Clear Sky Science · en
Cardiovascular ageing: hallmarks, signaling pathways, diseases and therapeutic targets
Why Our Hearts Age Before We Feel Old
Many people imagine aging as gray hair and slower steps, but deep inside the body another, quieter kind of aging is unfolding. Our hearts and blood vessels gradually stiffen and lose flexibility, and this hidden process turns into the single biggest driver of heart attacks, strokes, and heart failure worldwide. This review article brings together decades of research to explain why the cardiovascular system ages, how that aging helps trigger many major diseases, and what science is beginning to do about it—from lifestyle changes to experimental “rejuvenation” therapies.
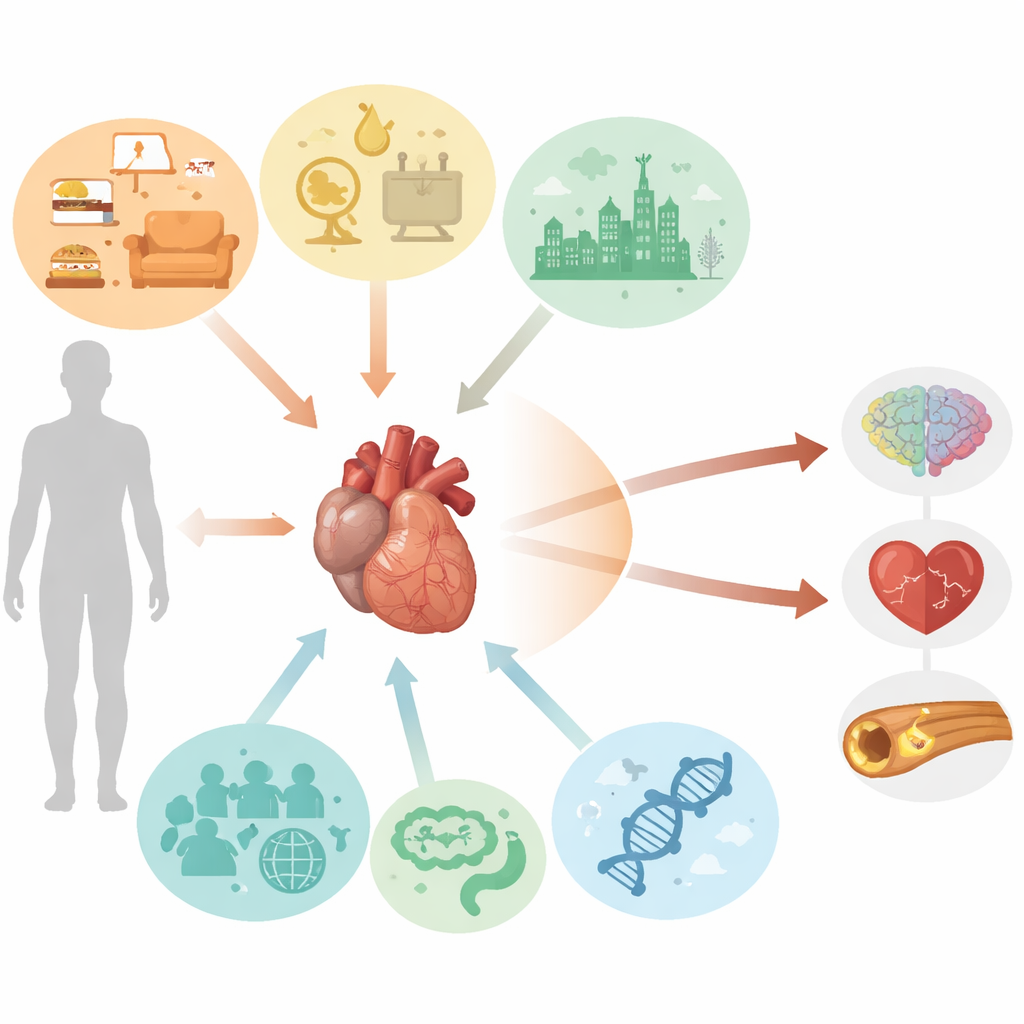
Everyday Influences That Age the Heart
The authors first describe cardiovascular aging as more than just the passage of time. Five broad categories of influences weave together to speed or slow the process: lifestyle habits, metabolic and hormonal balance, environmental exposures, genetic and epigenetic makeup, and features of our biology and social world. Smoking, inactivity, excess calories, and chronic stress hasten damage to blood vessels. Obesity, high blood pressure, diabetes, kidney disease, and metabolic syndrome create a biochemical environment that batters vessel walls. Air pollution, heavy metals, radiation, certain chemotherapy drugs, and even spaceflight can all push the cardiovascular system toward premature wear. At the same time, inherited gene variants, age-related shifts in how genes are switched on or off, gut microbes, chronic viral infections, sex hormones, and ethnic and social disparities further shape who ages faster, and how.
