Clear Sky Science · en
High-fidelity bioassembly of organoids and spheroids using inertial droplet microfluidics for precision oncology and tumor microenvironment modeling
Why tiny 3D tissues matter for cancer care
Doctors and researchers increasingly rely on miniature versions of tumors and organs grown in the lab to predict how real patients will respond to treatment. These three-dimensional cell clusters, called spheroids and organoids, mimic the structure and behavior of tissues in the body far better than flat cell layers on a dish. Yet today’s methods for growing them are often slow, wasteful, and inconsistent, making it hard to scale them up for drug testing or personalized cancer therapy. This paper introduces a new platform, called OsciSphere, that aims to mass-produce highly uniform 3D mini-tissues quickly and reliably, using equipment that fits into standard laboratory workflows. 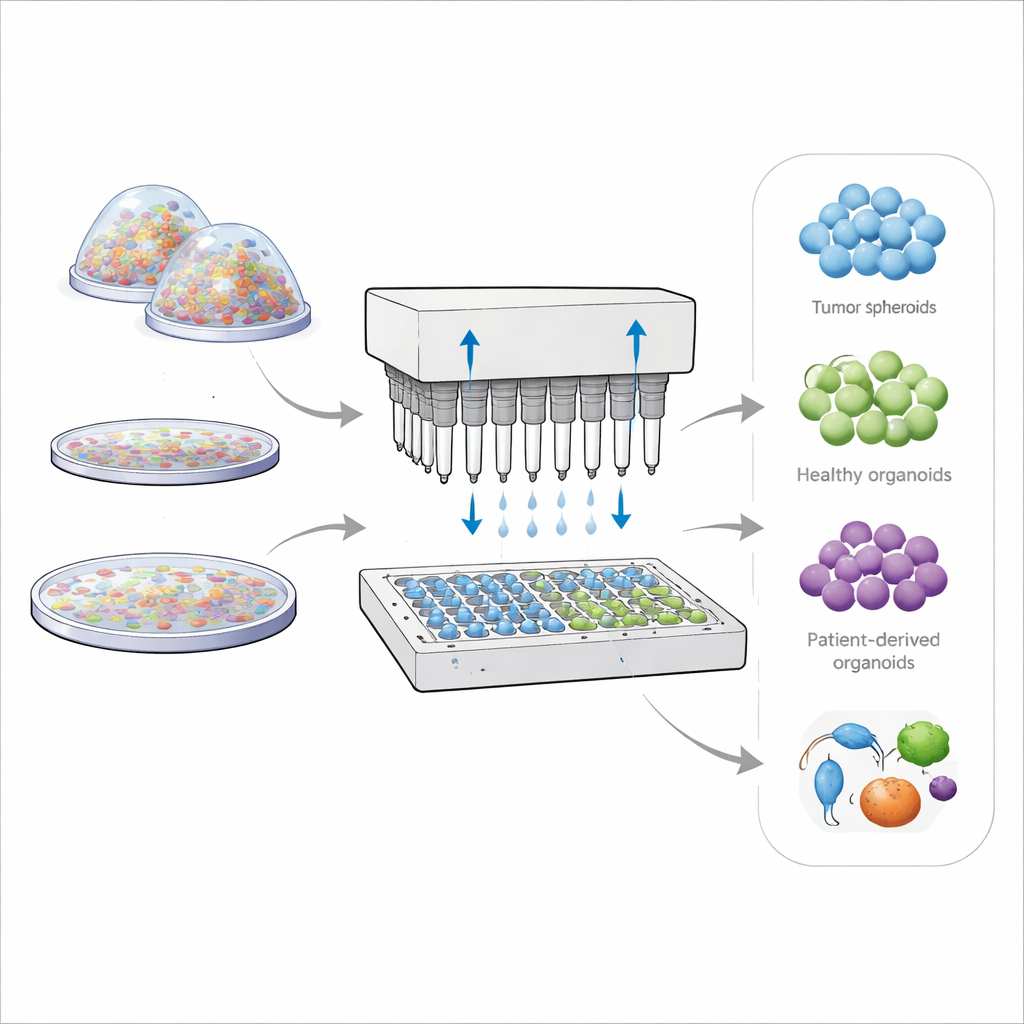
The problem with today’s 3D cell cultures
Most current approaches to 3D cultures fall into two camps: “scaffold-free” systems that let cells naturally clump together, and “scaffold-based” systems that embed them in a gel. Simpler methods like hanging drops or low-adhesion plates tend to give wildly different-sized clusters from well to well, which makes drug-response data noisy and hard to compare. Gel domes made from materials such as Matrigel support more lifelike tissue structures, but they are bulky and difficult to handle. Inside these large domes, oxygen and nutrients do not reach all cells equally, leading to dead cores and uneven growth that distort how the tissues respond to drugs.
