Clear Sky Science · en
Complement receptor 3 (CR3)-dependent microglial synapse elimination drives Parkinson’s disease pathogenesis in systemic inflammation
Why this study matters for everyday health
Parkinson’s disease is best known for causing tremors and stiffness, but long before these symptoms appear, subtle damage is already unfolding in the brain. This study shows how a strong immune reaction in the body – such as from infection or chronic inflammation – can quietly strip away the tiny connections between brain cells that control movement, setting the stage for Parkinson’s-like degeneration. Understanding this early “silent” phase may open a window for treatments that prevent or slow the disease before major damage occurs.
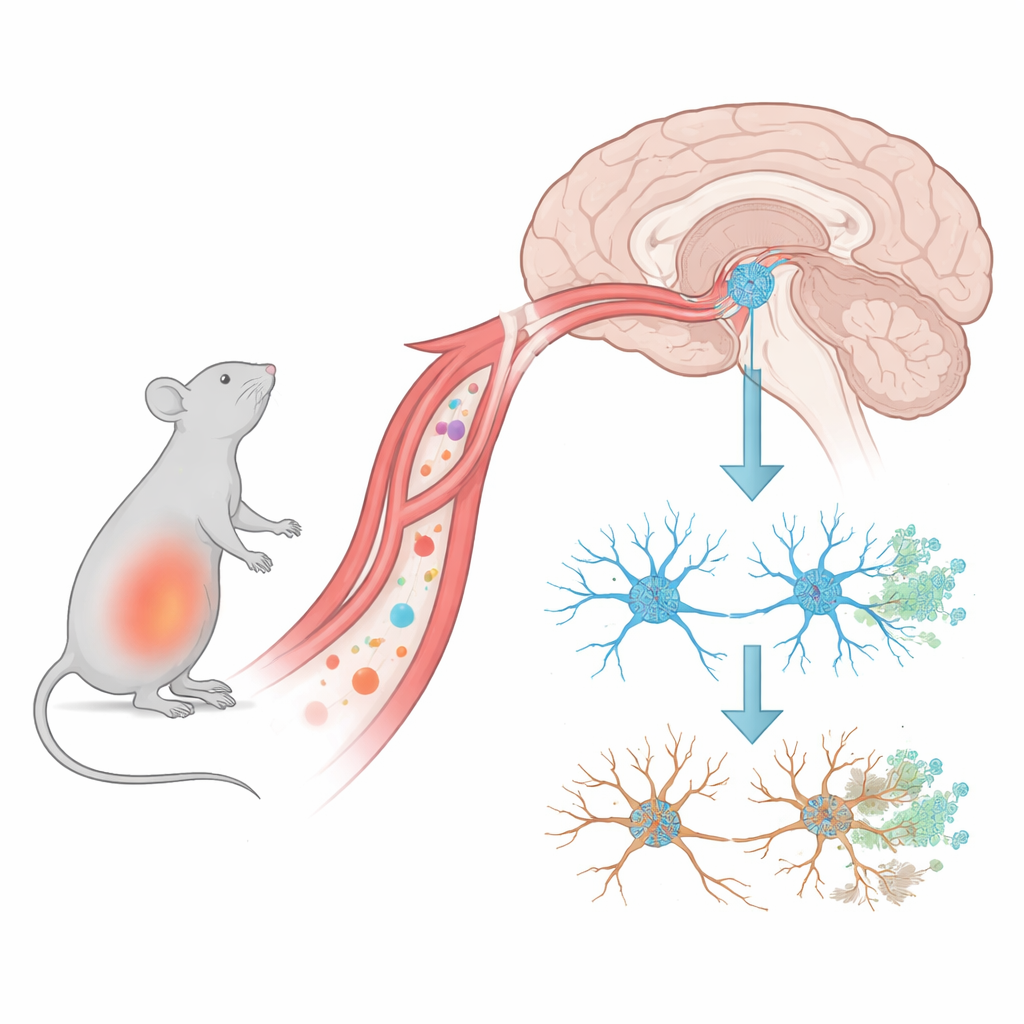
Early trouble at the brain’s communication points
The authors used mice exposed to a bacterial component called LPS to mimic a burst of body-wide inflammation. They then tracked changes in a midbrain region that houses dopamine-producing nerve cells, which are crucial for smooth movement and are heavily affected in Parkinson’s disease. They found that within just one day of the final inflammatory hit, the number of synapses – the microscopic contact points where nerve cells talk to each other – had already dropped. In contrast, the dopamine-producing nerve cells themselves did not noticeably die until about two weeks later. This timing suggests that the earliest damage in this Parkinson’s-like state is not the death of neurons, but the quiet loss of their connections.
Brain immune cells as overzealous cleaners
The brain’s resident immune cells, called microglia, act like sentinels and housekeepers: they survey the environment and remove debris, including weak or unnecessary synapses. In the inflamed mice, the team saw that microglia in the midbrain became highly activated as soon as synapse loss began. These cells swelled in size, changed shape, and showed increased levels of internal “digestive” structures, signs that they were in an aggressive cleaning mode. Three-dimensional imaging revealed that microglia were actually swallowing up both the sending and receiving sides of synapses. This intense pruning was strongest one day after inflammation and then gradually waned, even though the earlier wave of synapse loss set the stage for later neuron death.
