Clear Sky Science · en
RRx-001 inhibits G6PD to deplete NADPH and trigger disulfidptosis coupled with DAMP-mediated immunogenic cell death in hepatocellular carcinoma
Why this study matters for liver cancer
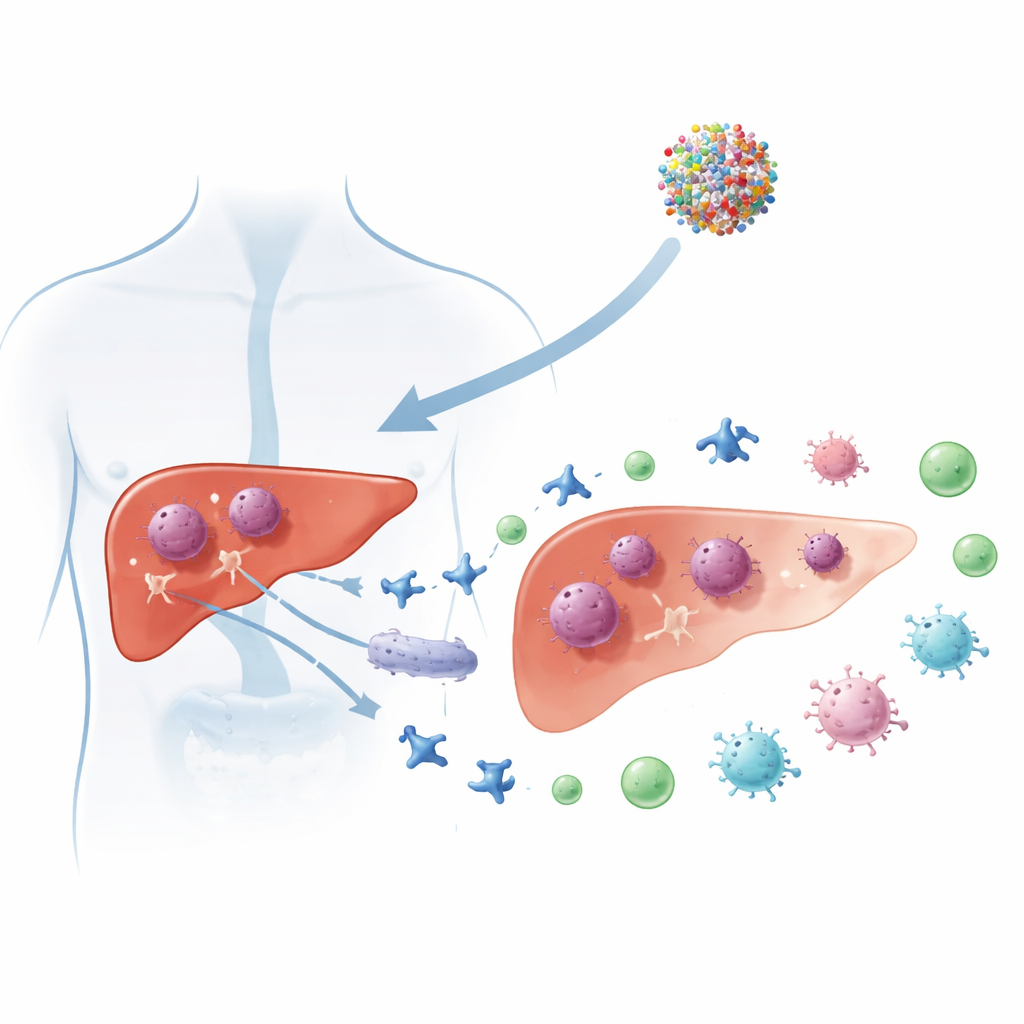
Primary liver cancer, especially hepatocellular carcinoma, remains one of the deadliest cancers worldwide, with most patients diagnosed too late for curative surgery. This study explores whether a clinical-stage experimental drug, RRx-001, can attack liver tumors in an unusually smart way: by cutting off a key source of cellular “battery power” and at the same time turning dying cancer cells into a vaccine-like signal that wakes up the immune system.