What an Aging Heart and Arteries Look Like Inside
With advancing age, the heart and blood vessels undergo striking structural and functional changes. The heart muscle accumulates scar-like fibrosis and abnormal protein deposits, its chambers enlarge or thicken, and its ability to relax and pump efficiently declines. Small coronary vessels lose elasticity and density, limiting blood flow when demand rises. In the arteries, the once-supple walls become thicker, stiffer, and more calcified. The inner lining cells that normally release vessel-relaxing signals become fewer and more dysfunctional, while muscle cells in the wall change identity, moving away from contraction and toward growth and scarring. These changes erode the cardiovascular system’s ability to adapt to stress, setting the stage for common late-life problems such as atrial fibrillation, heart failure with preserved ejection fraction, and difficult-to-treat high blood pressure.
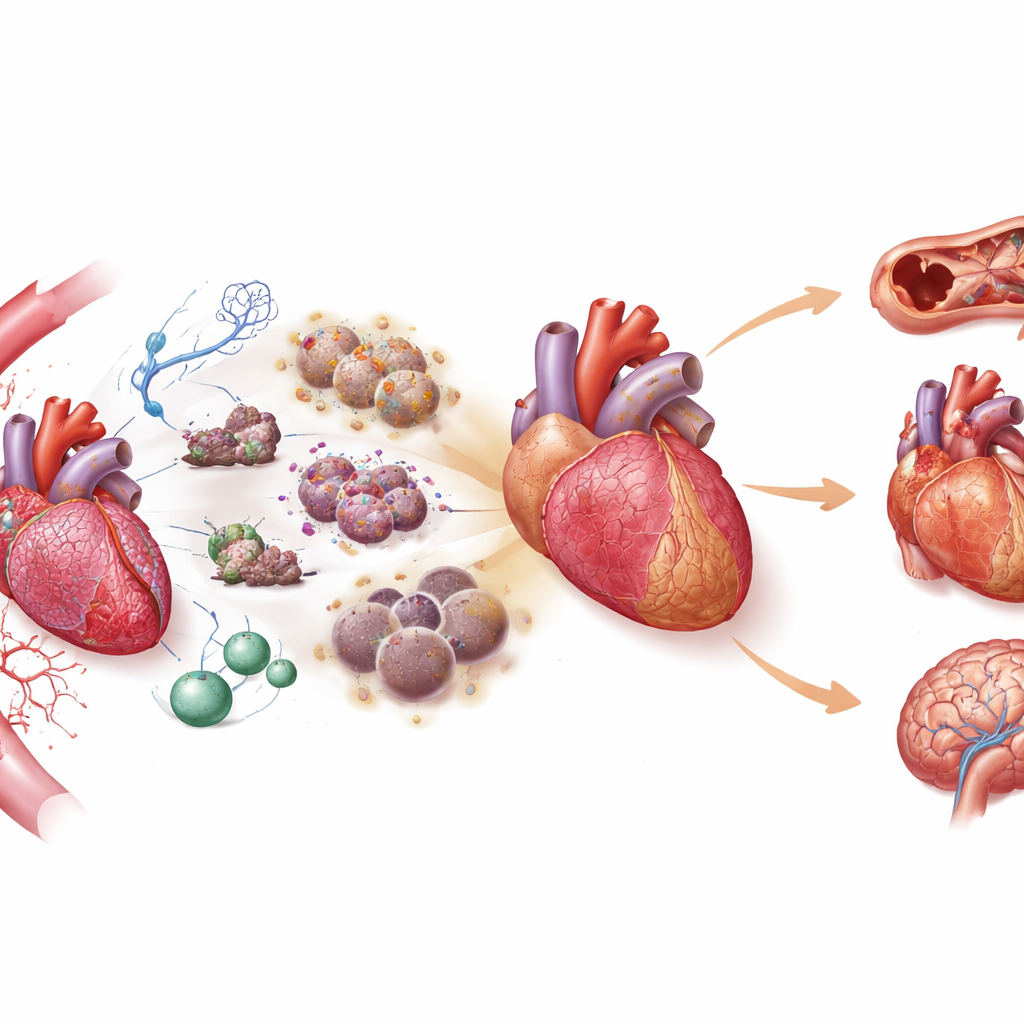
The Inner Hallmarks of Cardiovascular Aging
To make sense of the complexity, the paper organizes twelve “hallmarks” of cardiovascular aging into three levels: molecular, cellular, and systemic. At the molecular level, DNA damage, epigenetic drift, failures in protein quality control, faltering mitochondria, and chronic low-grade inflammation slowly undermine cell health. At the cellular level, key players such as heart muscle cells, vessel-lining cells, immune cells, and stem cells lose their ability to repair, recycle, and renew; many enter a state called senescence, in which they no longer divide but release inflammatory and tissue-remodeling substances. At the systemic level, major signaling systems that normally help the body respond to stress—such as the hormone network that regulates blood pressure and salt balance, the “fight-or-flight” stress response, growth-factor pathways, and the way cells sense mechanical forces—become chronically overactivated or mis-tuned. These hallmarks constantly interact, so a disturbance in one, like mitochondrial damage, can ripple outward to amplify inflammation, senescence, and tissue stiffening.
How an Old Heart Drives Many Different Diseases
Cardiovascular aging does not stay confined to the chest. It forms a common soil for atherosclerosis, aneurysms, and heart failure, and it also feeds into disorders far from the heart. Senescent and dysfunctional vessel cells encourage fatty plaques, weaken the aortic wall, and make plaques more fragile. Loss of protein quality control and mitochondrial health in the heart muscle encourages