Clear Sky Science · en
NSUN2 promoted tumor growth and metastatic via m5C-regulation of YAP through ALYREF/YBX1 axis in NSCLC
Why this lung cancer study matters
Lung cancer is the deadliest cancer worldwide, and most deaths come from non-small cell lung cancer (NSCLC), which often spreads before it is caught. This study digs into the molecular "wiring" that helps lung tumors grow and seed metastases throughout the body. By uncovering a specific chain of molecules that boosts a powerful growth switch inside cancer cells, the research points to new drug targets that could slow or stop the disease while sparing healthy tissue.
A hidden layer of control in cancer cells
Our genes are carried in DNA, but it is RNA—the working copy of genetic information—that tells cells which proteins to build and when. In recent years, scientists have discovered that RNAs themselves carry chemical marks, much like notes in the margins of a script, that can change how long they last and how strongly they are read. One such mark is called m5C, a small chemical tag placed on the RNA base cytosine. This work shows that in NSCLC, an enzyme called NSUN2 adds extra m5C marks to the RNA that encodes YAP, a master regulator of growth and survival. Tumors from patients had higher NSUN2 and YAP levels than nearby normal tissue, and patients with more of either protein tended to fare worse.
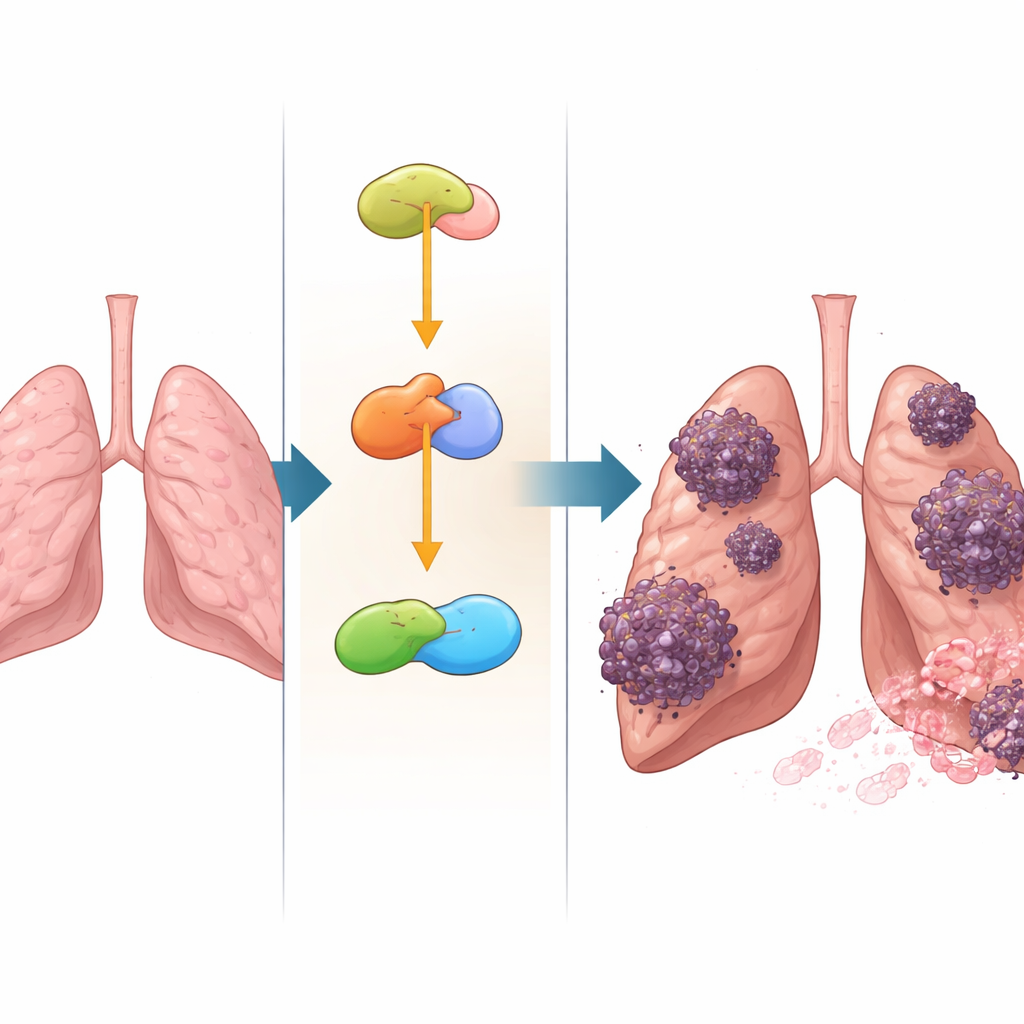
Turning up a dangerous growth switch
YAP acts like a central throttle for cell proliferation, movement, and shape change. When overactive, it can push cells to divide faster, invade surrounding tissue, and break away to form metastases. The researchers found that NSUN2 and YAP drive many of the same cancer behaviors in NSCLC cells: boosting cell division, migration, invasion through