Clear Sky Science · en
Effects of crosslinker role reversal on the properties of hydrosilylation-cured silicone elastomers
Why this study matters for everyday materials
Silicone rubbers are everywhere, from phone cases and kitchenware to medical devices and soft robots. Yet the way these materials are chemically “set” can quietly change how stiff, stretchy, or long lasting they are. This article explores what happens when chemists swap the roles of two key ingredients during curing and shows how that simple change reshapes the internal structure and performance of silicone elastomers.
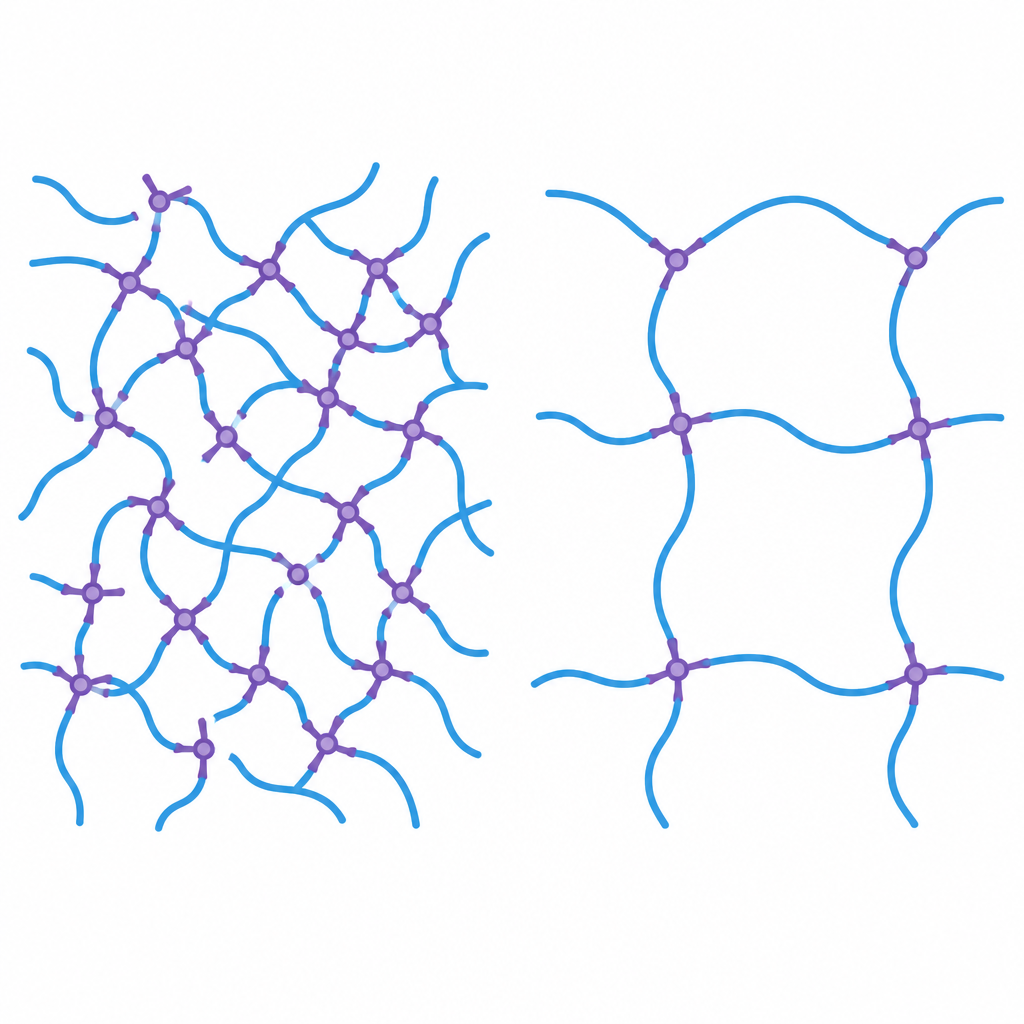
Two ways to tie silicone chains together
The researchers focus on polydimethylsiloxane, or PDMS, the workhorse silicone used in many products because it stays flexible, tolerates heat and cold, and is gentle to living tissue. To turn liquid PDMS into rubber, small molecules called crosslinkers connect long polymer chains into a network. In the “traditional” recipe, the long chains carry vinyl groups while the crosslinkers carry reactive silicon hydride groups. The team asked what would happen if they reversed these roles, so the long chains carry hydride groups and the crosslinkers carry vinyl groups, and whether this would change how the network forms and behaves.
Peering into the building blocks
Using advanced nuclear magnetic resonance (NMR) methods, the authors first mapped the fine structure of the two crosslinkers. They found that the hydride crosslinker tends to have its reactive groups clustered in short blocks along the chain, while the vinyl crosslinker has its reactive groups more evenly alternated with nonreactive units. This subtle difference turns out to matter a lot. Blocky hydride segments encourage regions of very dense linking, while a more alternating vinyl layout promotes a smoother, more uniform spacing of links once the network forms.
