Clear Sky Science · en
A phase 2 trial of burosumab for treatment of fibroblast growth factor-23-mediated hypophosphatemia in children and adults with fibrous dysplasia
Why This Matters for Patients and Families
Fibrous dysplasia is a rare bone disorder that can cause painful fractures, bowed limbs, and loss of mobility, often beginning in childhood. Many people with this condition also lose too much phosphate, a mineral that bones need to stay strong. Standard treatments to replace phosphate can be hard to tolerate and often do not fully fix the problem. This study tested a targeted medicine called burosumab in children and adults with severe fibrous dysplasia to see whether it could safely restore phosphate levels, support bone health, and improve everyday function.
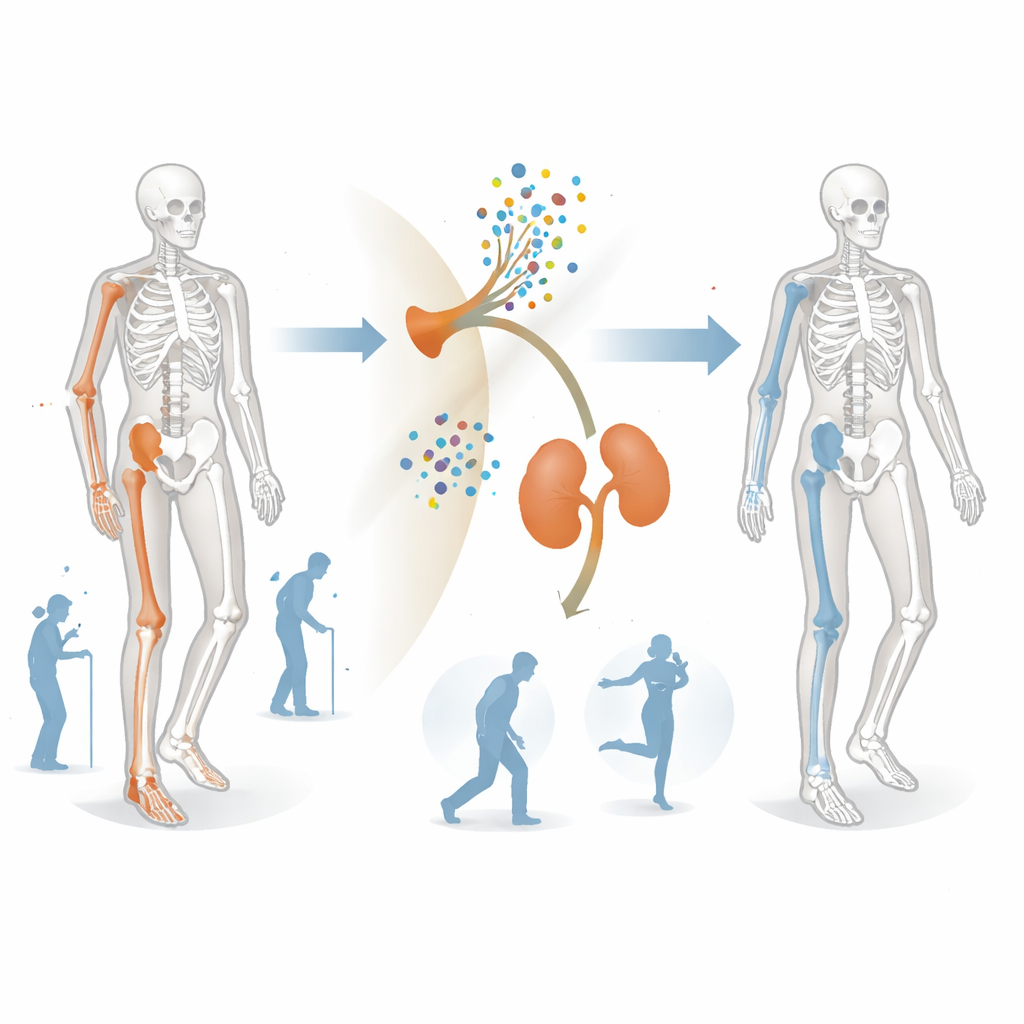
A Rare Bone Disease with Heavy Daily Burden
In fibrous dysplasia, normal bone is partly replaced by soft, fibrous tissue, leaving affected bones weak, easily fractured, and prone to bending out of shape. Some patients also have skin spots and hormone problems, a combination known as McCune–Albright syndrome. Many participants in this trial had a very high amount of their skeleton involved and already lived with major disability: two‑thirds needed devices such as wheelchairs, walkers, or crutches. On top of the faulty bone, their bodies produced excess amounts of a hormone that causes the kidneys to spill phosphate into the urine, leading to low phosphate in the blood and further weakening the skeleton.
Limitations of Older Treatments
Until now, doctors have tried to correct low phosphate in fibrous dysplasia with large doses of oral phosphate and activated vitamin D. These medicines can upset the stomach, stress the kidneys, and trigger the body to make even more of the phosphate‑wasting hormone, trapping patients in a difficult cycle. Even with treatment, many people cannot reach truly normal phosphate levels and continue to fracture, need surgery, or lose mobility. Earlier work suggested that patients with phosphate in the middle to upper part of the normal range have fewer bone complications, but it was unknown whether aiming that high would be both safe and effective.
