Clear Sky Science · en
NFATC2-mediated CST1 upregulation drives cholangiocarcinoma growth and metastasis
Why this liver cancer story matters
Intrahepatic cholangiocarcinoma is a deadly cancer that starts in the small bile ducts inside the liver. It is often discovered late, spreads quickly, and responds poorly to standard treatments. This study uncovers a hidden molecular chain that helps these tumor cells avoid aging, grow relentlessly, and spread to distant organs. By exposing this chain, the work points to new ways doctors might slow or stop this hard‑to‑treat cancer.
A closer look at a quiet but deadly liver cancer
Cholangiocarcinoma accounts for only a fraction of liver cancers, but it causes a disproportionate number of cancer deaths worldwide. One reason is that early symptoms are vague or absent, so many patients are diagnosed only after the disease has advanced. The authors began by mining large public gene‑expression databases, along with tumor samples from their own hospital, to search for genes that are consistently more active in bile‑duct tumors than in normal tissue and that also predict poor patient survival. Among hundreds of altered genes, one stood out: CST1, which encodes a small secreted protein called cystatin SN. Patients whose tumors had high levels of CST1 lived significantly shorter lives, marking CST1 as both a warning sign and a potential driver of the disease.
How a single protein boosts growth and spread
To test CST1’s role, the researchers used bile‑duct cancer cell lines in which they could dial CST1 levels up or down. When they forced cells to make more CST1, the cells multiplied faster, moved more readily through porous barriers, and more easily invaded through a gel mimicking surrounding tissue. When they reduced CST1, cell growth slowed, cells stalled in an early phase of the cell cycle, and their ability to migrate and invade dropped. In mice, tumors engineered to overproduce CST1 formed larger nodules in the liver and seeded more metastases in the lungs, while tumors with CST1 turned down grew more slowly and spread less. Together, these experiments show that CST1 is not just a bystander marker, but an active promoter of tumor growth and dissemination.
Turning off cellular aging to escape natural brakes
