in many different parasite strains.
A shared pattern across diverse patients and parasites
Most patients – nearly three-quarters – showed a strikingly similar antibody pattern. For Tc24-C4, their antibodies strongly recognized one main region in the middle of the protein and two weaker regions nearby. For TSA1-C4, they shared four main target regions, mostly near one end of the protein. These “immunodominant” spots were usually located on the outside surfaces of the folded proteins, where antibodies can easily reach them. When the team compared parasite genomes representing several major Trypanosoma cruzi lineages, they found that these key regions were highly conserved: the amino acid sequences changed very little from strain to strain, and copies of the Tc24 epitopes were abundant across genomes. This suggests that antibodies against these hotspots could, in principle, recognize a wide range of circulating parasites.
When immune responses broke the mold
Not every patient fit this dominant pattern. About one-quarter had little or no antibody binding to the main hotspots, instead recognizing scattered alternative regions, or in a few cases barely reacting to the vaccine proteins at all. The researchers examined whether this difference could be explained by standard diagnostic test results, parasite levels in the blood, age, pregnancy history, or the patients’ HLA genes, which help shape immune responses. None of these factors clearly separated the groups. Limited parasite genotyping also showed that individuals with the same dominant antibody pattern could be infected with very different mixtures of T. cruzi strains. This hints that other factors, such as how much of each protein the parasites express, how epitopes are displayed on their surface, or gradual “exhaustion” of parts of the immune system, may shape each person’s antibody profile.
What this means for future vaccines
For a vaccine meant to treat already infected people, it is encouraging that most patients naturally focus their antibodies on a small set of stable, shared regions of Tc24 and TSA1 that are present in many parasite strains and exposed on the protein surface. These regions offer clear targets for boosting protective immunity with vaccination and for tracking how antibody responses change in future clinical trials. At the same time, the minority of patients with atypical or weak recognition reminds us that not everyone’s immune system responds in the same way. Overall, this work provides a detailed map of where human antibodies land on two leading Chagas vaccine candidates and establishes a baseline for judging whether vaccination successfully reshapes those responses to better control this long-lasting, heart-damaging infection.
Citation: Dumonteil, E., Herrera, C. Epitope mapping of vaccine antigens Tc24 and TSA1 with antibodies from Trypanosoma cruzi-infected patients. Genes Immun 27, 195–202 (2026). https://doi.org/10.1038/s41435-026-00380-8
Keywords: Chagas disease, Trypanosoma cruzi, therapeutic vaccine, antibody epitopes, Tc24 and TSA1