Clear Sky Science · en
ZDHHC9 palmitoylates LAMTOR1 to promote renal cell carcinoma malignant progression
Why this kidney cancer study matters
Renal cell carcinoma, a common and often deadly form of kidney cancer, frequently becomes resistant to available drugs, leaving patients with few options. This study uncovers a hidden molecular switch inside cancer cells that helps drive their rapid growth and may explain why some tumors shrug off current treatments. By pinpointing this switch, the work suggests a new way to weaken kidney tumors and make existing drugs work better.
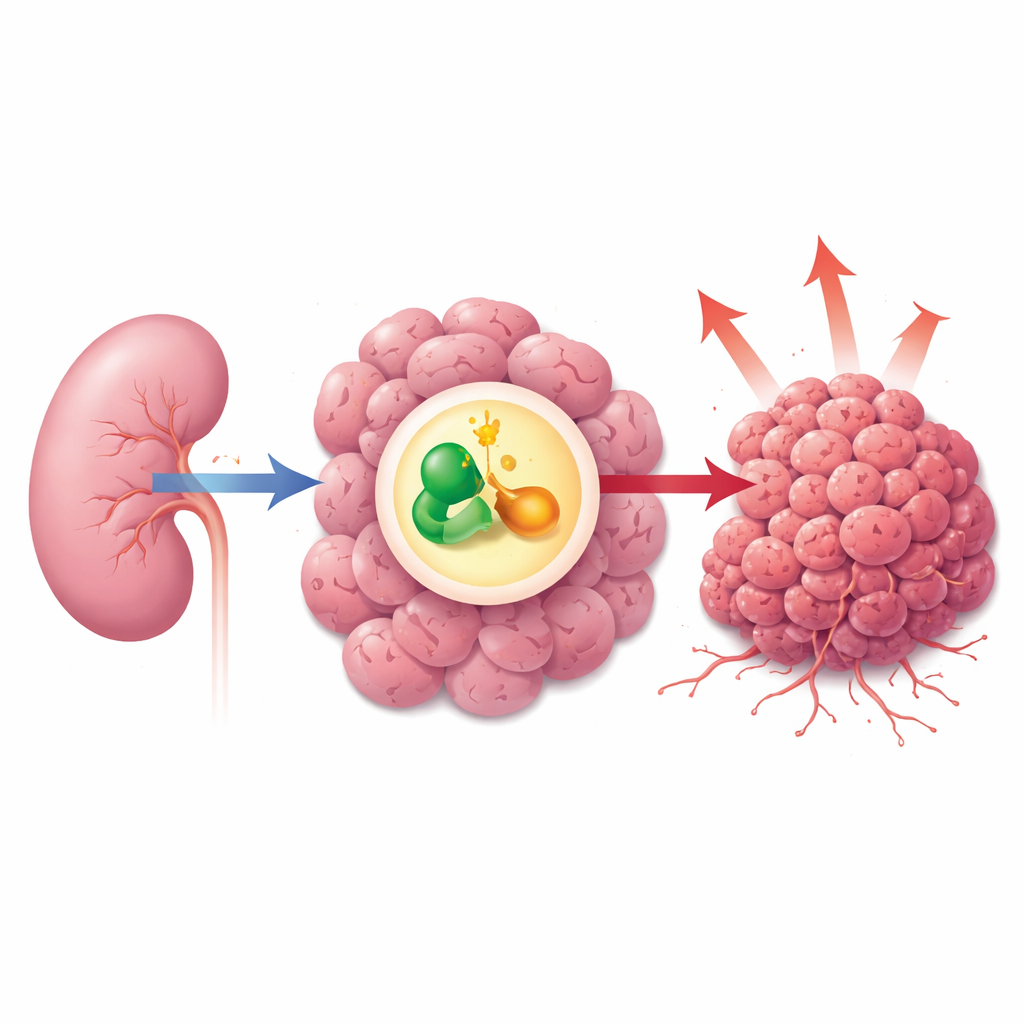
A growth switch deep inside the cell
Cancer cells rely on powerful internal growth circuits to divide and spread. One major circuit is the mTOR pathway, which senses nutrients and energy and then tells cells whether to grow or stay quiet. In more than half of kidney cancers, this pathway is abnormally active. Doctors already use mTOR-blocking drugs such as rapamycin, but tumors often adapt and regain their growth advantage. The authors suspected that a lesser-known protein called ZDHHC9 might be one of the hidden controllers that keeps this pathway switched on in renal cell carcinoma.
Finding an overactive helper in kidney tumors
The researchers first compared tumor samples from kidney cancer patients with nearby normal kidney tissue. They found that ZDHHC9 levels were consistently higher in cancer cells than in healthy cells, both at the RNA and protein level. When they lowered ZDHHC9 in kidney cancer cell lines, the cells divided more slowly, formed fewer colonies, and underwent more programmed cell death. In mice, tumors built from cells lacking ZDHHC9 grew much more slowly and weighed less than control tumors. Together, these results show that ZDHHC9 behaves like an “accelerator pedal” for kidney tumor growth.
