Clear Sky Science · en
Distinct metabolomic and proteomic signatures in Parkinson’s disease patients with REM sleep behavior disorder
Why Sleep and the Gut Matter in Parkinson’s
Many people think of Parkinson’s disease mainly as a movement disorder, marked by tremor and stiffness. But long before these symptoms appear, subtle changes in sleep and digestion may already be under way. This study explores why people with Parkinson’s who also act out their dreams during deep sleep—a condition called REM sleep behavior disorder—often have worse symptoms and faster disease progression. By looking carefully at molecules in the blood and microbes in the gut, the researchers uncover how changes in metabolism and gut bacteria may help drive this more aggressive form of Parkinson’s.
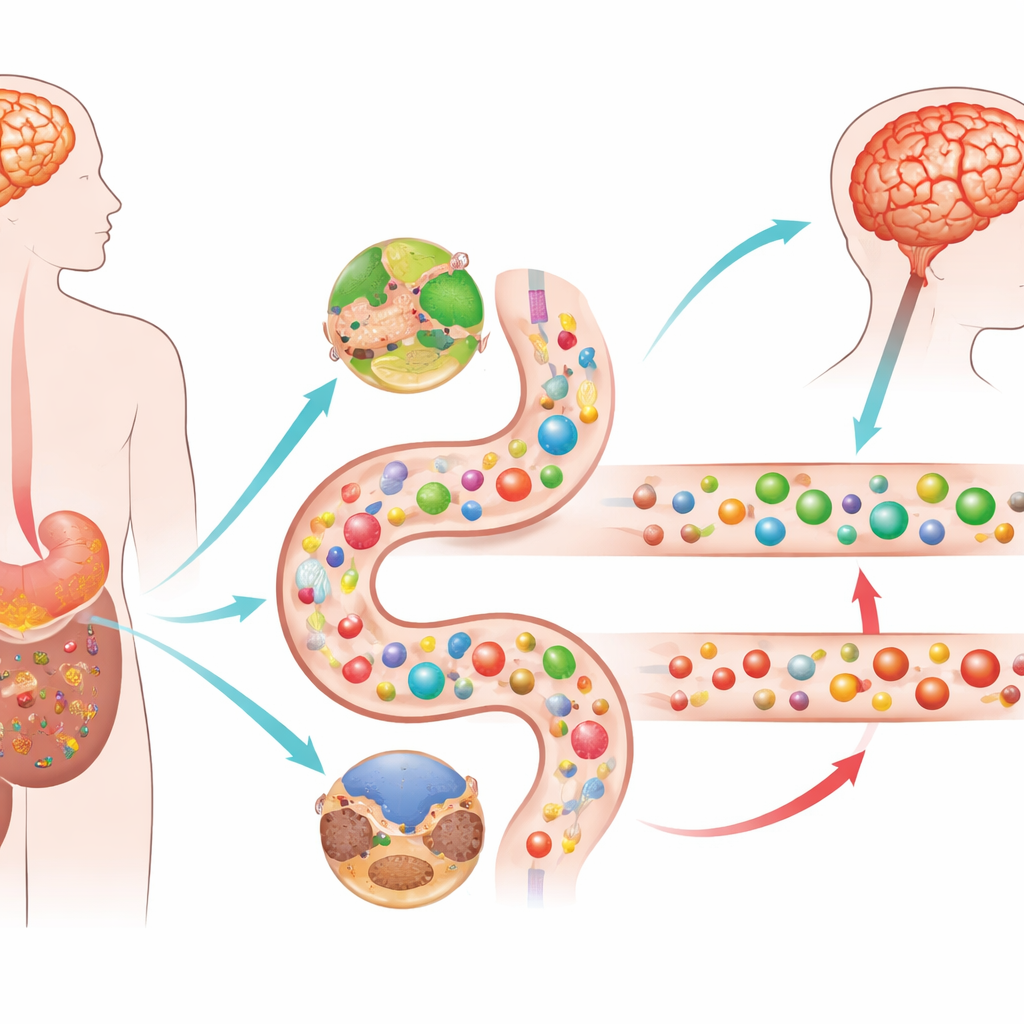
Two Faces of the Same Disease
The researchers studied over 400 participants, including people with Parkinson’s with and without dream-enacting sleep behavior, people with isolated REM sleep behavior disorder but no movement symptoms yet, and healthy volunteers. They measured hundreds of small molecules (metabolites and fats) and inflammation-related proteins in blood samples. Both Parkinson’s groups, regardless of sleep problems, showed broad shifts in how their bodies process energy. Chemicals from the cell’s main power-generating cycle built up, while many carbohydrates and healthy fats declined. At the same time, immune and inflammatory signaling in the blood was clearly ramped up, pointing to a body-wide stress response, not just a problem in the brain.
Energy, Waste, and Fats Out of Balance
The team found signs that brain and body cells in Parkinson’s are relying more on quick, less efficient sugar-burning and less on their usual energy cycle inside mitochondria. This shift leaves behind excess byproducts, such as lactic acid and certain organic acids, which can feed into a vicious circle of oxidative stress and cell damage. The urea cycle, which normally helps clear ammonia and keep brain chemistry stable, was also disturbed, potentially adding to nerve cell strain. At the same time, many complex fats that form cell membranes were reduced, while a particular group of breakdown products called lysophospholipids was increased. These lipid changes may weaken cell membranes and interfere with cellular cleanup systems that normally prevent the build-up of toxic proteins like alpha-synuclein.