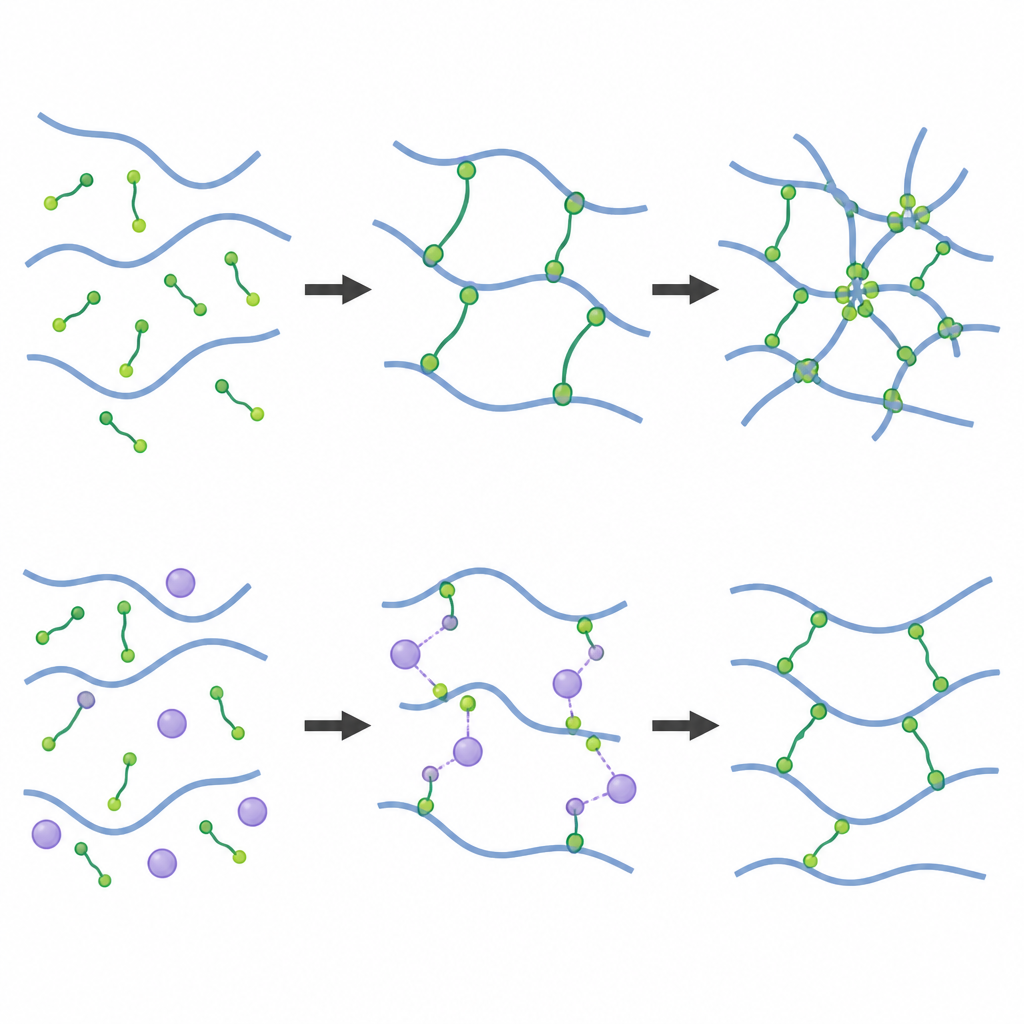
How curing speed shapes the internal network
The team then tracked how each system cures over time using rheology, which measures how a material resists flow, and in situ NMR, which follows the actual chemical groups as they react. When the hydride crosslinker is used, the mixture quickly turns from a liquid into a gel in a single step, whether at room temperature or higher heat. The very reactive hydride groups not only link to vinyl groups on polymer chains but also start reacting with each other. This creates a dense network with extra connections and patches of high stiffness. In contrast, when the vinyl crosslinker is used, the curing is slower and proceeds in stages. For a while, the platinum catalyst sits on nearby vinyl groups and holds them back, so the system thickens slowly before it finally links up into a rubbery solid.
What this means for softness, stretch, and uniformity
Mechanical tests showed that hydride-crosslinked elastomers are stiffer, stronger, and swell less in solvent, all signs of a tightly tied network. They also contain fewer loose chains that can be washed out. However, solid-state NMR and swelling experiments revealed that this network is more heterogeneous, with clusters of heavy crosslinking. In the vinyl-crosslinked elastomers, the opposite trend appears: the rubber is softer and more stretchable, with longer segments between crosslinks and a more uniform internal structure. Interestingly, adding more vinyl crosslinker does not significantly increase stiffness once all the reactive ends on the long chains are used, whereas adding more hydride crosslinker continues to change both density and uniformity of the network.
Implications for designing better silicones
To a non-specialist, the key message is that “who carries what” in the curing recipe strongly controls both how fast a silicone sets and what kind of internal web it forms. When the small crosslinker carries the very reactive hydride groups, the result is a firm, somewhat patchy network that can be tuned by changing how much crosslinker is used. When those hydride groups are moved to the long chains and the crosslinker carries vinyl groups, curing slows down and produces a gentler, more even network whose properties depend less on crosslinker excess. By understanding this role reversal, manufacturers can better choose curing systems that favor either toughness and rigidity or softness and uniform stretch, depending on the demands of applications from sealants to flexible electronics.
Citation: Yu, L., Enemark-Rasmussen, K., Madsen, F.B. et al. Effects of crosslinker role reversal on the properties of hydrosilylation-cured silicone elastomers. npj Soft Matter 2, 12 (2026). https://doi.org/10.1038/s44431-026-00023-y
Keywords: silicone elastomers, PDMS, hydrosilylation, polymer networks, crosslinking chemistry