artificial barriers, and a process called epithelial–mesenchymal transition, in which cells loosen their attachments and become more mobile. In mice, engineering lung cancer cells to make extra NSUN2 led to larger, more aggressive tumors and more metastatic spots in the lungs, while dialing down YAP blunted these effects. This showed that NSUN2’s impact on tumor growth depends heavily on its ability to raise YAP levels.
A relay team that protects cancer messages
NSUN2 does not act alone. Once it places m5C marks on YAP RNA, two RNA-binding proteins, ALYREF and YBX1, recognize and latch onto the modified message. The study reveals a relay: ALYREF first spots the marked RNA, then passes it to YBX1, and together they form a three-part complex with NSUN2. This complex shields YAP RNA from AGO2, a protein that normally helps degrade unwanted messages, and also helps recruit the translation machinery that turns RNA into protein. The result is that YAP RNA sticks around longer and is translated more efficiently, flooding the cell with YAP protein. Downstream, YAP switches on genes that drive cell-cycle progression, invasion, and tissue-remodeling enzymes, all hallmarks of aggressive cancer.
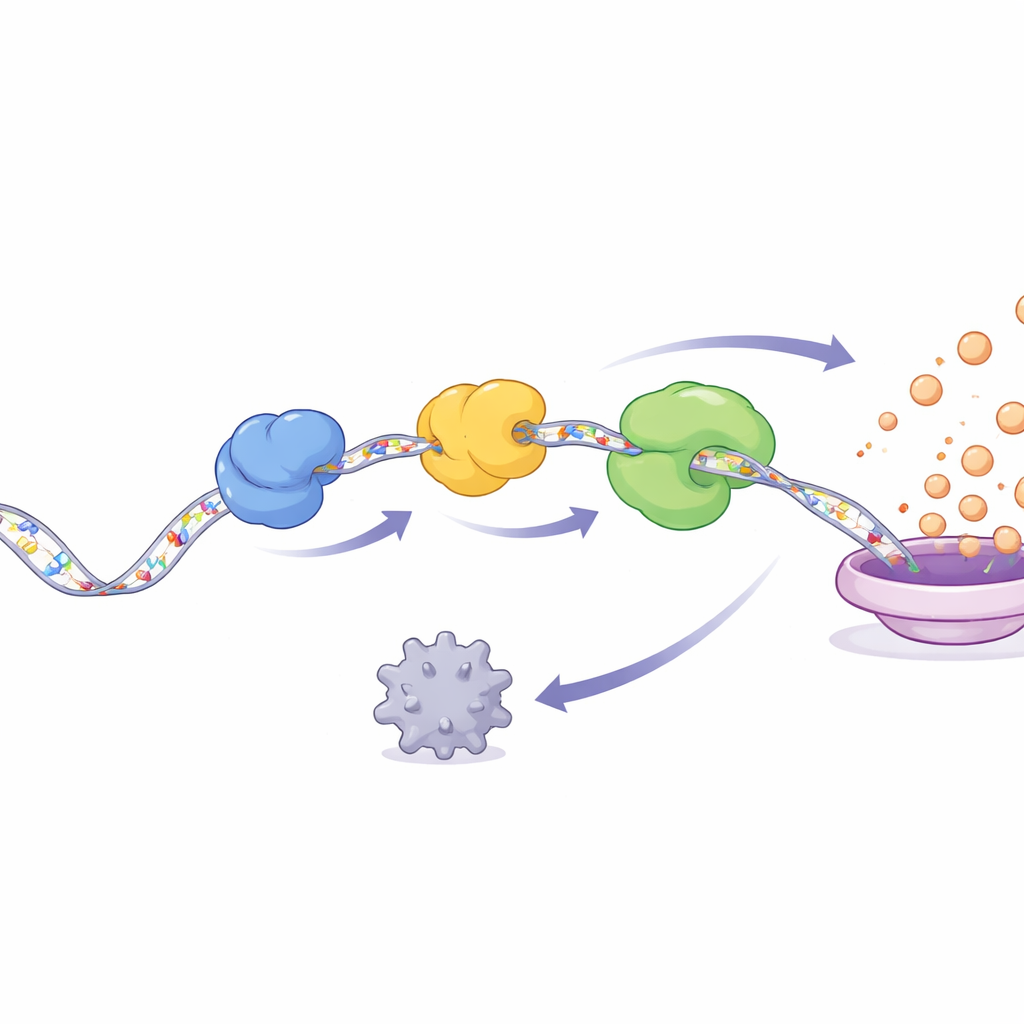
A self-reinforcing cancer loop
Strikingly, the relationship runs both ways. YAP partners with a DNA-binding protein called TEAD2 to turn on many genes, and the team found that NSUN2 is one of them. In lung cancer cells, active YAP–TEAD2 binds directly to the NSUN2 promoter region in DNA and cranks up NSUN2 production. That, in turn, increases m5C marks on YAP RNA, further stabilizing and amplifying YAP itself. This creates a positive feedback loop: more YAP leads to more NSUN2, which leads to even more YAP, locking cells into a high-growth, pro-metastatic state. Disrupting this loop, the researchers argue, could be a powerful way to slow NSCLC progression.
