Clear Sky Science · en
TWIST1 mediated transcriptional activation of SPON2 drives colorectal cancer peritoneal metastasis through stromal cell signaling network
Why this study matters for people with colon cancer
When colon or rectal cancer spreads into the abdominal cavity, coating the intestines and other organs, it becomes one of the most lethal forms of the disease. Standard chemotherapy and even aggressive surgery rarely cure it. This study asks a basic but urgent question: what hidden molecular conversation between tumor cells and their surroundings makes this type of spread so hard to stop—and can that conversation be interrupted?
A deadly spread inside the abdomen
Colorectal cancer that seeds the lining of the abdomen, called peritoneal metastasis, affects up to one third of patients with advanced disease and carries a grim outlook, with five‑year survival near zero. Surgeons can sometimes remove visible tumors and bathe the abdomen in heated chemotherapy, but tiny resistant deposits almost always remain and grow back. The authors argue that to improve outcomes, we must understand not just the tumor cells themselves, but also the unique environment of the peritoneum and how it encourages cancer to grow, spread, and evade treatment.
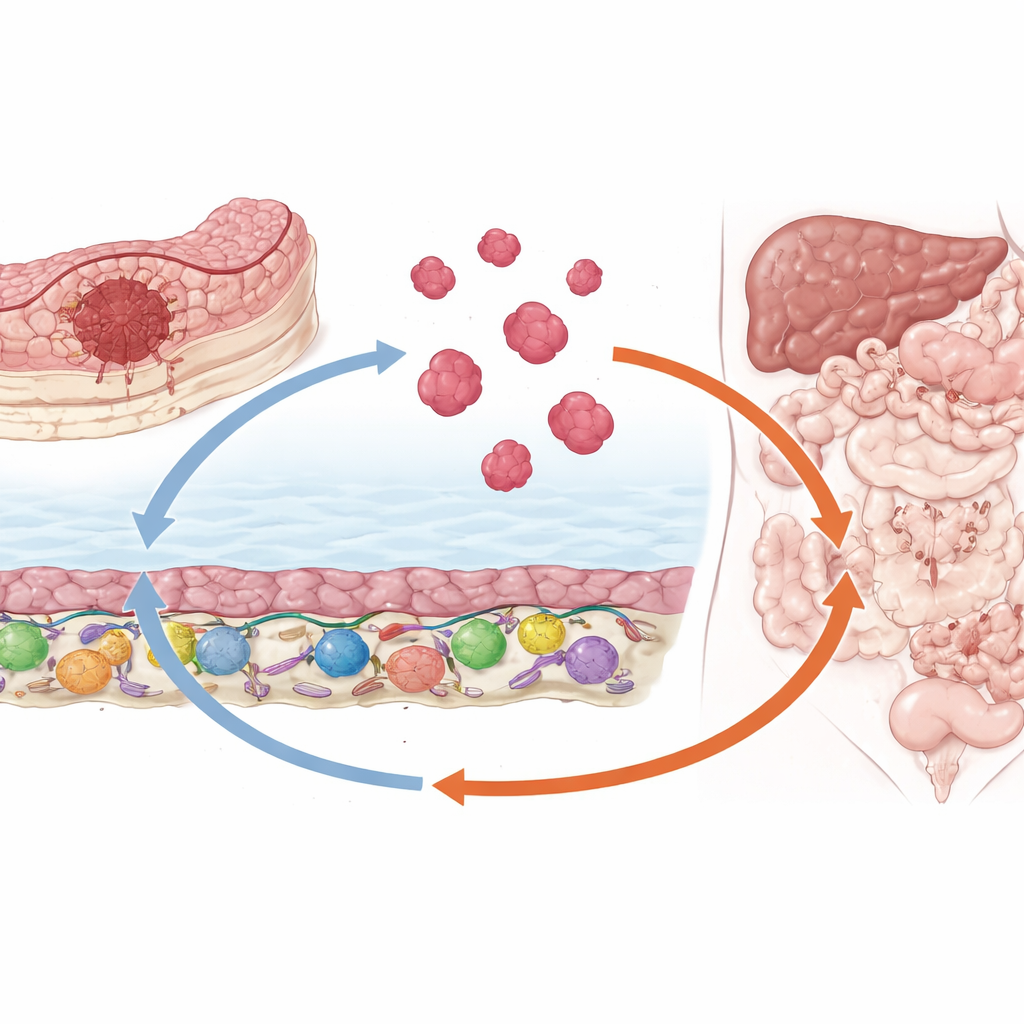
A three-part signal that switches tumors into high gear
Using patient samples, sophisticated gene‑reading methods, and mouse models, the researchers uncovered a three‑link signaling chain that appears to drive this dangerous pattern of spread. First, cells in the supporting tissue that lines the abdomen release a protein messenger called SPP1. Nearby colorectal cancer cells sense this signal and switch on a gene regulator named TWIST1, known for helping cells loosen their attachments and become more mobile. TWIST1 in turn activates production of another secreted protein, SPON2. Together, TWIST1 and SPON2 make cancer cells migrate more readily, invade surrounding tissue, and form three‑dimensional clusters that resemble the resistant “seeds” seen in patients’ abdominal cavities.
How cancer reshapes its neighborhood
The story does not stop at the cancer cell. SPON2, once secreted by tumor cells, feeds back onto the surrounding stromal cells—including mesothelial cells that form the slippery lining of the abdomen—and coaxes them into a more fibrous, cancer‑supporting state similar to activated fibroblasts. These remodeled stromal cells, in turn, produce even more SPP1, closing a self‑reinforcing loop between tumor and stroma. The loop thickens the tissue with collagen and other matrix components and floods the space with growth‑promoting signals, conditions that favor tumor expansion and the buildup of fluid (ascites) that often causes severe symptoms in patients.
