Clear Sky Science · en
The optimization of neuroprosthetic interfaces relying on biophysical and surrogate digital twins
Rewiring the Body with Smarter Implants
From easing chronic pain to restoring sight and movement after paralysis, tiny electrical devices that talk to our nerves are quietly transforming medicine. Yet today, many of these neuroprosthetic systems are tuned by trial and error at the bedside. This article explains how "digital twins"—computer replicas of a patient’s nerves and brain—could turn that guesswork into a more precise science, making implants safer, more effective, and easier to tailor to each individual.
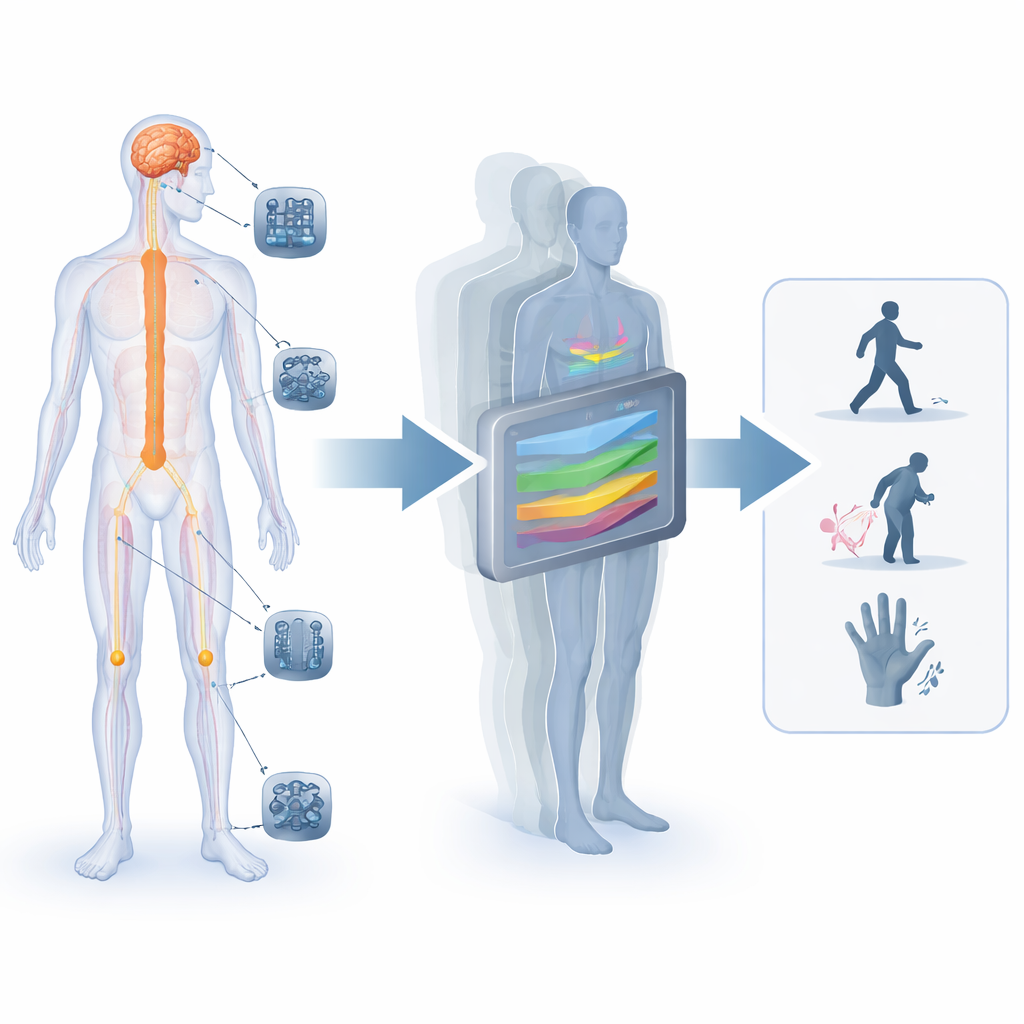
How Electricity Talks to Nerves
Our nervous system is an electrical communication network linking brain, spinal cord, organs, and limbs. Modern implants tap into this network in many ways: spinal cord stimulators to reduce pain or help people with spinal injuries walk; deep brain stimulators to ease Parkinson’s symptoms; retinal and optic-nerve implants to restore some vision; and devices on peripheral nerves to return touch or control muscles. Each technology must strike a delicate balance: strong enough signals to trigger useful sensations or movements, but not so strong or poorly aimed that they cause side effects, from hoarseness during vagus nerve stimulation to uncomfortable tingling during spinal stimulation.
Building a Virtual Copy of the Patient
Because it is impossible—and unethical—to test every electrode design or stimulation pattern directly in people, researchers are turning to detailed computer models. These "biophysical digital twins" use medical images and anatomical data to reconstruct the patient’s nerves, spinal cord, brain, or retina in three dimensions. Physics-based equations then estimate how electrical current flows through tissue, how individual nerve fibers respond, and how that activity might translate into a sensation, a muscle contraction, or a change in organ function. Although the same general framework is used across body regions, it can be adjusted to capture the unique structures of a peripheral nerve, spinal cord, retina, or deep brain nucleus.
From Simulation to Optimization
Once a digital twin can predict how nerves react to stimulation, it becomes a testing ground for improvement. Instead of manually trying a handful of settings, computer algorithms can automatically search through thousands of possibilities, adjusting controllable knobs such as which electrodes are active, how strong the pulses are, or how often they are delivered. The goal might be to recruit as many helpful fibers as possible while avoiding those that trigger pain or unwanted movements, or to shape an electric field that is tightly focused on a target brain region. In some cases, these optimization routines also tune hidden aspects of the model itself—such as the internal wiring of a nerve bundle—so that predicted responses match what doctors observe in the clinic.
