Clear Sky Science · en
Macrophage metabolic reprogramming by vanadium released from glucose-responsive bio-gel accelerates diabetic wound repair
Why stubborn diabetic wounds matter
For many people with diabetes, a small blister or cut on the foot can quietly turn into a chronic, hard-to-heal ulcer that threatens infection, amputation, or even death. These wounds often resist standard dressings and antibiotics because high blood sugar quietly scrambles the local immune system and energy supply. This study explores a new kind of “smart” wound gel that releases a trace metal, vanadium, in response to high glucose. The goal is to gently retune the metabolism of key immune cells so that they switch from fueling inflammation to actively rebuilding tissue.
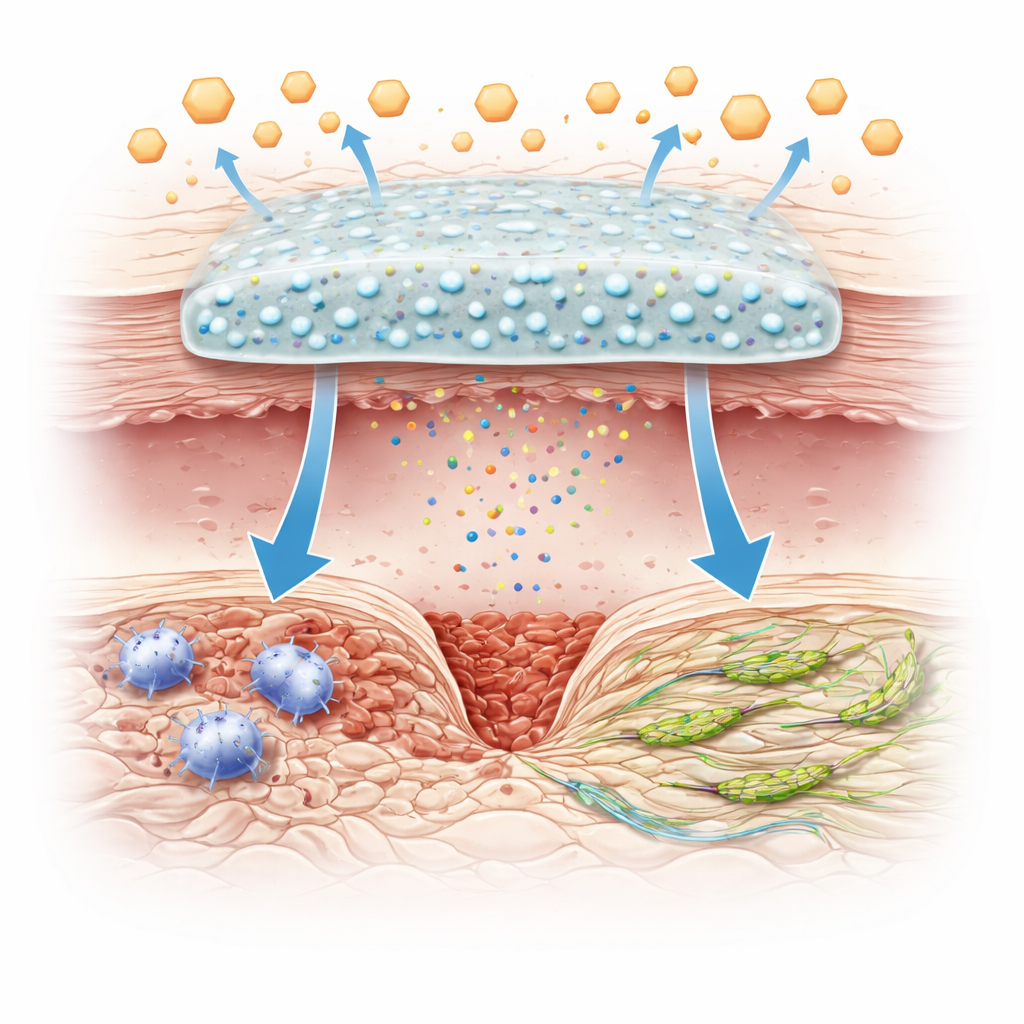
How immune cells get stuck
Macrophages are front-line immune cells that also act as on-site repair crews. Early in wound healing they take on a warrior-like form that attacks germs and stirs up inflammation; later, they are supposed to shift into a nurturing mode that calms inflammation and supports new tissue growth. This shift is tightly linked to how the cells make energy: the pro-inflammatory state relies on fast, sugar-burning chemistry, while the pro-healing state uses slower, oxygen-based energy from mitochondria, the cell’s powerhouses. In diabetic wounds, constant high sugar and poor insulin signaling derail this energy switch, trapping macrophages in the angry, inflammatory mode and keeping the wound in a chronic, nonhealing state.