A chip-free way to make uniform mini-tissues
OsciSphere solves these problems by turning thick, cell-laden gel into many identical tiny droplets directly inside common lab plates, without using complex microfluidic chips. The system uses an array of oscillating pipette tips that move back and forth at controlled speeds and amplitudes. This motion harnesses inertia rather than delicate surface forces to pinch off equal-sized gel droplets into an oil layer resting above culture medium. Temperature control keeps the gel liquid during this step, then warming solidifies the droplets into small spheres that are gently transferred into the medium below. Each solid gel sphere becomes a mini “home” where cells can assemble into uniform tumor spheroids or organoids, using only a fraction of the gel normally required. 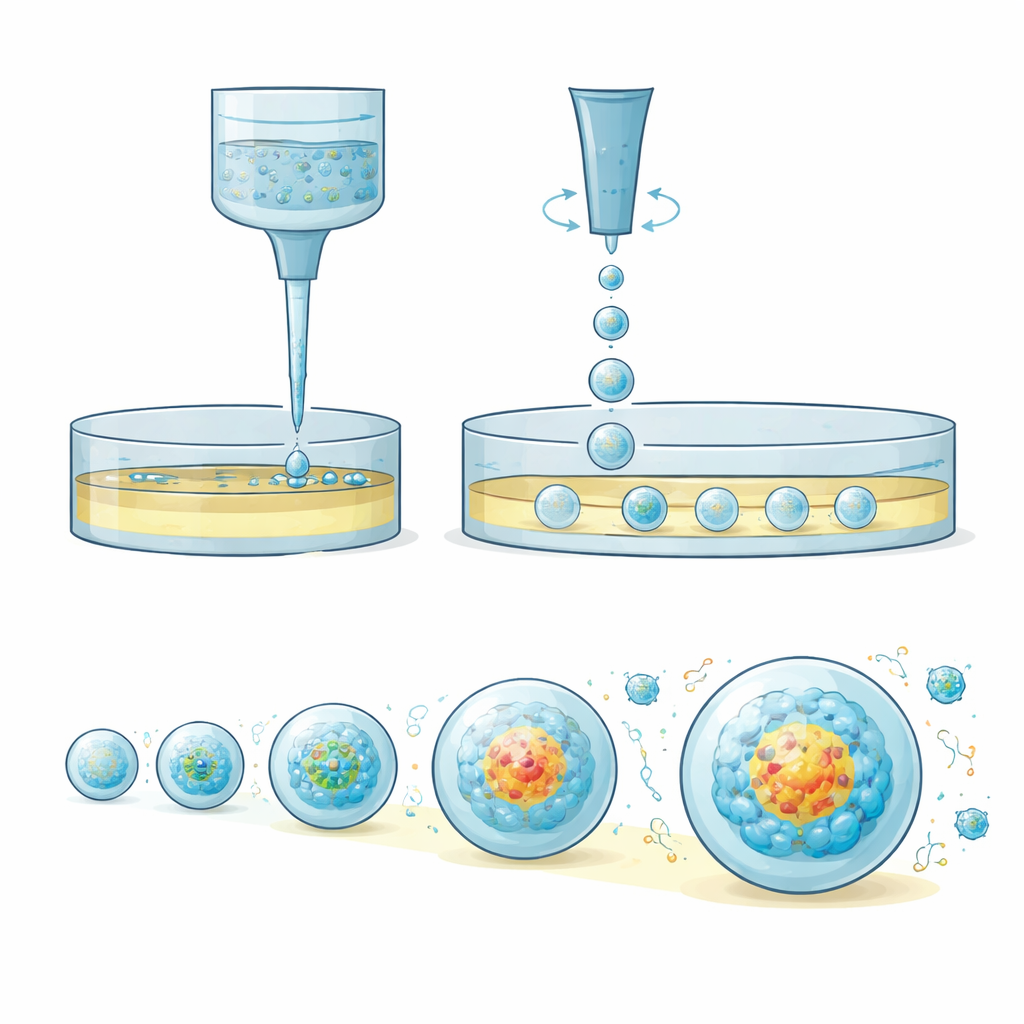
Building more realistic tumor and organ models
Because every droplet is nearly the same size and contains a precisely tuned number of cells, OsciSphere produces 3D tumor spheroids with tight control over diameter and shape. The authors show that these mini tumor models reproduce key hallmarks of real cancers: slower, more realistic growth rates, internal chemical gradients, stress signals, and gene programs associated with invasion and stem-like behavior. When used to grow miniaturized intestinal organoids, the small gel spheres avoid the diffusion bottlenecks of large domes. The organoids grow faster, show more advanced structures resembling the lining of the gut, and remain uniformly alive throughout, rather than developing dead centers that skew experimental results.
From drug tests to microbiome and immune studies
Once reliable arrays of mini-tissues are in place, the platform becomes a powerful testing engine. Tumor spheroids created with OsciSphere show drug resistance patterns that more closely match what doctors see in patients, while organoids in tiny gels are more honestly exposed to chemotherapy than those buried in thick domes. This allowed the team to rapidly screen combinations of standard colorectal cancer drugs and identify doses that work together effectively at relatively low concentrations. They went further by exposing tumor spheroids to secreted molecules from dozens of gut bacteria, quickly pinpointing species whose metabolites strongly suppress tumor growth and trigger cancer cell death pathways. Finally, they used patient-derived tumor organoids and a patient’s own immune cells to model how immune therapies work, showing that the small, accessible microspheres allow immune cells to infiltrate and become activated in ways that bulky domes do not.
How this could change precision oncology
In plain terms, OsciSphere offers a practical route to grow thousands of near-identical mini-tumors and mini-organs that behave more like tissues inside the body, using tools many laboratories already have. By shrinking the gel environment down to tiny, uniform spheres and tightly controlling how many cells go into each one, the platform improves the realism, consistency, and speed of 3D models used for cancer research. This, in turn, makes drug testing, microbiome studies, and immune-therapy evaluation more trustworthy and scalable. If widely adopted, such technology could help bring truly personalized treatment—where therapies are screened on a patient’s own miniature tumors before they are given in the clinic—closer to everyday cancer care.
Citation: Li, Y., Cao, Z., Xu, Y. et al. High-fidelity bioassembly of organoids and spheroids using inertial droplet microfluidics for precision oncology and tumor microenvironment modeling. Microsyst Nanoeng 12, 152 (2026). https://doi.org/10.1038/s41378-026-01244-x
Keywords: organoids, tumor spheroids, microfluidics, precision oncology, tumor microenvironment