How ZDHHC9 boosts the cell’s growth engine
ZDHHC9 belongs to a family of enzymes that attach small fat-like molecules to other proteins, a chemical tweak that can change where those proteins sit inside the cell and how they interact. Using genetic and biochemical tests, the team discovered that ZDHHC9 physically binds to another protein called LAMTOR1, which acts as a docking platform on the surface of lysosomes—small compartments that help coordinate mTOR signaling. ZDHHC9 adds fat groups to two specific positions on LAMTOR1, which anchors LAMTOR1 more firmly at the lysosome. This, in turn, makes it easier for the mTOR complex to gather at that site and become activated.
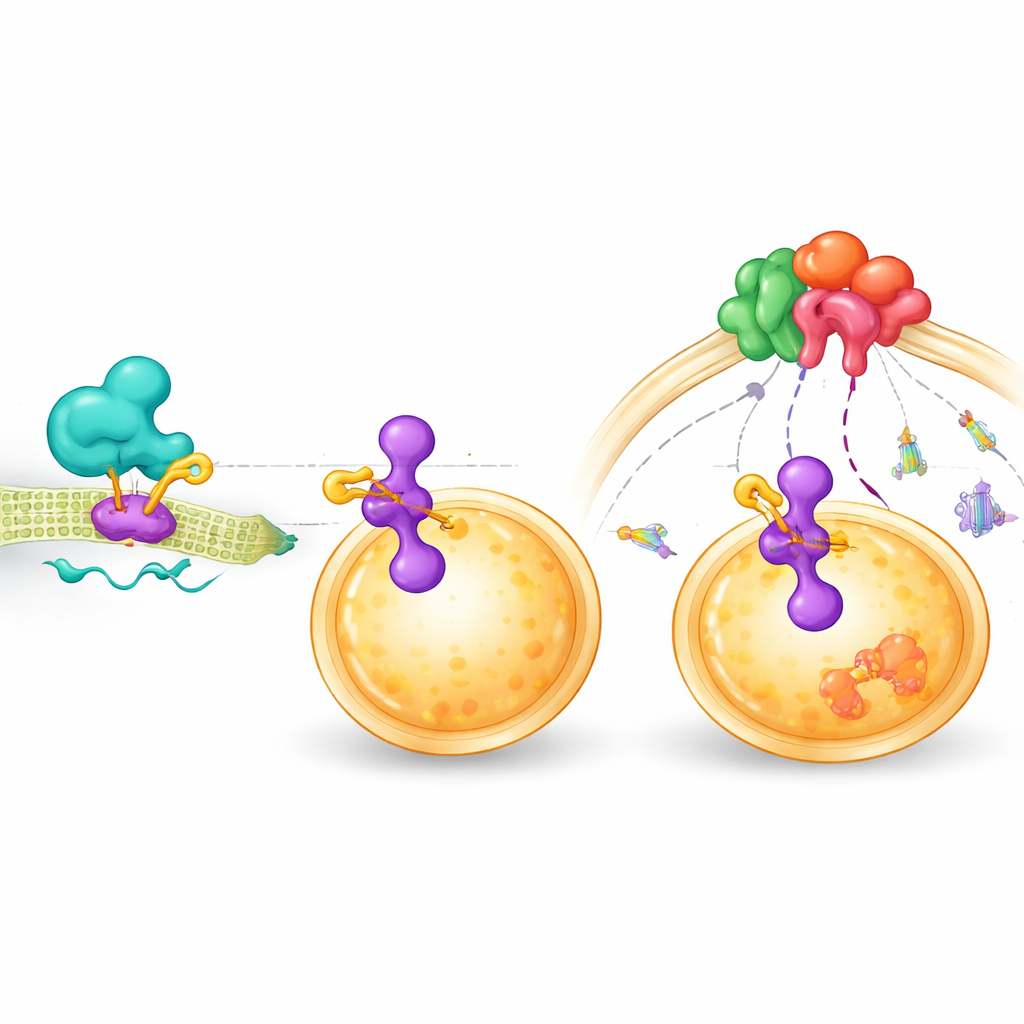
Disrupting the chain from signal to tumor growth
When the researchers blocked this fat-adding step with a chemical inhibitor, or when they altered either the working site of ZDHHC9 or the key sites on LAMTOR1, the chain reaction was interrupted. Less mTOR reached the lysosome, and its downstream growth signals dropped. Cells treated this way showed reduced survival and more signs of self-destruction. Importantly, when ZDHHC9 was reduced, standard mTOR-blocking drugs such as rapamycin and AZD-8055 became more effective, both in cell cultures and in mouse tumors. The combined treatment slowed tumor growth more strongly than either approach alone and triggered higher levels of cell death markers in the cancer tissue.
What this means for future kidney cancer treatment
To a layperson, the study shows that kidney tumors are not driven by a single faulty switch, but by a chain of helpers that keep the growth engine humming. ZDHHC9 acts as a hidden technician, modifying LAMTOR1 so that the mTOR growth hub is locked onto the cell’s inner surface and kept active. By targeting ZDHHC9, doctors might be able to loosen this grip, making tumors more sensitive to existing mTOR drugs and slowing disease progression. While this work is still at the experimental stage, it highlights a promising new target that could one day improve treatment for patients with renal cell carcinoma.
Citation: Liu, B., Hou, T., Liu, X. et al. ZDHHC9 palmitoylates LAMTOR1 to promote renal cell carcinoma malignant progression. Cell Death Dis 17, 323 (2026). https://doi.org/10.1038/s41419-026-08558-8
Keywords: renal cell carcinoma, mTOR signaling, protein palmitoylation, ZDHHC9, LAMTOR1