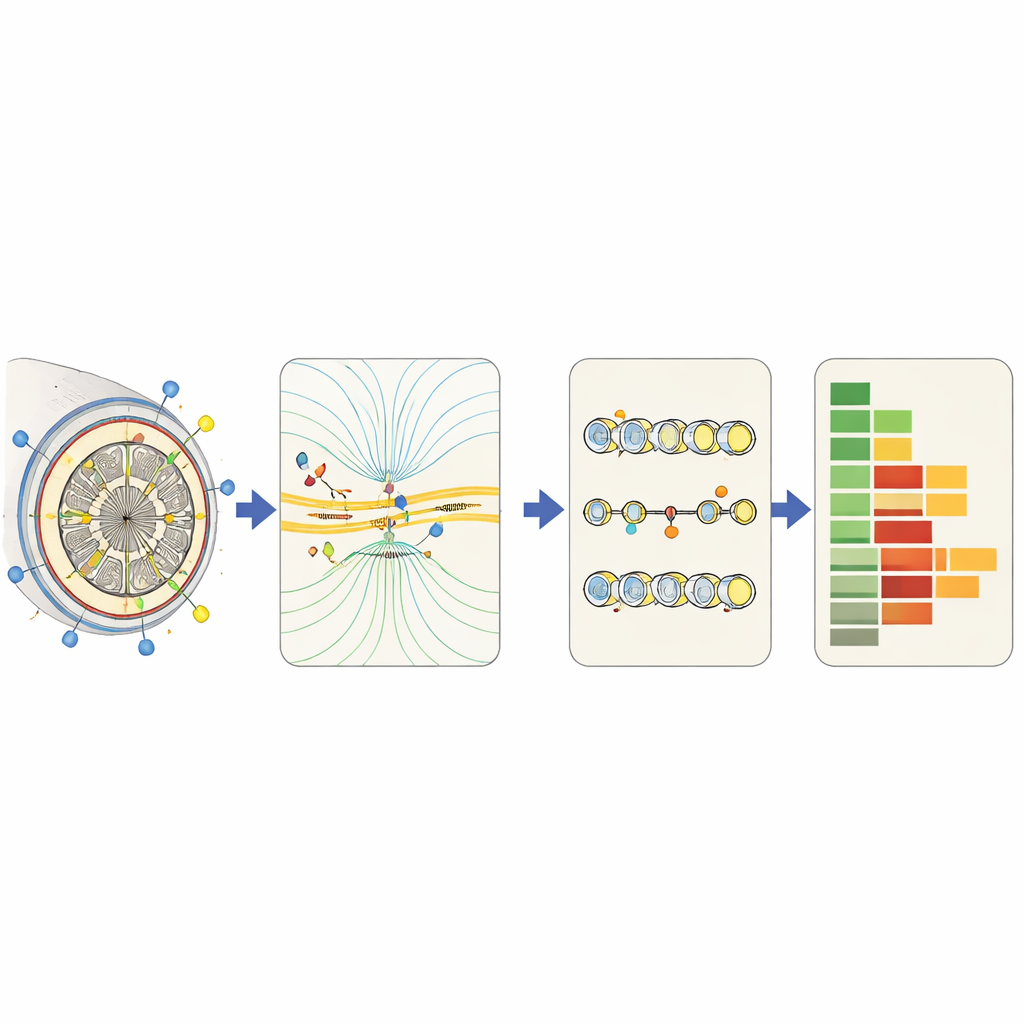
Making Heavy Computations Fast Enough
Highly detailed simulations can be slow, especially when each change in settings requires computing electrical fields and the firing of thousands of nerve fibers. To make the process practical, researchers are building "surrogate" models: streamlined predictors that learn, from many full simulations, how patterns of electric field relate to nerve activity. Some are based on simplified physics; others use machine learning to classify whether a fiber will fire, or to estimate its firing rate, from a compact set of features. Coarser-grained approaches track average activity in regions of tissue or across the whole brain, which is less precise but fast enough to explore large design spaces and to link stimulation to high-level effects such as engaging specific brain circuits.
Letting Implants Learn from Data
Even the best digital twin is only an approximation, and real patients change over time as tissues heal, adapt, or scar. To keep up, many teams complement model-based methods with "data-driven" optimization that learns directly from the body’s responses. Here, the implant tries different settings while sensors record nerve signals, muscle activity, movement, heart rate, or even patient-reported sensations. Statistical algorithms such as Bayesian optimization then propose the next set of settings to test, balancing cautious exploration with the desire to quickly reach effective, comfortable stimulation. In the future, biophysical models and data-driven learners may be combined, with the model providing smart starting points and the data-driven layer fine-tuning them to each individual.
From Virtual Lab to Everyday Medicine
The article concludes that digital twins and their faster surrogate cousins could profoundly change how neuroprosthetic devices are designed and used. By predicting outcomes before surgery, narrowing the range of safe and promising settings, and continually adapting to the patient, these tools could reduce reliance on animal testing and lengthy manual tuning sessions. Significant hurdles remain—regulatory approval, integration into busy clinics, and the need to predict meaningful outcomes like movement and sensation rather than just nerve activation. Still, the authors argue that, if developed and validated transparently, computational modeling will become a key partner to clinicians, helping neuroprosthetic technologies deliver more reliable, personalized benefits to people living with neurological disease and injury.
Citation: Verardo, C., Fossati, V., Toni, L. et al. The optimization of neuroprosthetic interfaces relying on biophysical and surrogate digital twins. npj Biomed. Innov. 3, 28 (2026). https://doi.org/10.1038/s44385-026-00076-8
Keywords: neuroprosthetics, digital twins, neural stimulation, computational modeling, Bayesian optimization