Normal cells have a built‑in safety system called senescence: when they accumulate enough damage, they enter a permanent rest state and stop dividing. This acts as a barrier against cancer. The team found that lowering CST1 in cancer cells switched this safety system back on. Cells with reduced CST1 showed classic signs of senescence, including changes in shape, increased activity of a senescence‑linked enzyme, and shifts in inflammatory factors they secreted. Surprisingly, although CST1 also changed how cells handled certain small molecules used to build DNA, attempts to rescue growth by supplying extra building blocks failed. This suggested that CST1’s main power lies not in feeding DNA synthesis, but in helping tumor cells sidestep the senescence program that would otherwise hold them in check.
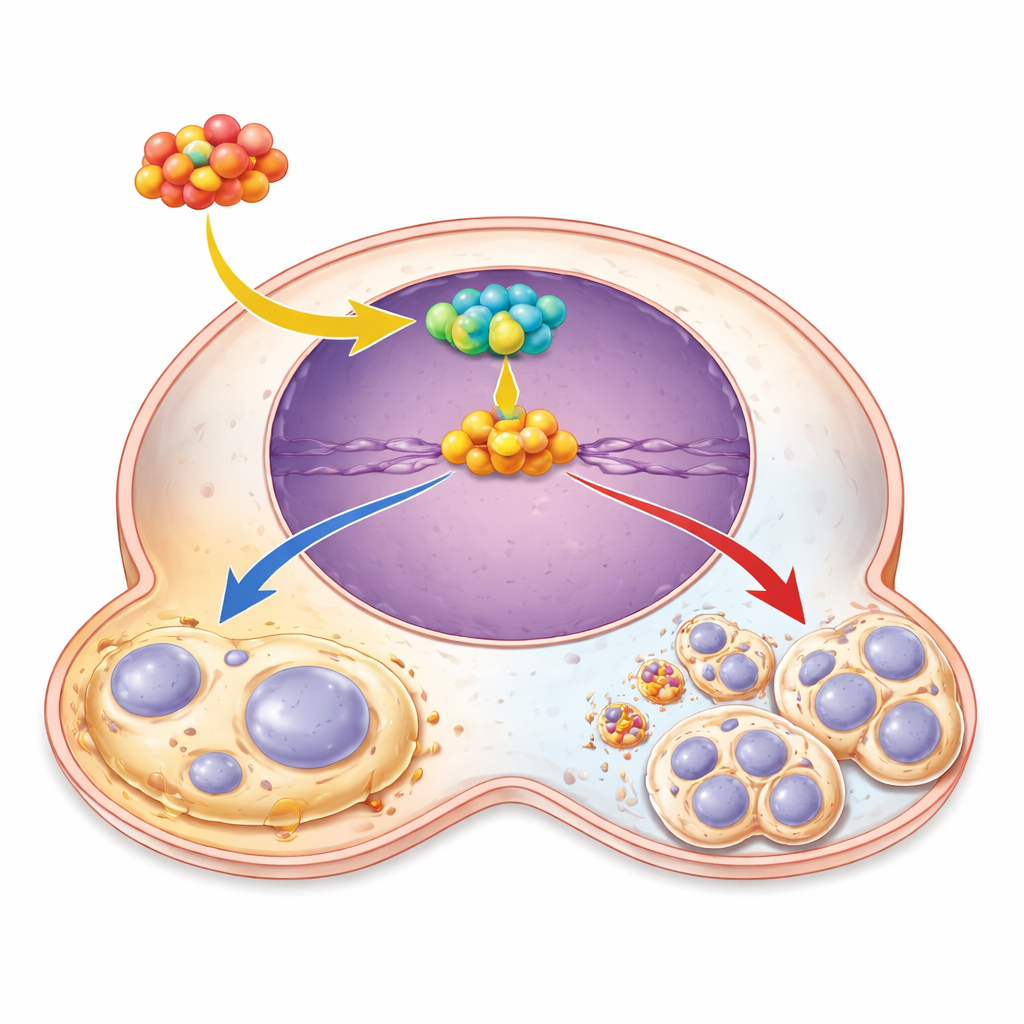
An oncogenic relay: from NFATC2 to CST1 to SOX4
Digging deeper, the researchers pieced together a three‑step molecular relay inside cancer cells. At the top is NFATC2, a transcription factor that binds to DNA and turns specific genes on. They showed that NFATC2 docks directly on the CST1 gene’s control region and boosts its activity. When NFATC2 levels were high, CST1 went up; when NFATC2 was silenced, CST1 levels fell and tumor‑promoting behaviors weakened. Downstream of CST1, they identified another key player, the transcription factor SOX4, which has been linked to blocking senescence in other cancers. CST1 raised SOX4 levels, and when CST1 was knocked down, adding extra SOX4 was enough to restore cell growth and reduce senescence signals. This positions SOX4 as the main executor of CST1’s anti‑aging, pro‑growth effects.
What this means for future treatments
By mapping this NFATC2–CST1–SOX4 relay, the study reveals how bile‑duct cancer cells disable their internal aging brakes, allowing unchecked growth and spread. To a layperson, the message is that these tumors co‑opt a small network of proteins to keep cells forever young and dividing. Disrupting this chain at any point—blocking NFATC2’s control over CST1, reducing CST1 itself, or cutting off SOX4—could restore the cell’s natural tendency to slow down and stop. While more work is needed before these insights translate into new drugs, CST1 in particular emerges as a promising target for therapies and as a marker that might help identify patients at higher risk.
Citation: Zhao, W., Zhao, J., Li, K. et al. NFATC2-mediated CST1 upregulation drives cholangiocarcinoma growth and metastasis. Cell Death Discov. 12, 187 (2026). https://doi.org/10.1038/s41420-026-03036-8
Keywords: cholangiocarcinoma, CST1, cellular senescence, cancer metastasis, NFATC2 SOX4 pathway