New ways to target lung tumors
Because enzymes and RNA-binding proteins can be targeted by small molecules, the team tested experimental inhibitors that interfere with NSUN2’s catalytic activity and lower YBX1 levels. In lung cancer cell lines, each compound reduced YAP protein, slowed cell growth, curtailed movement and invasion, and nudged cells back toward a less invasive state. In mouse models, using both drugs together shrank tumors more than either alone and cut down on metastatic lesions in the lungs. Importantly, these agents did little harm to normal lung cells in culture, hinting at a potential therapeutic window.
What this could mean for patients
For a non-specialist, the key message is that this study uncovers a specific molecular circuit that lung cancer cells use to amplify a central growth signal. NSUN2 chemically tags the YAP message, ALYREF and YBX1 protect and translate it, and YAP in turn boosts NSUN2, creating a runaway loop that fuels tumor growth and spread. By showing that blocking NSUN2’s marking function and YBX1’s support role can break this loop in cells and in mice, the work lays groundwork for future drugs that act at the RNA level rather than directly on DNA or proteins alone. While these compounds are still at a preclinical stage, the concept of targeting m5C marks on cancer-promoting RNAs offers a fresh angle in the fight against NSCLC.
Citation: Li, R., Jin, D., Shao, S. et al. NSUN2 promoted tumor growth and metastatic via m5C-regulation of YAP through ALYREF/YBX1 axis in NSCLC. Cell Death Dis 17, 299 (2026). https://doi.org/10.1038/s41419-025-08353-x
Keywords: non-small cell lung cancer, RNA modification, YAP signaling, NSUN2, metastasis