heart failure. In the brain, stiff arteries, leaky barrier vessels, and sluggish fluid clearance systems contribute to cognitive decline and neurodegenerative diseases such as Alzheimer’s and Parkinson’s. In diabetes, aged endothelial cells worsen microvascular complications, while high blood sugar in turn accelerates vascular aging, creating a vicious circle. Similar feedback loops likely link obesity and disordered blood fats with progressive damage to vessels and organs.
Can We Turn Back the Cardiovascular Clock?
The review highlights a growing toolbox of potential “rejuvenation” strategies. Some are familiar: quitting smoking, improving diet quality, maintaining healthy weight, and regular physical activity all slow vascular stiffening and improve heart performance at any age. Others are more experimental. Drugs that eliminate or reprogram senescent cells, compounds that tweak energy-sensing pathways like mTOR, AMPK, and sirtuins, and targeted anti-inflammatory therapies are being tested in animals and early human trials. Researchers are also exploring neuro–heart connections, showing that sleep, stress, and mental health directly affect vascular aging, as well as stem-cell and extracellular-vesicle approaches to repair damaged vessels. At the same time, new “aging clocks” based on DNA marks or blood proteins aim to measure how old a person’s cardiovascular system really is, and to track whether interventions truly slow or reverse its decline.
What This Means for Healthy Aging
The central message for lay readers is that cardiovascular aging is not a simple, automatic countdown tied only to birthdays. It is a dynamic process driven by interacting molecular damage, cellular wear, and system-wide miscommunication—yet many of its levers are modifiable. By understanding the hallmarks and triggers of cardiovascular aging, scientists are beginning to design therapies that could delay, blunt, or sometimes partially reverse the deterioration that leads to heart attacks, strokes, heart failure, and even some forms of dementia. While much work remains, the review argues that protecting the “age” of our arteries and heart may be one of the most powerful ways to extend not just life span, but years of healthy, independent living.
Citation: Zheng, P., Yan, W., Ding, Y. et al. Cardiovascular ageing: hallmarks, signaling pathways, diseases and therapeutic targets. Sig Transduct Target Ther 11, 142 (2026). https://doi.org/10.1038/s41392-026-02630-7
Keywords: cardiovascular aging, vascular health, cellular senescence, heart failure, atherosclerosis