Clear Sky Science · en
Artificial intelligence differentiates prefibrotic primary myelofibrosis with thrombocytosis from essential thrombocythemia using digitized bone marrow biopsy images
Why this matters for people with blood disorders
Doctors often struggle to tell apart two look-alike blood conditions that carry very different long-term risks. This study shows how artificial intelligence can read bone marrow images on a computer screen to separate these diseases more accurately, and even points to subtle tissue features that humans may be overlooking. For patients, that could eventually mean better diagnosis, clearer risk information, and more tailored treatment.
Two similar diseases with very different futures
Essential thrombocythemia and prefibrotic primary myelofibrosis are both rare bone marrow diseases that cause too many platelets, the blood cells that help with clotting. Under the microscope, their bone marrow samples can look quite similar, so even expert pathologists sometimes disagree on which diagnosis to give. The distinction is important because people with the prefibrotic form are more likely to develop severe scarring of the marrow, leukemia, and life-threatening complications than those with essential thrombocythemia. Current guidelines rely on experts visually judging cell shapes and subtle scarring, a process that is both subjective and time consuming.
Teaching a computer to read bone marrow slides
The researchers built a two-part artificial intelligence system that works directly from routinely stained bone marrow biopsy images. First, they used a powerful pre-trained model, originally exposed to tens of thousands of cancer slides, to extract detailed visual patterns from each small region of the biopsy. Then they applied an “attention” method that lets the algorithm assign higher importance to regions that most strongly support one diagnosis or the other. Trained on 200 Italian patients whose diagnoses had been carefully confirmed, and then tested on an independent group from a U.S. cancer center, the system distinguished the two diseases with high accuracy. It correctly separated prefibrotic primary myelofibrosis from essential thrombocythemia in more than nine out of ten patients in the validation group, with a particularly strong ability to avoid falsely labeling someone as having the higher-risk disease. 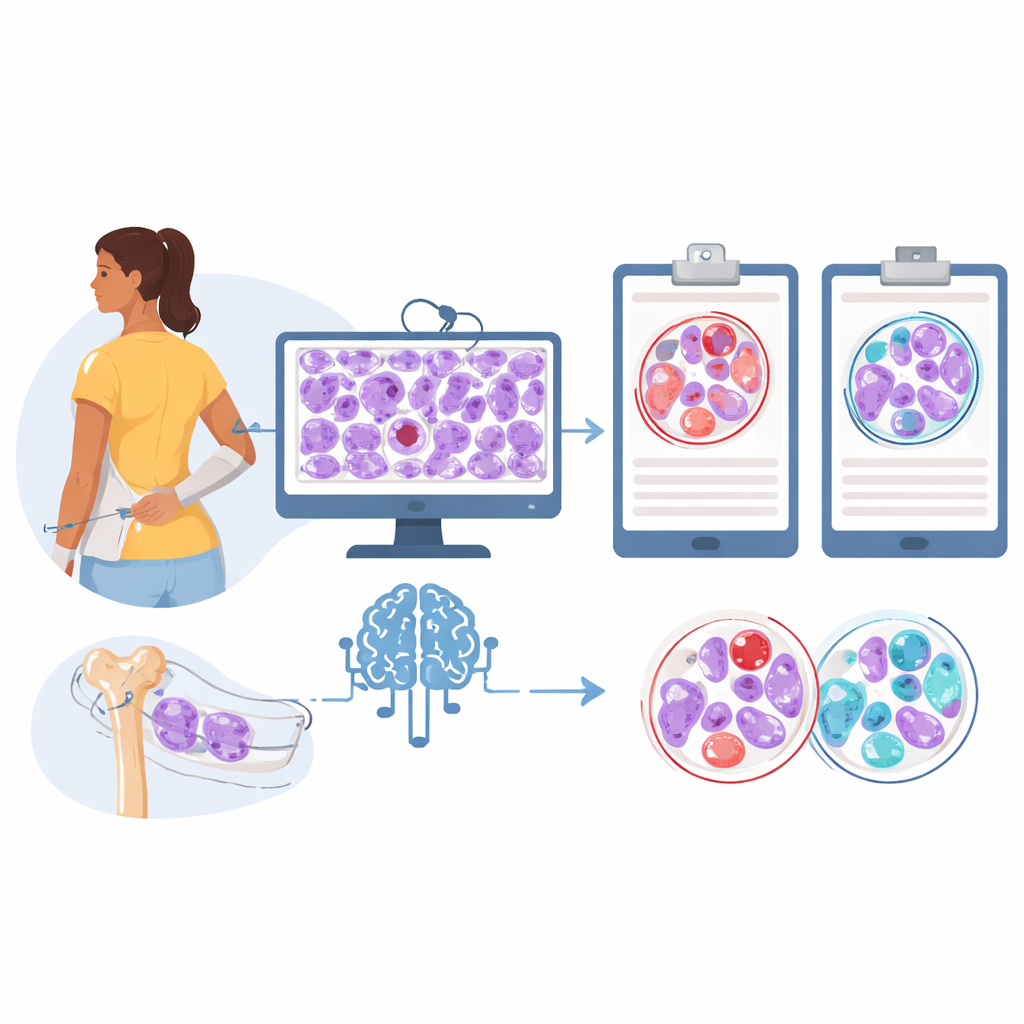
Using synthetic images to see what the AI sees
To understand what visual cues the model was relying on, the team added a generative component that can create realistic-looking bone marrow patches typical of each disease. They selected both real and computer-generated image tiles that the classifier handled confidently and asked three expert hematopathologists, working blindly, to label them. For real images, human experts agreed with the AI’s diagnosis most of the time when they were willing to commit to an answer. But for many synthetic images, especially those mimicking essential thrombocythemia, pathologists often disagreed with the algorithm or felt they could not safely choose a diagnosis, even though the AI classified these tiles correctly according to the known patient data.