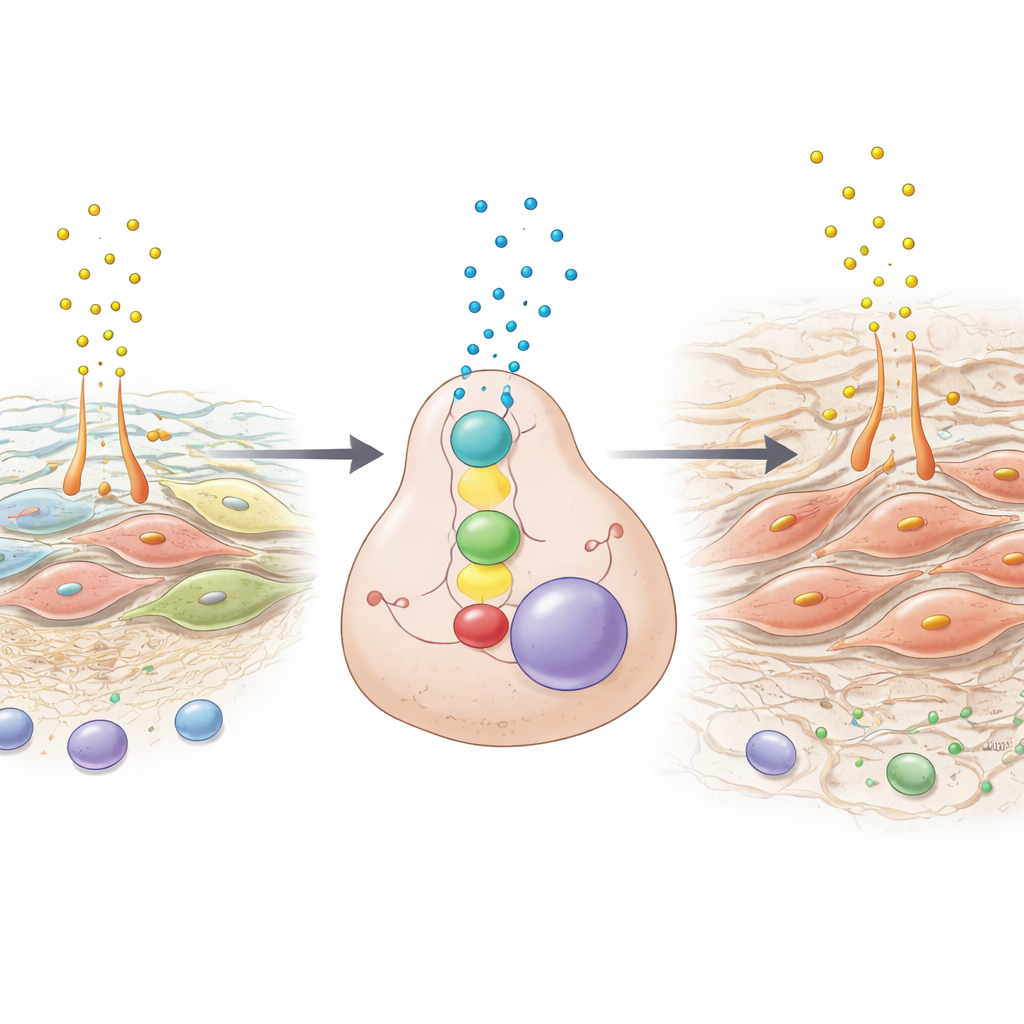
Blocking the loop slows tumors and invites immune attack
In mouse models that closely mimic human peritoneal metastasis, the team disabled individual parts of this circuit. Knocking out TWIST1 or SPON2 in tumor cells, or removing SPP1 from host stromal cells, sharply reduced the number and size of abdominal tumor deposits, lowered fluid accumulation, and thinned the fibrous stroma. At the same time, tumors became more accessible to the immune system: there was a marked increase in cancer‑killing CD8 T cells entering the tumor, without major changes in other immune cell types. Laboratory experiments showed that drugs which block the PI3K–AKT pathway—a key growth signal activated by SPP1—could also dampen TWIST1 and SPON2, hinting at possible ways existing agents might weaken this malignant dialogue.
What this could mean for future treatment
For a layperson, the essence of this work is that the authors have mapped a specific three‑step conversation—SPP1 to TWIST1 to SPON2—between colon cancer cells and the lining of the abdomen that helps the cancer spread, build scar‑like support tissue, shut out immune attack, and generate ascites. By disrupting any link in this chain, tumors in mice become smaller, less fibrotic, and more vulnerable to immune cells. This suggests that measuring these proteins could help identify high‑risk patients, and that therapies aimed at SPP1, TWIST1, SPON2, or the signaling routes they control might one day turn a nearly untreatable pattern of spread into a more manageable disease.
Citation: Zhou, Z., La Ferlita, A., Palavalli, M.H. et al. TWIST1 mediated transcriptional activation of SPON2 drives colorectal cancer peritoneal metastasis through stromal cell signaling network. Oncogene 45, 1613–1626 (2026). https://doi.org/10.1038/s41388-026-03743-7
Keywords: colorectal cancer, peritoneal metastasis, tumor microenvironment, TWIST1 SPON2 SPP1, cancer stroma