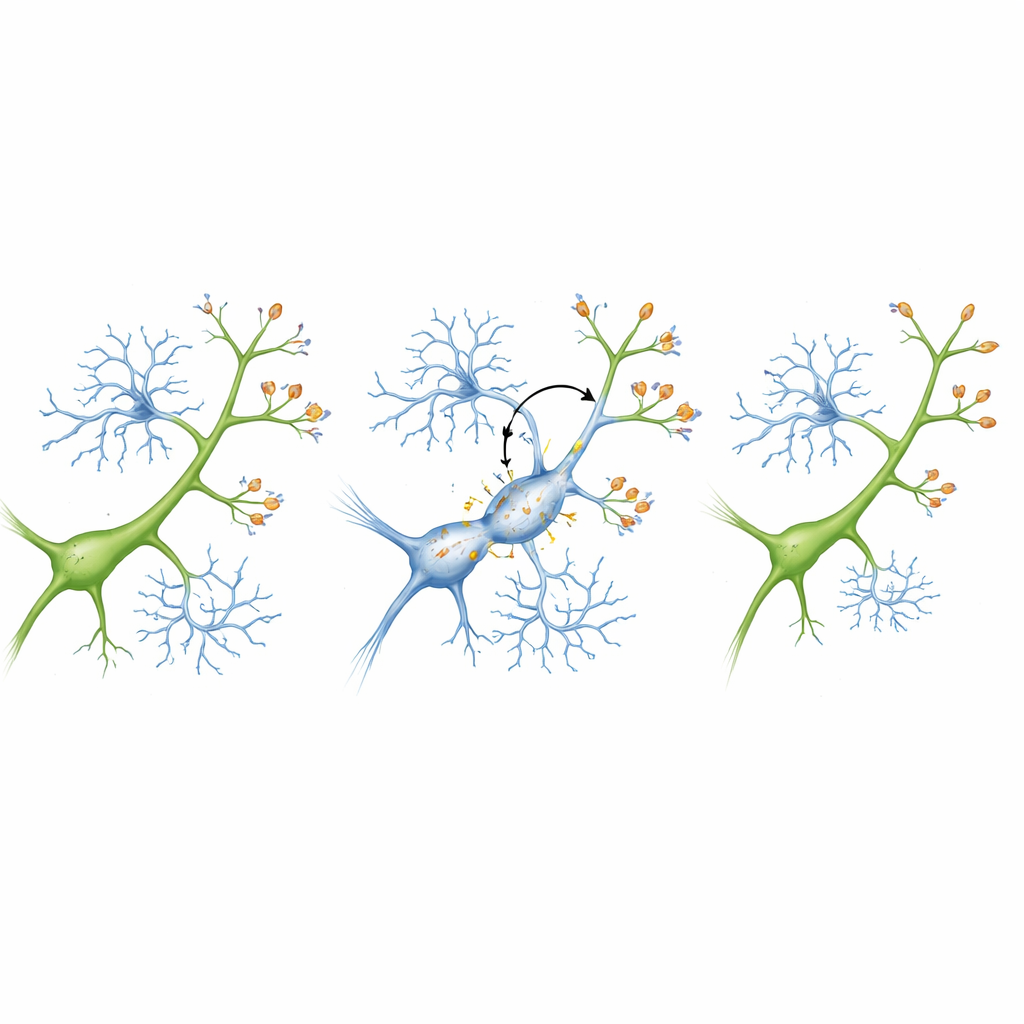
A tagging system that marks synapses for removal
To understand why microglia suddenly turned on healthy-looking synapses, the researchers focused on a molecular “tagging” system known as the complement pathway. In this system, small proteins coat targets that are meant to be cleared away, and microglia carry receptors that recognize these tags. The team found that one complement protein, C3, became more abundant on synapses in the inflamed mice, while a matching receptor, CR3, increased on microglia. This pairing effectively flagged synapses for removal. When the authors recreated the system in lab dishes containing neurons and microglia, inflammation alone did not harm isolated neurons. But when microglia were present, inflammation sharply reduced synapse numbers – and blocking C3 or reducing CR3 on microglia protected those synapses and kept neurons healthier.
Switching off the damaging pathway in living brains
The researchers then tested whether shutting down CR3 specifically in microglia could protect the brains of living mice. They used a viral tool to reduce CR3 only in microglia before inducing systemic inflammation. In these animals, complement activity was dampened, microglia were less activated, and they engulfed far fewer synapses. As a result, synapse density in the midbrain was better preserved soon after inflammation, and weeks later the dopamine-producing neurons were far less likely to die. Microglia also showed a more normal, branching shape rather than the compact, attack-ready form seen under unchecked inflammation, suggesting that blocking CR3 kept them from entering a harmful overdrive state.
What this means for future Parkinson’s care
This work reframes early Parkinson’s-like damage as a problem of misguided cleanup rather than immediate cell death. Systemic inflammation appears to turn on a synapse-tagging system that causes microglia to strip away crucial connections through the C3–CR3 pathway; only later do dopamine-producing neurons succumb. For a layperson, the takeaway is that brain health depends not only on the survival of nerve cells but also on the preservation of their communication lines, especially during bouts of strong immune activation in the body. By targeting the specific molecules that tell microglia which synapses to eat, future therapies may be able to protect these connections and slow or prevent the progression of Parkinson’s disease.
Citation: Cai, L., Zhang, Y., Li, J. et al. Complement receptor 3 (CR3)-dependent microglial synapse elimination drives Parkinson’s disease pathogenesis in systemic inflammation. Cell Death Dis 17, 319 (2026). https://doi.org/10.1038/s41419-026-08557-9
Keywords: Parkinson’s disease, microglia, synapse loss, neuroinflammation, complement system