A surprising role for fat versus cells in the marrow
The contrast between human and machine prompted a closer look at the tissue makeup in both real and generated images. Using another image analysis tool, the researchers measured how much of each tile was filled with fat spaces versus blood-forming cells. Across the board, samples linked to essential thrombocythemia contained more fat, while those linked to prefibrotic primary myelofibrosis were more densely packed with cells. This pattern appeared in both real patient images and computer-generated images, and it was even more exaggerated in the synthetic essential thrombocythemia slides. Heatmaps of the model’s “attention” also showed that the algorithm naturally focused on active blood-forming areas rather than bone or artifacts, suggesting it had learned biologically meaningful cues rather than shortcuts. 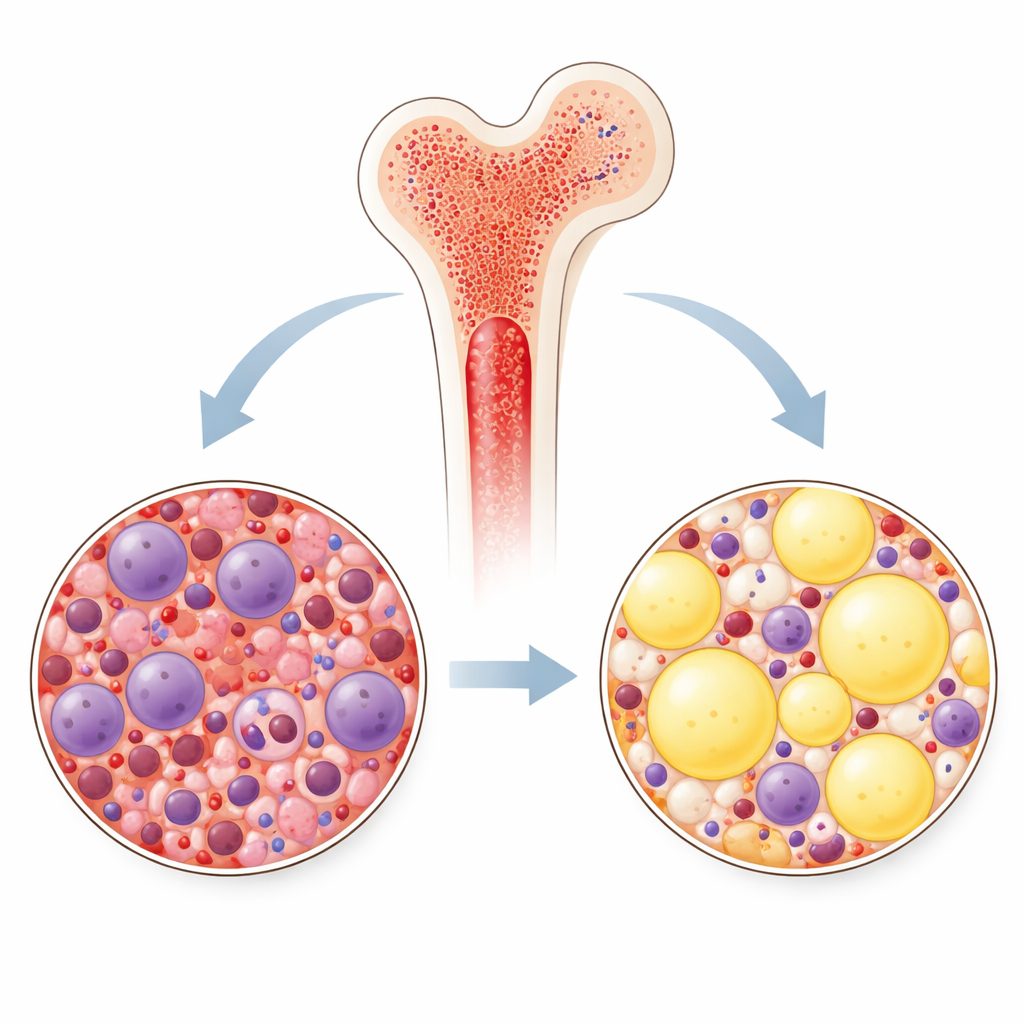
What this could mean for diagnosis and treatment
In practical terms, the study suggests that a carefully trained AI system can help pathologists separate these two look-alike marrow diseases using standard microscope slides, and can uncover useful tissue features, like the balance of fat and cells, that are not part of current checklists. The authors stress that their tool is not meant to replace expert review, especially given the modest number of patients studied so far. Instead, it could act as a second reader, especially in centers that see fewer such cases, and it might help refine which patients are enrolled in clinical trials by excluding those with the more aggressive prefibrotic disease. Over time, combining this kind of image-based AI with clinical and genetic data could support more confident, personalized care for people living with these rare blood disorders.
Citation: Srisuwananukorn, A., Loscocco, G.G., Dolezal, J.M. et al. Artificial intelligence differentiates prefibrotic primary myelofibrosis with thrombocytosis from essential thrombocythemia using digitized bone marrow biopsy images. Leukemia 40, 1018–1026 (2026). https://doi.org/10.1038/s41375-026-02893-7
Keywords: artificial intelligence, bone marrow biopsy, myeloproliferative neoplasms, essential thrombocythemia, prefibrotic myelofibrosis