Clear Sky Science · en
Cortisol-resistant CAR-NK cells overcome steroid-induced immunosuppression in lung cancer
Why Stress Hormones Matter in Lung Cancer
Lung tumors do not simply grow on their own; they also reshape their surroundings to keep the body’s defenses at bay. This study reveals that lung cancers create a pocket rich in cortisol—the body’s main stress hormone—which weakens natural killer (NK) cells, a frontline immune force against cancer. The researchers then engineer a new type of immune cell therapy that can ignore cortisol’s brake signals, restoring its ability to attack lung tumors even when patients need steroid drugs.
A Hidden Hormone Cloud Around Tumors
The team began by carefully measuring many different steroid molecules in pieces of human lung tumors. They found that cortisol was the most abundant steroid inside these tumors, present at surprisingly high levels compared with other hormones. Using single-cell genetic maps of thousands of cells from lung cancers, they discovered that several cell types help build this hormone-rich niche. Immune cells, cancer cells, and support cells within the tumor can make steroid precursors from cholesterol, while certain fibroblasts and macrophages excel at converting inactive cortisone back into active cortisol. Together, they create a continuous local supply of this potent stress hormone right where immune cells are trying to fight.
How Cortisol Silences Natural Killers
Natural killer cells are designed to recognize and destroy abnormal cells quickly, including many cancer cells. Yet inside lung tumors, NK cells often look exhausted and ineffective. By exposing human NK cells to cortisol and studying changes in gene activity, the researchers showed that cortisol pushes these cells toward a sluggish, less aggressive state. Important genes linked to cell killing and movement were turned down, while genes linked to inhibition and stress were turned up. When they examined NK cells taken directly from lung tumors, the ones carrying higher levels of the cortisol receptor displayed weaker attack programs and stronger “dysfunction” and hypoxia (low-oxygen stress) signatures. Across many immune and support cell types in lung cancer, higher cortisol responses went hand in hand with stronger hypoxia signals, suggesting that stress hormone and oxygen deprivation work together to blunt immunity.
Blocking the Hormone Signal Reawakens Immunity
To test whether cutting off cortisol signaling could restore tumor-fighting power, the scientists used mouse models of lung cancer and treated them with a drug that blocks the cortisol receptor, known as the glucocorticoid receptor. Mice receiving this blocker developed smaller tumors than untreated animals. Their tumor-invading NK cells produced more killing molecules and activation markers, while inhibitory receptors and markers of hypoxic stress went down. T cells inside the tumors were also more active, but immune cells in the spleen remained largely unchanged, indicating that the drug mainly lifted suppression inside the tumor microenvironment rather than triggering widespread immune activation. In a lung metastasis model, blocking the receptor similarly reduced the number and size of tumor spots in the lungs.