A new way for cancer cells to die
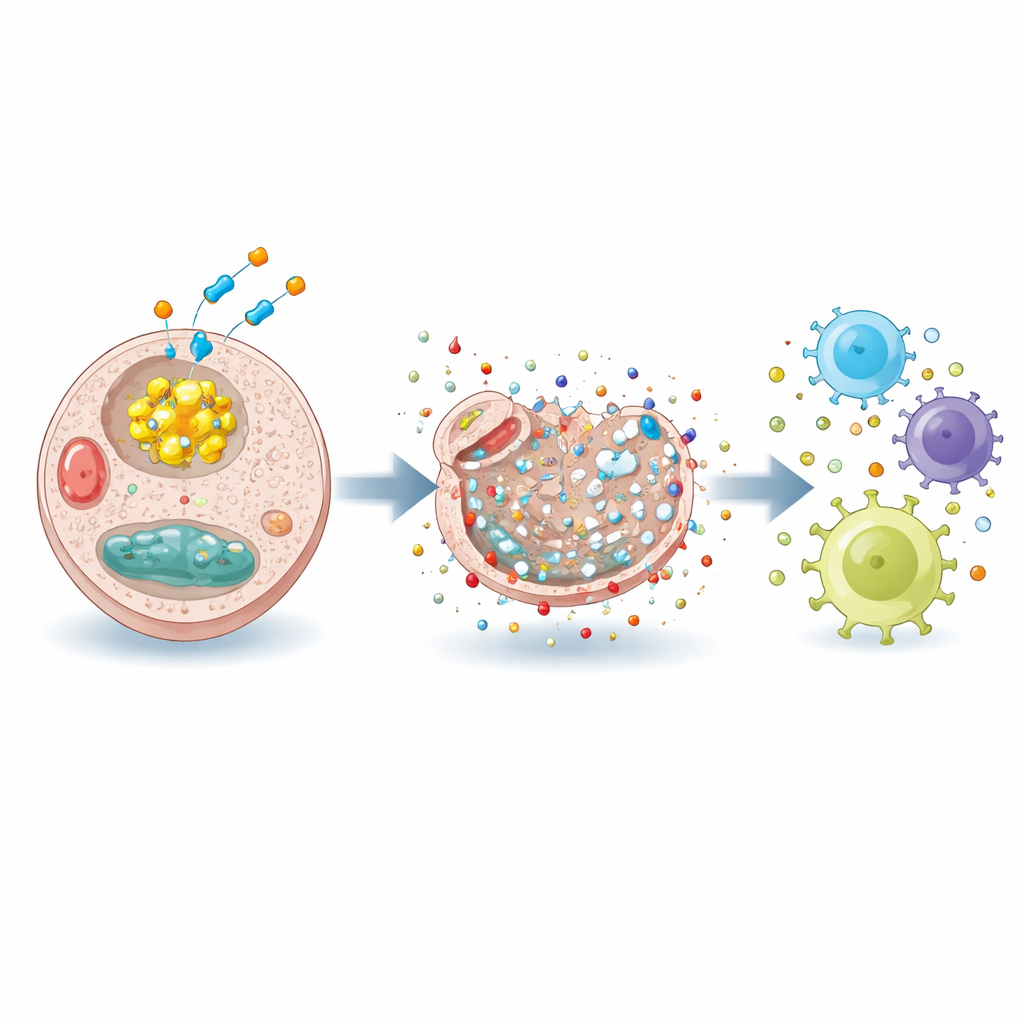
Scientists increasingly view cancer treatment as a matter of controlling how tumor cells live and die. Beyond well-known forms of cell death like apoptosis, researchers have recently identified “disulfidptosis,” a type of programmed death that happens when cells can no longer manage certain chemical bonds called disulfides in their internal skeleton. When this balance collapses, the scaffolding that keeps cells in shape contracts and falls apart, leading to rapid cell death. The promise of disulfidptosis is that it exploits a weakness in cancer cells’ altered metabolism, potentially allowing more selective tumor targeting.
Targeting the tumor’s power supply
Cancer cells rely heavily on the molecule NADPH to keep their internal chemistry stable and to neutralize harmful reactive molecules. A major source of NADPH is a metabolic route called the pentose phosphate pathway, controlled by an enzyme named G6PD. In this study, the researchers compared different compounds that block several NADPH-producing routes and found that RRx-001 was the most potent at killing liver cancer cells in the lab. Unlike other inhibitors, the cell death caused by RRx-001 could be partly reversed by a chemical that specifically breaks disulfide bonds, and the cells showed a characteristic collapse of their internal actin fibers. These hallmarks indicated that RRx-001 was triggering disulfidptosis, not just generic toxicity.
From metabolic collapse to immune alarm
By blocking G6PD, RRx-001 drained NADPH, disturbed the balance between protective and oxidized glutathione, and allowed damaging reactive oxygen and nitrogen species to build up. This chemical stress pushed liver cancer cells into disulfidptosis, but it also did something more: it caused the cells to expose and release so-called danger signals, or DAMPs, including molecules such as calreticulin and heat shock proteins. Under an electron microscope, treated cells showed damaged organelles and disrupted membranes, changes consistent with a form of cell death that can rally the immune system rather than pass silently.
Turning tumors from “cold” to “hot”
The team then tested RRx-001 in mice carrying liver tumors. Tumors treated with the drug grew more slowly and weighed less than those in untreated animals. Detailed analysis of the tumor tissue revealed increased infiltration of helper (CD4+) and killer (CD8+) T cells, a shift in macrophages toward a more inflammatory, tumor-fighting type, and reduced levels of PD-L1, a surface protein that tumors use to switch off immune attacks. Gene activity patterns in the tumors showed activation of T cell receptor signaling and T helper cell differentiation pathways, all signs that the immune system had been mobilized rather than suppressed.
What this could mean for future treatments
Together, the findings suggest that RRx-001 attacks liver cancer on two fronts: it exploits a metabolic Achilles’ heel by starving cells of NADPH and forcing them into disulfidptosis, and it simultaneously turns their death into an immune-stimulating event known as immunogenic cell death. For patients, this raises the possibility of treatments that not only shrink tumors directly but also help the body recognize and fight cancer more effectively. The authors propose that RRx-001 could be especially powerful when combined with modern immune checkpoint inhibitors, potentially converting hard-to-treat “cold” liver tumors into “hot” ones that respond better to immunotherapy.
Citation: Huang, H., He, Y., Chen, J. et al. RRx-001 inhibits G6PD to deplete NADPH and trigger disulfidptosis coupled with DAMP-mediated immunogenic cell death in hepatocellular carcinoma. Cell Death Discov. 12, 194 (2026). https://doi.org/10.1038/s41420-026-03032-y
Keywords: hepatocellular carcinoma, cancer metabolism, immunogenic cell death, tumor immunotherapy, RRx-001