A smart gel that responds to sugar
To tackle this problem, the researchers created tiny glass-like spheres that contain vanadium, built from calcium, silicon, phosphorus, and vanadium oxides. These spheres were then embedded into a soft hydrogel made from modified gelatin and a chitosan-based component that swells in response to glucose. Under high-sugar conditions like those in a diabetic wound, the gel takes up more water, its internal network loosens, and vanadium-containing particles and ions are released in a controlled way. Laboratory tests showed that loading the gel with these spheres did not harm its structure, but strengthened its mechanical properties and allowed a steady, glucose-triggered release of vanadium.
Speeding up repair in diabetic mice
When the gel was placed on full-thickness skin wounds in diabetic mice, healing improved dramatically compared with untreated wounds or gels without vanadium. The vanadium-loaded dressing led to faster closure of the wound area, better re-formation of the outer skin layers, and more orderly collagen deposition, which are all hallmarks of healthy repair. Chemical profiling of the tissue showed that the treatment reduced the build-up of acidic by-products and other metabolic waste linked to diabetes. At the same time, signals of excessive inflammation, such as the molecules TNF-α and IL-6, dropped, while anti-inflammatory signals and new blood vessel growth increased. Staining for macrophage markers revealed more cells in the pro-healing state and fewer in the pro-inflammatory state at early time points, indicating that the gel actively reshaped the immune environment, not just covered the wound.
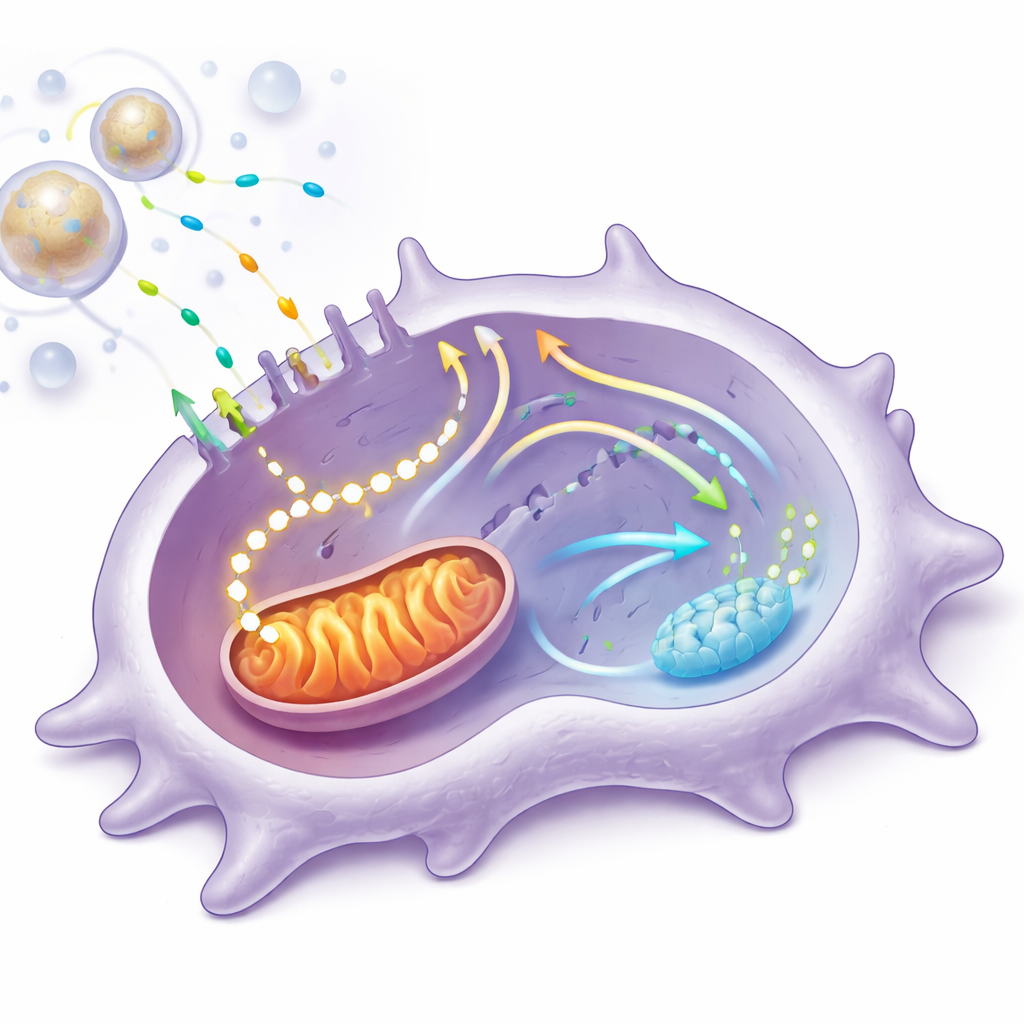
Rewiring cellular energy use
Digging deeper, the team isolated macrophages and exposed them to vanadium-releasing extracts under high-glucose, inflammatory conditions. These cells shifted their behavior toward a healing phenotype: genes and proteins tied to inflammation went down, while those associated with tissue repair went up. Advanced metabolic testing showed that vanadium nudged the cells to take up more glucose and channel it into mitochondrial respiration rather than simple sugar fermentation. Oxygen consumption and ATP output from mitochondria rose, and the internal structure of the mitochondria became larger and more interconnected—signs of a stronger, more efficient energy system. Blocking the entry of sugar-derived fuel into mitochondria largely erased these benefits, showing that the extra mitochondrial energy was essential for the macrophage’s personality change.
