Clear Sky Science · en
Expanding the genetic landscape of inherited metabolic diseases using long-read sequencing and transcriptomic profiling
Why hidden DNA changes matter for rare diseases
For many families living with rare inherited metabolic diseases, today’s most advanced genetic tests still fail to deliver a clear answer. This study explores why so many cases remain a mystery and shows how newer ways of reading DNA and measuring gene activity can uncover hidden glitches that traditional tests overlook. By zooming in on seven puzzling patients, the researchers reveal unusual types of genetic changes and offer a roadmap for improving diagnosis and care.
When standard genetic tests are not enough
Inherited metabolic diseases are a large group of conditions in which the body cannot correctly process certain fats, sugars, or other molecules. Doctors often first spot them through abnormal blood or urine tests in newborns or young children. The next step is usually to scan the protein-coding parts of the genome using exome or short-read genome sequencing. Yet even with these powerful tools, many patients only receive a partial answer, such as one faulty gene copy in a disorder that should require two, or no convincing change at all. The seven individuals in this study fell into that “diagnostic gap”: clinical and biochemical tests strongly pointed to a metabolic disease, but genetic testing left key questions unresolved.
Reading DNA in longer pieces and listening to RNA
To tackle these tough cases, the team combined two modern approaches. First, they used long-read sequencing, a technology that reads DNA in much larger pieces than standard tests, and focused it on the specific genes already suspected from metabolism results. Long reads make it easier to detect structural changes, such as duplicated segments, deep insertions, and complex rearrangements. Second, they analyzed RNA, the molecules produced when genes are turned on, to see how these DNA changes actually affect gene messages—whether exons are skipped, extra pieces are spliced in, or expression levels drop. Together, these tools provided both a structural map and a functional readout of each patient’s genetic landscape.
Unusual genetic culprits come to light
This combined strategy paid off. In six of the seven patients, the researchers discovered “missing” disease-causing variants that earlier tests had missed. They found a duplicated exon that likely disrupts a key mitochondrial gene, and two deep intronic changes that create extra segments in gene messages and trigger early stop signals. Strikingly, three patients carried new insertions of mobile DNA fragments called transposable elements. Two of these landed within metabolic genes themselves, leading to exon skipping and unstable messages. The third sat just beyond a glucose transporter gene, in a region that helps control when and how strongly that gene is switched on. In several cases, the team used tailored laboratory assays—such as miniature gene constructs and reporter systems—to prove that these unusual variants damaged normal gene function.
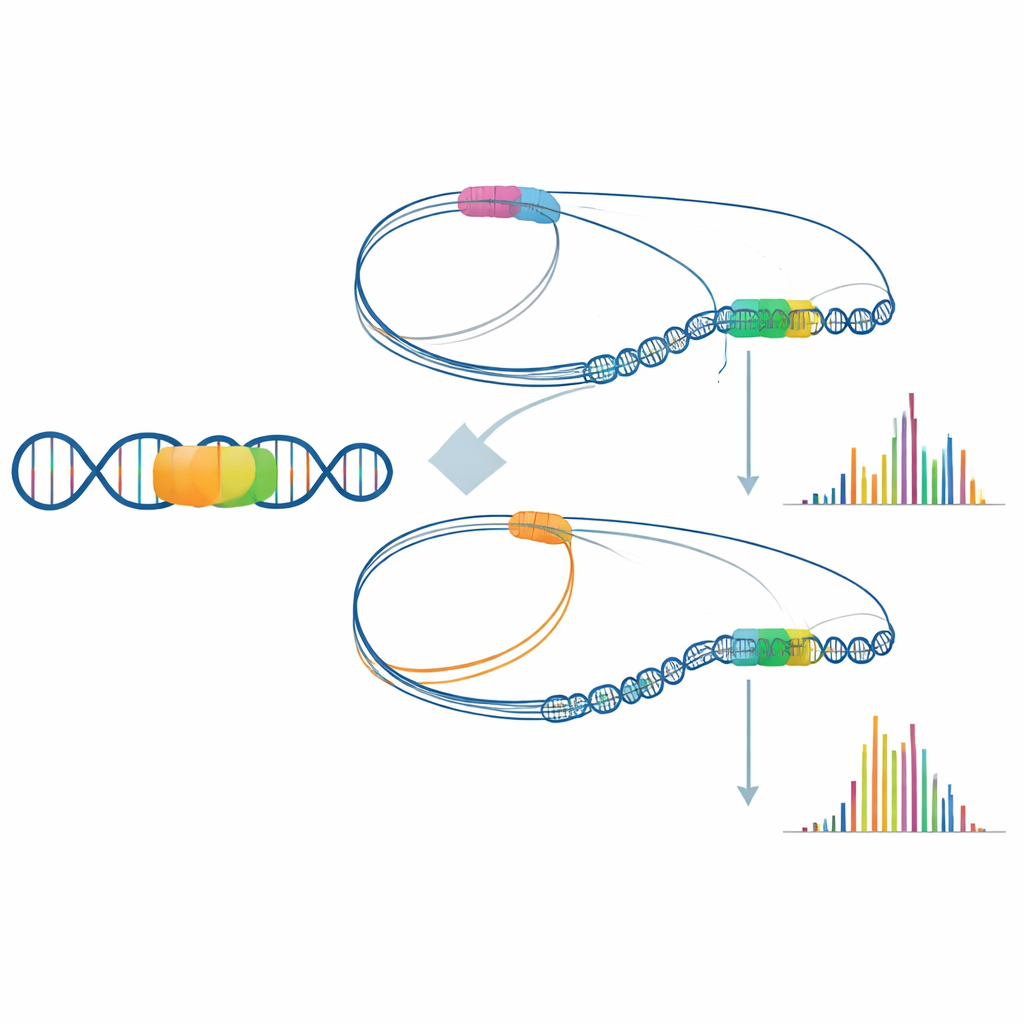
How 3D genome folding can cause disease
One especially revealing case involved the gene that moves glucose into brain cells. Although the gene’s coding sequence was intact, the patient’s cells produced much less of its RNA. Long-read sequencing uncovered a large mobile DNA insertion located several thousand bases away from the gene. By mapping the three-dimensional folding of the surrounding chromatin, the researchers showed that, in healthy cells, the gene’s control region regularly contacts an upstream cluster of enhancer elements, forming an “active hub” that supports strong expression. In the patient, these enhancer contacts were partially rewired toward the new insertion site, weakening the link between enhancer and gene. This subtle rewiring of genome architecture likely contributes to the energy shortage in the patient’s brain.
What this means for patients and future screening
Overall, the study demonstrates that focusing long-read sequencing on suspected genes, combined with careful RNA and functional assays, can uncover a wider range of disease-causing changes than standard methods alone. It highlights that mobile DNA insertions, deep intronic variants, and disruptions of the genome’s 3D folding are underappreciated contributors to inherited metabolic diseases. As genomic testing becomes more common, especially in newborns, incorporating these advanced tools and building better reference datasets could help more families move from uncertainty to precise diagnosis and, ultimately, more targeted treatment options.
Citation: Soriano-Sexto, A., Sánchez-Lijarcio, O., Beccari, L. et al. Expanding the genetic landscape of inherited metabolic diseases using long-read sequencing and transcriptomic profiling. Eur J Hum Genet 34, 543–553 (2026). https://doi.org/10.1038/s41431-025-01995-7
Keywords: inherited metabolic diseases, long-read sequencing, transposable elements, 3D genome organization, rare disease diagnosis