A Targeted Antibody as a New Approach
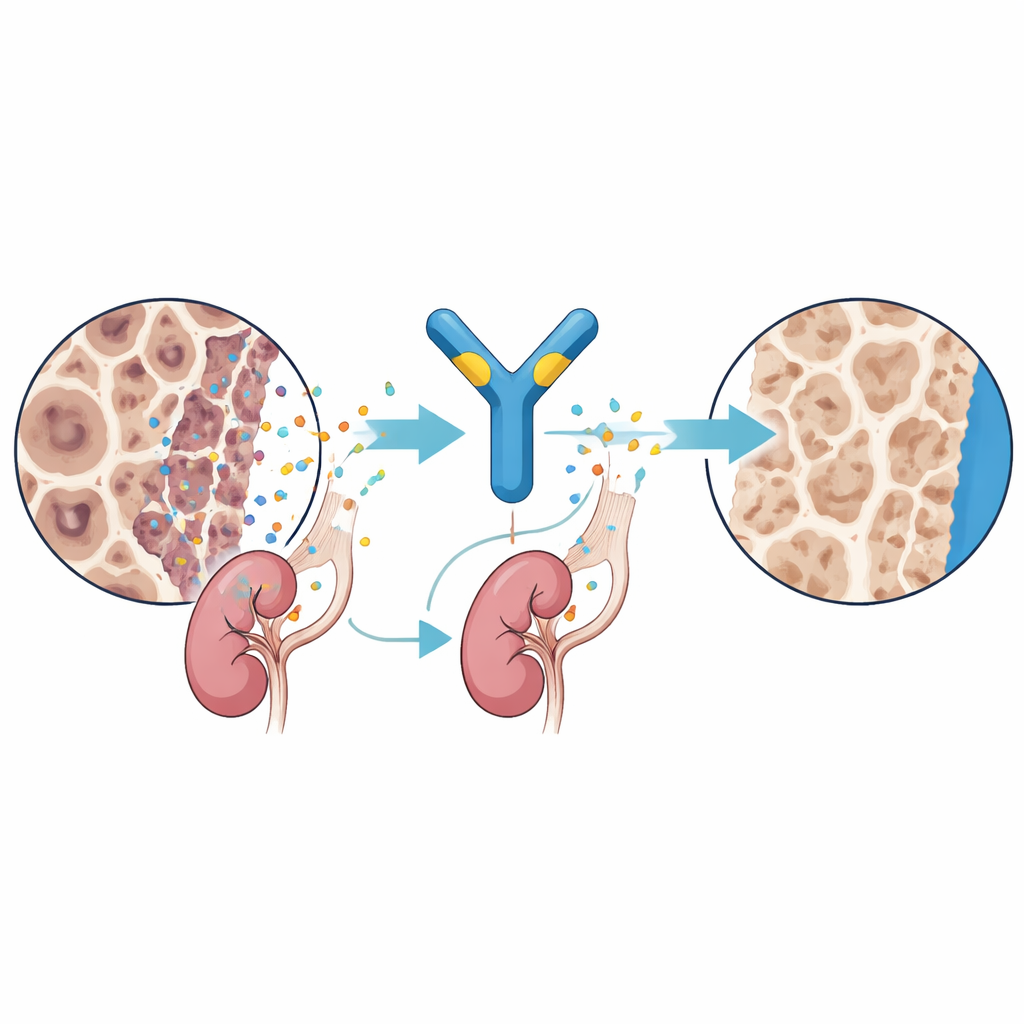
Burosumab is a laboratory‑made antibody that binds to the phosphate‑wasting hormone and blocks its effect, allowing the kidneys to hold onto more phosphate. It is already approved for other rare conditions caused by the same hormone, but had not been systematically tested in fibrous dysplasia. In this phase 2 study, 12 participants (7 children and 5 adults) stopped their old phosphate and vitamin D pills and received burosumab injections for 48 weeks. Doses were adjusted over time to place each person’s phosphate level in the mid‑to‑high normal range for their age and sex, with frequent checks of blood, urine, imaging, and symptoms.
What Changed with Treatment
By the end of the study, every participant reached the target phosphate range. On average, their phosphate status moved from clearly low to solidly normal. Other lab measures tied to mineral balance improved as well, and a key marker of bone turnover, alkaline phosphatase, dropped by nearly half, suggesting less stressed, better‑mineralized bone. In children, questionnaires showed steady trends toward less pain, less fatigue, and better mobility, even though the study was too small to prove statistical changes in all scores. Two girls with extremely limited mobility experienced striking gains: one went from full‑time wheelchair use to walking independently, and another progressed from being unable to bear weight to walking with a walker for short distances. In adults, benefits were more modest and less consistent, likely reflecting long‑standing damage that is harder to reverse.
Safety and Effects on Bone Lesions
Side effects were generally mild. Some participants had episodes of high phosphate, but these were small, temporary rises that resolved when doctors lowered the burosumab dose according to the study plan. Injection‑site reactions were mild and did not lead anyone to stop treatment. Importantly, detailed bone scans and biopsies of the abnormal tissue showed no signs that burosumab made the lesions grow faster or become more aggressive. Kidney function and calcium levels stayed stable, and no one developed new kidney calcifications.
What This Means Going Forward
For people with fibrous dysplasia complicated by phosphate loss, this study shows that burosumab can reliably restore phosphate to a healthier range, improve laboratory signs of bone health, and may offer meaningful functional gains, especially when started in severely affected children. While the trial was small and had no comparison group, the combination of better chemistry, encouraging mobility changes, and a reassuring safety profile suggests that burosumab could become a preferred tool to limit disability in this challenging disease. Larger and longer studies will be needed, but for many families, this targeted medicine offers a hopeful new option beyond traditional phosphate pills.
Citation: de Jong, O., Gun, Z.H., Asante-Otoo, A. et al. A phase 2 trial of burosumab for treatment of fibroblast growth factor-23-mediated hypophosphatemia in children and adults with fibrous dysplasia. Bone Res 14, 47 (2026). https://doi.org/10.1038/s41413-026-00523-7
Keywords: fibrous dysplasia, burosumab, hypophosphatemia, rare bone disease, FGF23