A signaling pathway that links sugar, energy, and healing
The study further mapped the signaling cascade inside macrophages. Vanadium boosted activity of the insulin receptor on the cell surface and switched on the downstream PI3K–AKT pathway, which is known to regulate glucose handling. This in turn increased the presence of a glucose transporter (GLUT4) on the cell membrane, allowing more sugar to flow in. Enzymes of the cell’s central energy cycle became more active, and levels of citrate and its derivative acetyl-CoA rose, feeding not only energy production but also subtle chemical changes to the cell’s DNA-packaging proteins. One particular histone modification, linked to turning on healing-related genes, increased. When the researchers chemically blocked the insulin receptor, PI3K, or GLUT4, both the metabolic boost and the macrophage’s shift into a repair mode were sharply reduced. Together, these results show that vanadium works as a localized insulin-mimicking signal that restores a broken link between high glucose, efficient mitochondrial energy use, and pro-healing immune behavior.
What this could mean for patients
This work suggests that tackling diabetic wounds may require more than simply adding growth factors or cells on top of a metabolically hostile environment. By designing a glucose-responsive gel that delivers vanadium exactly where sugar is high, the researchers were able to reprogram the wound’s immune cells from the inside out, calming chronic inflammation and promoting regeneration in mice. While long-term safety studies are still needed—especially to monitor potential vanadium build-up—the approach offers a promising, cell-free strategy that aligns with existing clinical practice of applying topical dressings. If translated successfully to humans, such smart materials could turn stubborn diabetic ulcers into wounds that finally remember how to heal.
Citation: Li, J., Li, Z., Han, L. et al. Macrophage metabolic reprogramming by vanadium released from glucose-responsive bio-gel accelerates diabetic wound repair. Sig Transduct Target Ther 11, 148 (2026). https://doi.org/10.1038/s41392-026-02647-y
Keywords: diabetic wound healing, macrophage polarization, vanadium biomaterials, glucose-responsive hydrogel, immune metabolism