When the Gut Turns Proteins into Poisons
The clearest distinction emerged when the researchers compared Parkinson’s patients with and without the dream-enacting sleep behavior. Those with the sleep disorder carried strikingly higher levels of several molecules made by gut bacteria when they ferment dietary proteins and certain amino acids. These include p-cresol and related compounds, as well as phenylacetylglutamine, which have been linked to oxidative stress, damage to blood vessels, and inflammation. People with isolated REM sleep behavior disorder—considered an early warning state for Parkinson’s—showed a similar pattern, suggesting that this toxic “chemical signature” appears before classic motor symptoms. Metagenomic analysis of stool samples supported this picture: in these individuals, gut microbes were geared less toward breaking down dietary fiber and more toward digesting protein and the mucus lining of the gut, favoring production of these harmful metabolites.
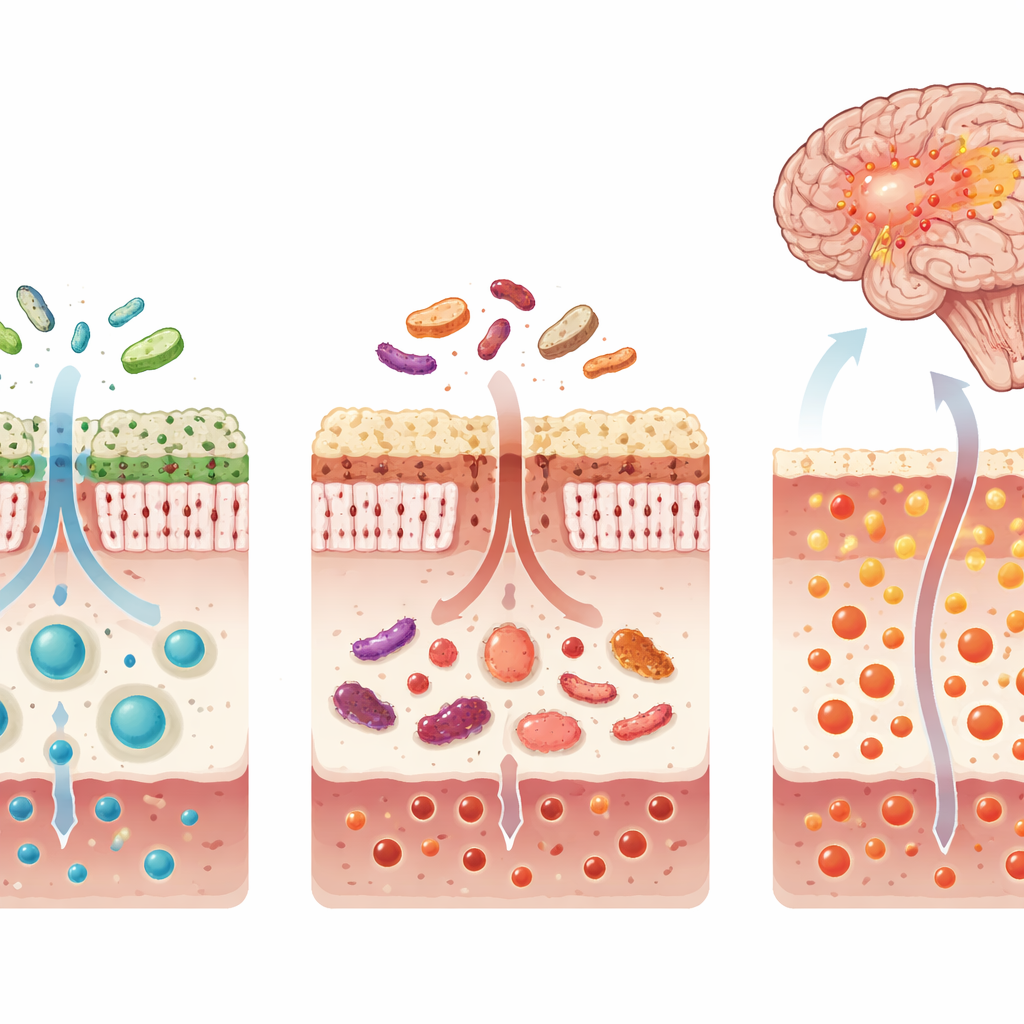
From Gut Chemistry to Brain Inflammation
Many of the gut-derived toxins found at higher levels in the aggressive sleep-related subtype of Parkinson’s were tightly linked to blood proteins involved in immune activation, oxidative stress, and intestinal inflammation. At the same time, building blocks for the key antioxidant glutathione—especially the amino acid glycine—were reduced, and byproducts that signal impaired antioxidant production were increased. Together, this suggests a double hit: more toxic compounds entering the bloodstream from the gut and a weakened internal defense system in the brain and body. Using these blood molecules, the researchers built a nine-metabolite panel that could reliably distinguish Parkinson’s patients with REM sleep behavior disorder from those without it in two independent groups of patients.
What This Means for Patients and the Future
To a non-specialist, the message is that Parkinson’s is not just a brain disease, and not all Parkinson’s is alike. People who act out their dreams in REM sleep appear to follow a “body-first” pathway, in which gut microbes and their chemical products may help trigger or speed up brain damage. The study shows that this pathway leaves a recognizable fingerprint in the blood—shifts in energy use, fat balance, and gut-derived toxins—that can already be seen in people who have the sleep disorder but not yet the movement problems. These insights point toward new possibilities: blood tests to flag high-risk individuals earlier, and therapies that target gut bacteria, diet, or specific metabolic pathways to slow or alter the course of this particularly aggressive form of Parkinson’s.
Citation: Shao, Y., Wang, J., Liu, Y. et al. Distinct metabolomic and proteomic signatures in Parkinson’s disease patients with REM sleep behavior disorder. Sig Transduct Target Ther 11, 115 (2026). https://doi.org/10.1038/s41392-026-02613-8
Keywords: Parkinson’s disease, REM sleep behavior disorder, gut microbiome, metabolomics, neuroinflammation