Clear Sky Science · en
A randomized trial to evaluate attitudes regarding pharmacogenomics among pregnant and pediatric populations: design and baseline characteristics
Why Your Genes Matter When You Take Medicine
When a doctor prescribes a drug, they often rely on trial and error to find the right medicine and dose. But our genes can influence how well a drug works and whether it causes side effects. This idea—using DNA to help choose medicines—is called pharmacogenomics. The study described here asks a simple but important question: how do pregnant people and families of children feel about this kind of testing, and what helps them understand and value it?
Personalized Medicine for Parents and Kids
The researchers focused on two groups who are usually left out of genetic drug studies: pregnant people and children with long‑term health conditions. These patients often take medicines that could be guided by genetic information, yet most of the existing research is based on non‑pregnant adults. Pregnancy and childhood bring major changes in how the body processes drugs, so simply borrowing advice from adult studies may not be safe or accurate. By learning what these groups know and feel about pharmacogenomic testing, the team hopes to make future research and clinical care more inclusive.
How the Study Was Set Up
The project, run at Vanderbilt University Medical Center, enrolled over 400 pregnant participants and more than 350 children along with their parents or guardians. Everyone filled out an online survey about their background, health, and experiences with medicines, as well as their understanding of basic genetic ideas like DNA and genes. They were then randomly assigned to one of two paths. All participants were offered a blood test that looked at specific genetic differences known to affect how certain drugs are handled by the body. Everyone who completed testing received results along with easy‑to‑understand explanations, and their doctors could also see these results in the medical record.
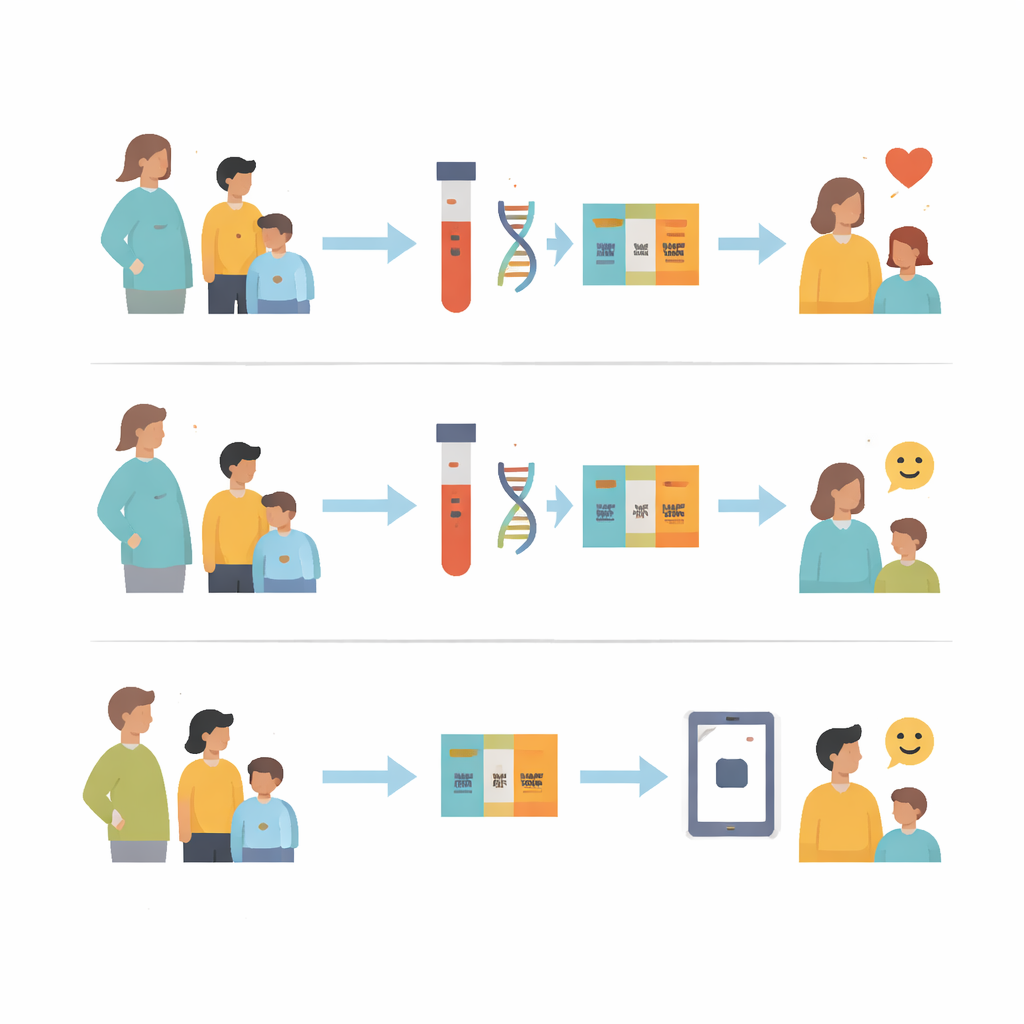
Testing, Education, and Attitudes
The key twist in the design was an educational video about pharmacogenomic testing. One group received the video at the same time as their test results, while the other group got the results first and the video later. After each step, participants were asked again about their knowledge and views. Before any education, most people in both groups already believed that genes affect how medicines work. They were familiar with basic genetic terms but much less comfortable with specialized ideas like “metabolizer status” or “precision medicine.” Once the researchers briefly defined pharmacogenomic testing as using DNA differences to pick the right medicine or dose, more than nine out of ten participants felt it could help them and their doctors choose safer and more effective treatments.
Who Followed Through With Testing
Not everyone who enrolled went on to complete the genetic test. In the pregnancy group, about 60 percent followed through. Those who did were more likely to be older, more highly educated, from higher‑income households, and already taking at least one daily medication. Participants who identified as Hispanic, were single, or had public insurance were less likely to complete testing. In the pediatric group, about two‑thirds completed the test. Children who were younger, whose caregivers were single, or who had public insurance were less likely to finish the process. These patterns suggest that social and economic factors, not just interest or curiosity, influence who actually receives genetic‑based care.
What This Means for Future Care
Overall, the study shows that pregnant patients and families of children are interested in using genetic information to guide medications and generally view it as helpful once it is clearly explained. Limited familiarity with technical language appears to be a fixable barrier: a simple definition and user‑friendly materials go a long way. At the same time, the uneven follow‑through with testing warns that without careful planning, pharmacogenomic services could widen existing gaps in care. By mapping out both enthusiasm and obstacles, this trial lays the groundwork for making DNA‑guided prescribing safer, more understandable, and more equitable for pregnant people and children.
Citation: Sundermann, A.C., Marryshow Batson, S.E., Jasper, E.A. et al. A randomized trial to evaluate attitudes regarding pharmacogenomics among pregnant and pediatric populations: design and baseline characteristics. Pharmacogenomics J 26, 16 (2026). https://doi.org/10.1038/s41397-026-00413-5
Keywords: pharmacogenomics, pregnancy, pediatrics, precision medicine, genetic testing