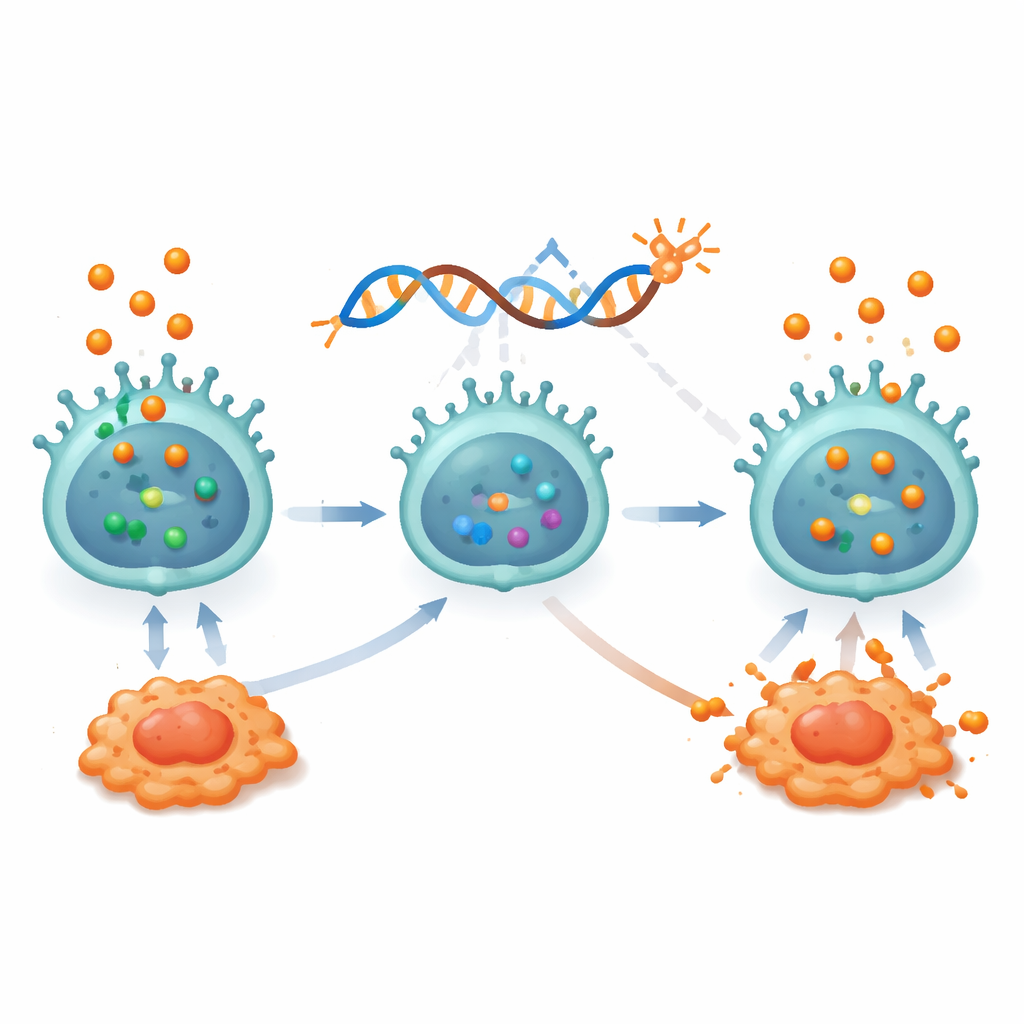
Designing Cortisol-Resistant Cancer Killers
Building on these insights, the researchers engineered a form of immunotherapy that could thrive in a steroid-rich setting. They created NK cells bearing a chimeric antigen receptor (CAR) that recognizes CEACAM5, a protein frequently found at high levels on lung cancer cells. These CAR-NK cells effectively killed CEACAM5-positive lung cancer lines in the lab, but their performance dropped sharply when cortisol was added: they killed fewer tumor cells, made fewer helpful cytokines, and began to display exhaustion markers. To overcome this, the team used gene-editing tools to delete the gene for the cortisol receptor, NR3C1, creating cortisol-resistant CAR-NK cells. These modified cells maintained strong killing, stayed activated, and avoided exhaustion even when bathed in cortisol or placed under hypoxia-like conditions. Detailed pathway analyses showed that, unlike standard CAR-NK cells, the edited cells preserved key internal signaling routes—especially PI3K–AKT and NF-κB—that drive NK-cell activation and survival.
Stronger Tumor Control in Steroid-Rich Lungs
Finally, the researchers tested both standard and cortisol-resistant CAR-NK cells in mice carrying lung metastases of human lung cancer. To mimic real clinical practice, all mice also received a synthetic steroid drug, modeling the situation in patients who need glucocorticoids for symptom control. Under these conditions, only the cortisol-resistant CAR-NK cells achieved robust and lasting tumor control, as shown by imaging and tissue examination; tumors were smaller and less widespread than in animals treated with regular CAR-NK cells. Importantly, the engineered cells did not cause noticeable tissue damage or high levels of inflammatory cytokines in the blood, suggesting a favorable safety profile.
What This Means for Future Cancer Care
This work identifies local cortisol signaling as a major, previously underappreciated roadblock to immune attack in lung cancer. Lung tumors exploit both hormone production and recycling to maintain a cortisol-rich, low-oxygen niche that saps NK cells of their killing power. By removing the cortisol receptor from CAR-NK cells, the researchers created an immune therapy that stays active even when steroids are present and the tumor environment is harsh. In the future, such cortisol-resistant cell therapies could allow doctors to combine life-saving immunotherapy with necessary steroid treatments, potentially improving outcomes for patients with lung and other steroid-producing solid tumors.
Citation: Chakraborty, S., Pramanik, J., Alviter-Raymundo, G. et al. Cortisol-resistant CAR-NK cells overcome steroid-induced immunosuppression in lung cancer. Sig Transduct Target Ther 11, 128 (2026). https://doi.org/10.1038/s41392-026-02638-z
Keywords: lung cancer immunotherapy, natural killer cells, cortisol and tumors, CAR-NK cell therapy, tumor microenvironment